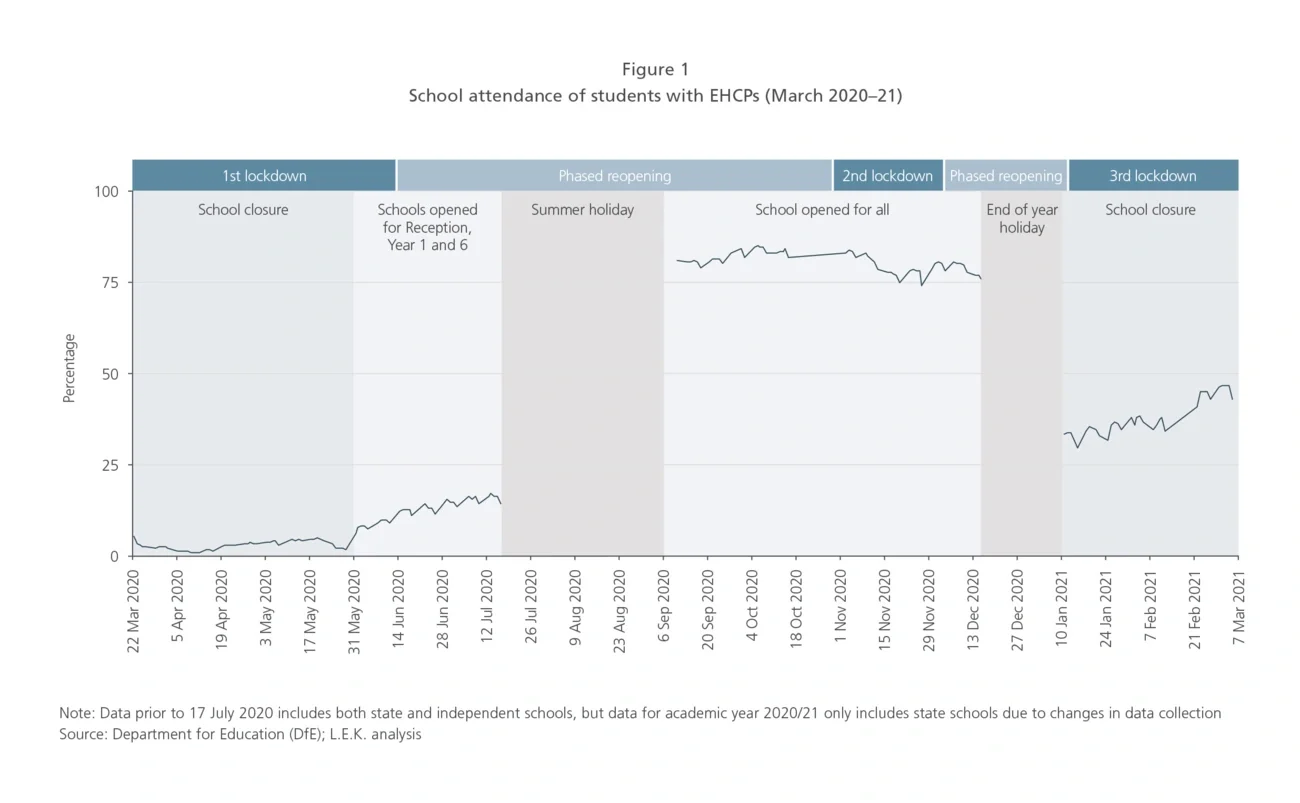
There is a backlog of SEN assessments
Well-established processes are in place in schools and local authorities (LAs) to identify and help children with special education needs. Teachers and social workers request EHCP assessments from commissioners, develop and formalise plans, and provide continuing support for children. But this has been severely disrupted by the pandemic.
In many cases, systems came to a grinding halt, creating a significant backlog of children in need (see Figure 2). Compared to the 2018-19 average, the number of new EHCP requests dropped 25% during March-July 2020, 16% on an annualised basis — equating to around 10,000 children not getting an EHCP in a timely manner.2 The number is increasing, and we expect to see a wave of demand for places at special needs schools. We anticipate that up to 1,000 children from the current backlog will benefit from a place in the independent sector.
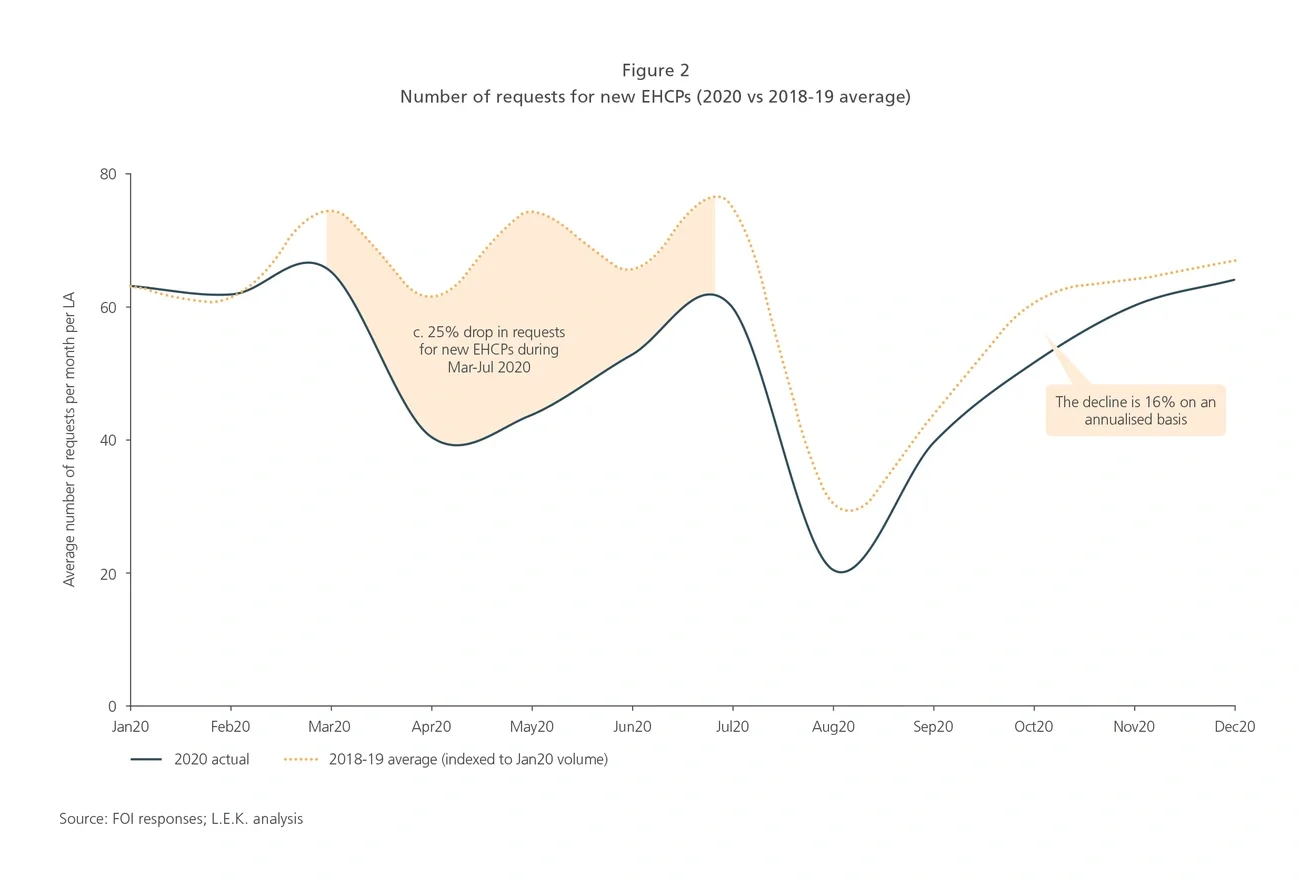
Reviews of existing EHCPs have also been delayed as LAs have struggled to conduct routine annual assessments during the pandemic, even remotely. When reviews did take place and a child’s needs assessed changed, requiring a change of school, parents and commissioners tended to keep the child in their existing school to avoid additional disruptions for them. Whilst this continuity offers some temporary relief, the child’s needs could escalate if the existing school cannot deliver the additional resources needed due to a lack of experience, expertise or bandwidth. The backlog of existing plan reviews is an extra burden for commissioners, and some of these delayed reviews will result in referrals to independent special needs schools rather than state operated schools — i.e. adding to demand from the identified backlog.
What next?
However the build-up of children with EHCP needs is not the pandemic’s only major impact on special education. The pandemic has triggered deeper-rooted and longer-lasting dynamics — e.g. slower speech, delayed social and emotional development, mental health of young children, and loss of learning in the early years. The impact of these factors will not come as an immediate wave; indeed, these will more likely lead to a stronger and faster rising tide that needs to be managed in the future. In many regions of the UK, independent providers have become vital partners to commissioners to address the needs of youngsters, and those providers’ services will be in high demand — especially for those with high-quality propositions.
Endnotes
1SEN support for those of lower complexity and EHCPs for those with more significant needs
2Department for Education (DfE ); FOI responses; L.E.K. analysis
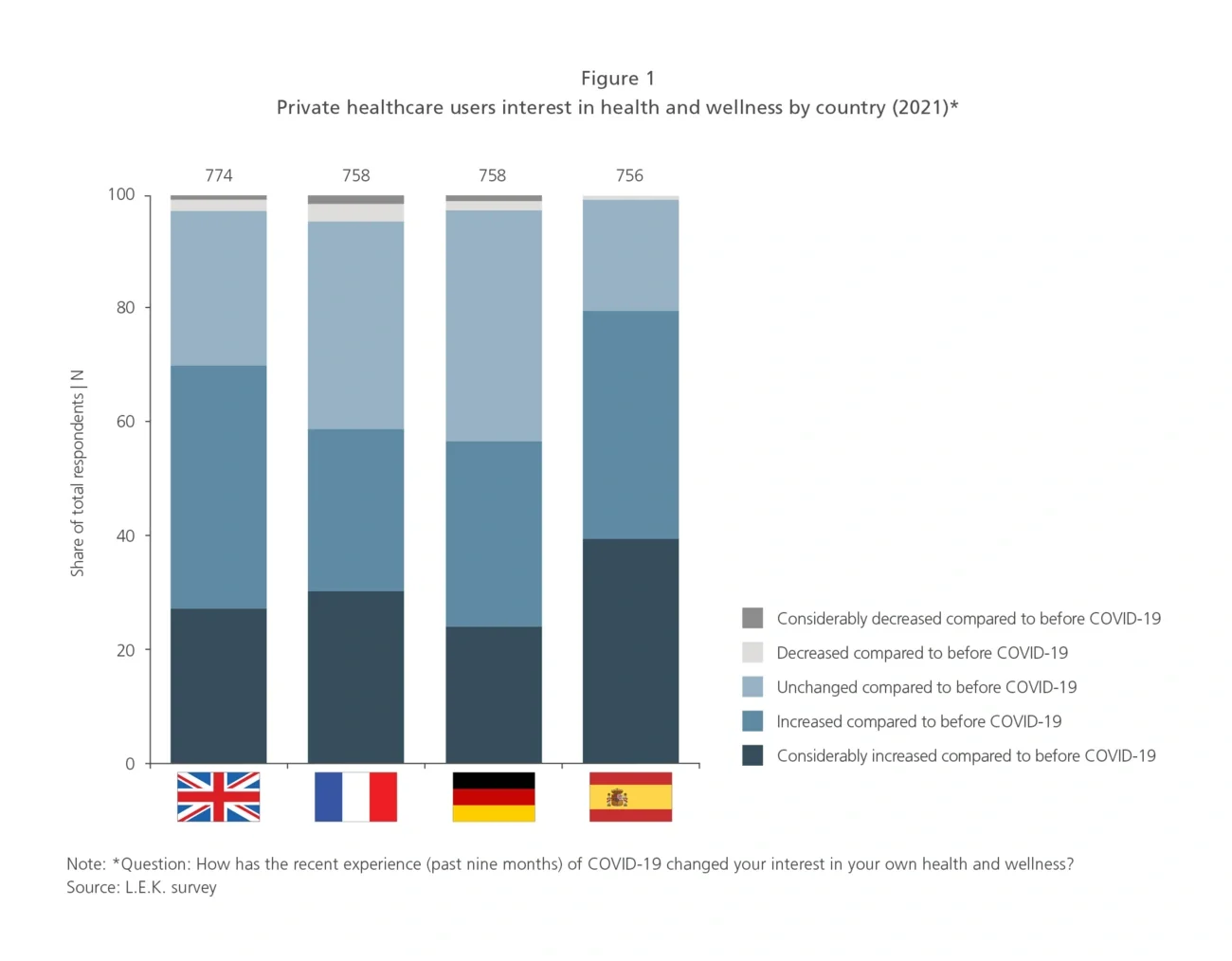
As a result, private healthcare users expect to resume their pre-pandemic levels of spending on their health or increase it in 2021 (UK, 85%; France, 60%; Germany, 60%; Spain, 90% — see Figure 2).
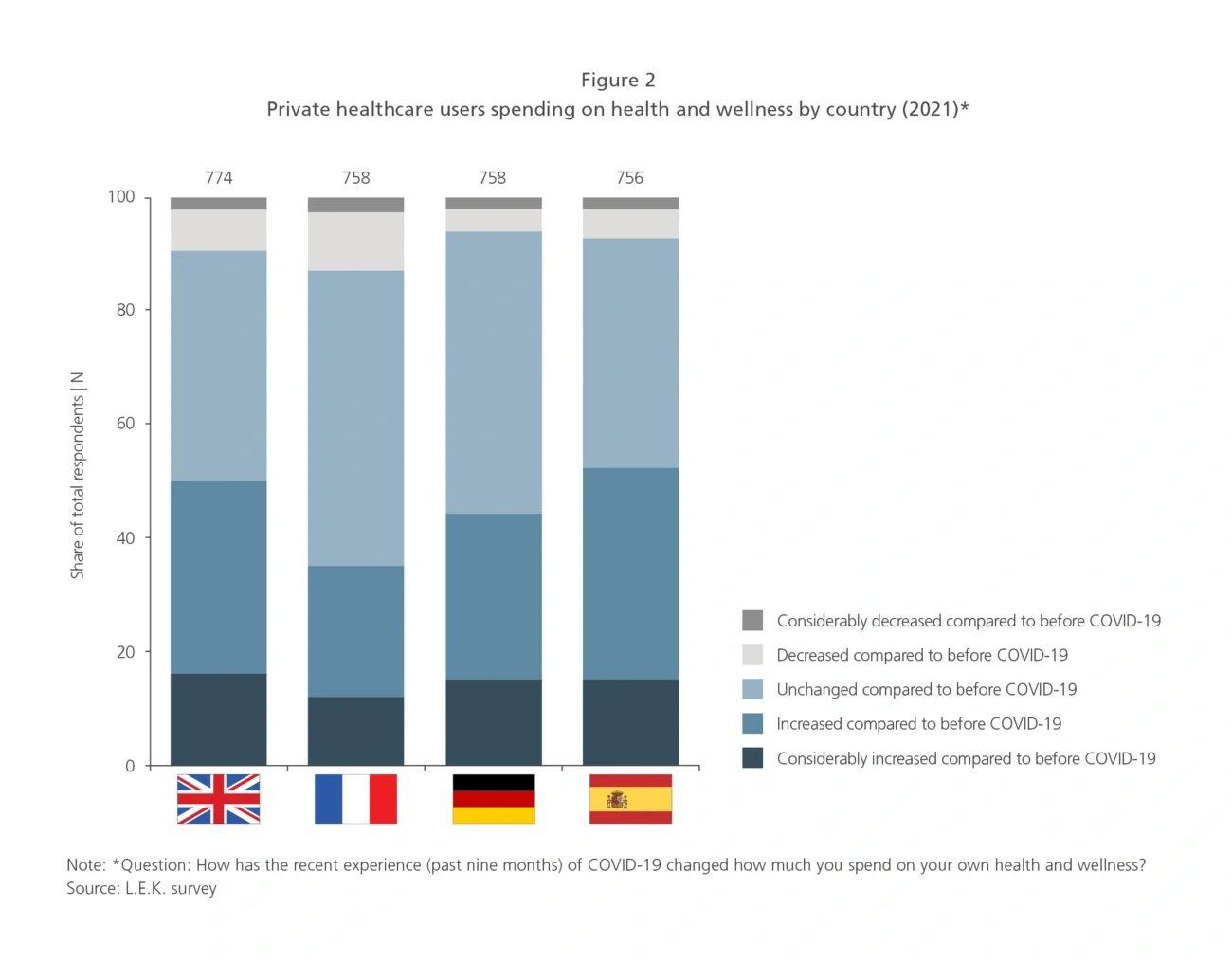
The most buoyant market is the UK, where spending is set to increase across all 15 of the private healthcare sub-sectors reviewed, with the top three areas being cosmetic skin treatments, orthopaedics and laser ophthalmology — see Figure 3. Growth is expected in all sub-sectors in Germany and Spain, apart from laser ophthalmology (Germany) and plastic surgery (Spain), which will remain static. But the outlook is more robust in Spain, with generally higher levels of spending anticipated, especially in laser ophthalmology, dental orthodontics and mental health treatment. France is the weakest market, as six sectors are set to see a decline in spending.
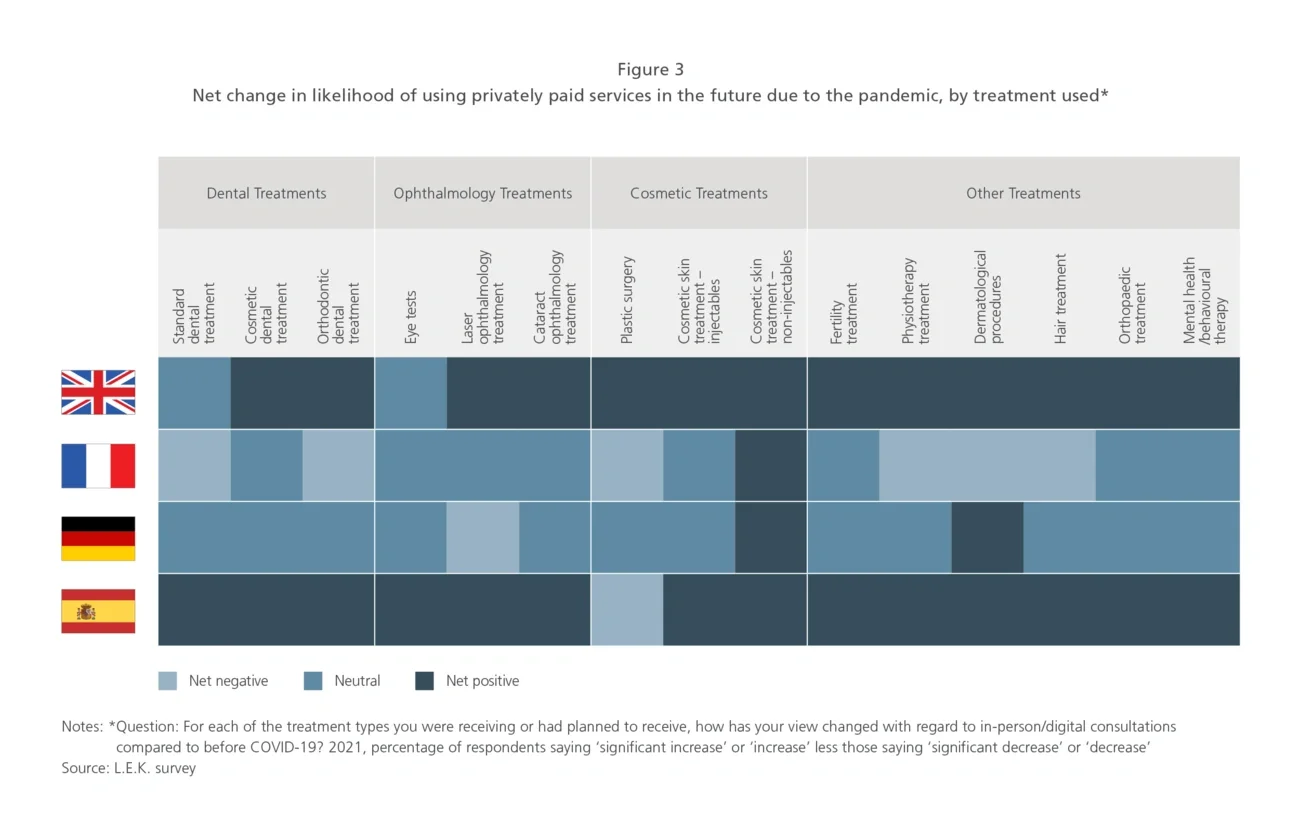
As private healthcare services companies forecast and plan their business post-pandemic, they should prepare for returning confidence and growth opportunities. They will need to build into their strategy an understanding of how the growth outlook varies by country and sub-sector. Patient familiarity with digital consultations as a result of the pandemic offers healthcare providers a further opportunity to enhance the patient experience through digital channels and improve margins by being able to undertake more consultations.
10-year Digital Transformation Roadmap for Leading Metals Producer and Manufacturer
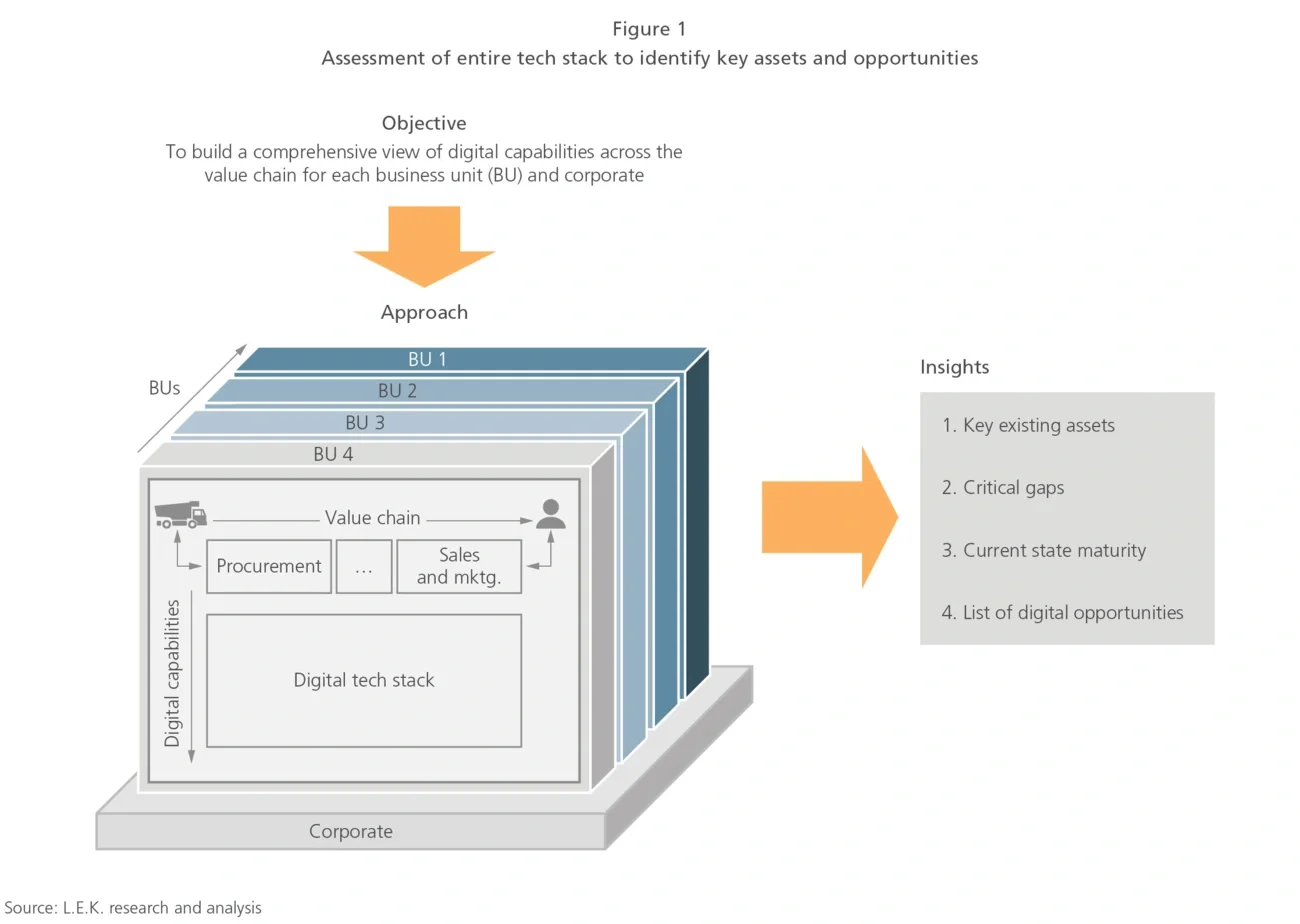
- Art of the possible: Highlighted dozens of relevant and potentially high-impact uses of technology to illustrate the range of outcomes that could be achieved through digital transformation initiatives
- Strategic imperatives: Assessed the client’s strategy and analyzed the competitive set to isolate the key digital capabilities the client would need to develop in order to maintain and enhance its market position
- Digital roadmap development: Built detailed 10-year digital transformation roadmaps for four BUs, totaling over 50 unique initiatives to meet the client’s strategic imperatives for continued profitable growth
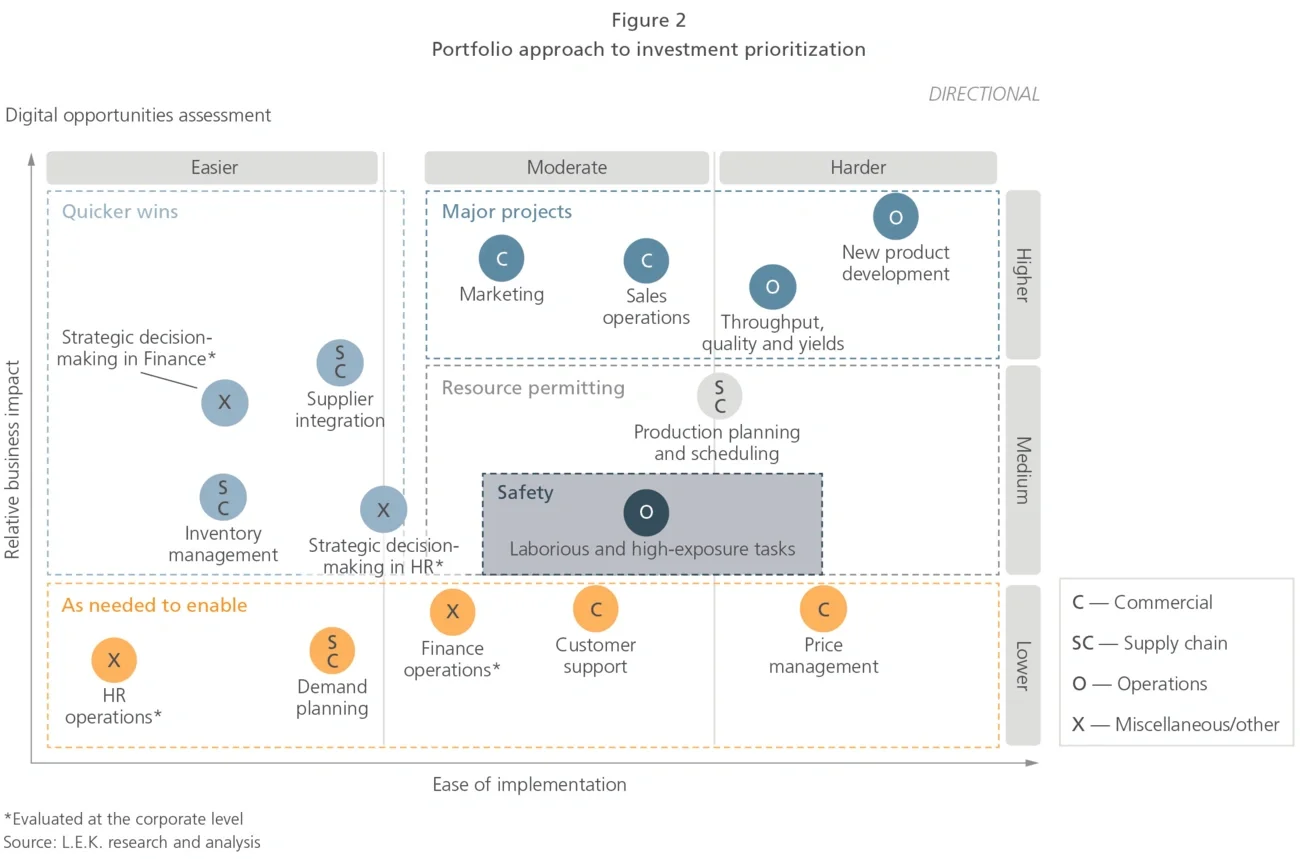
- Financial analysis: Developed a 10-year investment plan with expected timelines and ROIs for each initiative, taking a portfolio approach to investment prioritization using medium-term initiatives to drive momentum and fund the long term (see Figure 2)
- Key success factors: Recommended necessary, urgent changes required to the organizational design in order to execute the digitization roadmap
Results
The client now has a clear vision of more than 30 prioritized digital initiatives and recommendations for critical organizational changes, sequenced to account for technical dependencies, level of impact and ease/time of implementation. In total, the roadmap has the client poised to unlock hundreds of millions of dollars in cumulative EBITDA benefit over the next 10 years.
The release of the Williams-Shapps Plan for Rail (“the Plan”) is an important moment for the UK rail industry as it seeks to address long-standing and complex challenges including: a desire for more industry-wide strategic coordination, the balance of returns and risks between the public purse and private sectors, and the complicated incentives across the contractual boundary between track and train management.
Whilst stakeholders face substantial change, it is essential that the new industry model proposed in the Plan be able to build on the best of privatisation over the last 20 years and its successes, including increasing passenger numbers and revenue, launching new routes and services, and the significant rolling stock and infrastructure.
We set out below the opportunities facing industry stakeholders, the questions that must be addressed through implementation to ensure the best of privatisation is built on, and three near-term considerations to ensure the Plan is a success.
The impacts facing stakeholders
As with any transformation of this ambition and scale, the creation of Great British Railways (GBR) will provide significant opportunities and challenges for the industry’s stakeholders.
- The Department for Transport, Network Rail and the Rail Delivery Group will need significant momentum to face the scale of change required, as they seek to combine their skills and experiences to form a new organisation that is more than the sum of its parts. This new organisation will have an important mandate to create an industry strategic and financial blueprint which captures the vision for GBR as set out in the Plan.
- Some operator groups (i.e. owners of train operating companies) may prefer lower risk Passenger Service Contracts (PSCs) and must find ways to create value without direct influence over many commercial levers. Operator groups who left the market in the past will need to consider whether the new contracts give them an opportunity to return to GBR.
- Rolling stock leasing companies and manufacturers will take comfort that their business models are not required to change, but will still need to consider how best to accelerate, design and finance new, greener rolling stock to respond to decarbonisation targets.
- The Plan sets out that the ownership, planning and operation of infrastructure will be centralised within GBR, with the Office of Rail and Road taking on an independent reviewer role and ensuring GBR remains accountable for performance and efficiency. This approach creates significant strategic and financial planning opportunities to target infrastructure investment where there might be greatest returns for passengers, as well as ensuring all infrastructure is of at least a minimum grade across the country.
- Ticket retailing will have a new, state-owned competitor, which will undoubtedly disrupt the third-party retailer market. Pricing must be fair and competitive and incentives provided to ensure continued private sector participation and innovation.
- Green and digital suppliers should search for new opportunities to support major infrastructure delivery across the network, preparing the railways for a low-carbon and digital future.
- Taxpayers and passengers will hope the Plan delivers improvements in value for money from the rail network. For GBR to deliver this, it will need to be structured and incentivised to optimise the offering to customers and to drive growth in revenue — while also delivering reliability, investment and safety across the railway system.
Learning from privatisation
To be a success, it is crucial that the Plan builds on the learnings and benefits of the privatised model of the last 20 years.
Key issues to consider are:
- How will GBR’s success be defined and measured to incentivise improvements in customer proposition, reliability, safety and industry profitability, when direct revenue growth incentives are reduced?
- What is the best way for GBR to continue to attract and retain the expertise and capital that has driven significant industry improvements during privatisation?
- How can GBR best build a customer-centric organisation with the appropriate skills, expertise and incentives to put customer needs above those of infrastructure management?
- How can the opportunity presented by combining track and train under one organisation best be used to address the long-standing issue of congestion across the network, beyond simply minimising the costs of delay attribution?
- How can local and regional interests be fully reflected within a more centralised industry structure?
- How should the industry re-prioritise capital investment in light of a sizeable revenue gap that is likely to persist post-pandemic when compared with 2019?
- How should the industry adapt to the inevitable lower levels of demand over the medium term as the nation recovers from the pandemic? What will be the new normal level of demand reflecting changing travelling patterns?
- How can the rail industry best interact with other modes of transport to ensure a seamless passenger experience not only within the network, but also for door-to-door journeys?
Three near-term actions for success
The Williams-Shapps Plan for Rail sets out an ambitious and challenging vision for the industry, and much work is required to ensure the Plan’s vision is implemented successfully. We see three near-term actions necessary to ensure this:
- Current industry stakeholders must align to ensure there is no knowledge lost in this transformational change to the structure and governance of the industry.
- The design of GBR must integrate carefully planned new incentive structures and KPI performance scorecards that put customers first.
- The future industry structure must be designed to accommodate private sector participation beyond operating train services in order to enable and encourage innovation, development of expertise and capital investment.
We would welcome the opportunity to discuss the Plan and its impact. L.E.K. Consulting supports the rail industry in corporate strategy, business planning, revenue forecasting, organisational change, policy development and M&A. Please contact the UK Transport Team.
We look forward to working with stakeholders to make the vision of the Plan a reality.
‘An Opportunity for Change in UK Rail’ was written by Andrew Allum, Partner; John Goddard, Partner; Becrom Basu, Partner; Stuart Robertson, Principal and Emma Edgar, Principal, at L.E.K Consulting. All authors are based in London.

Unlocking the Benefits of Patient-Centric Healthcare
Many organizations have adopted patient-centric healthcare (PCH) over the years, although some have been challenged to deliver on the full potential of their aspiration. This special report highlights the best-practice uses of PCH and how benefits for the industry and patients emphasize the value this rapidly evolving healthcare sector approach provides.

Asia-Pacific in the Eye of AMR Storm
Preparedness, Policy And Patients – Learning From The Pandemic
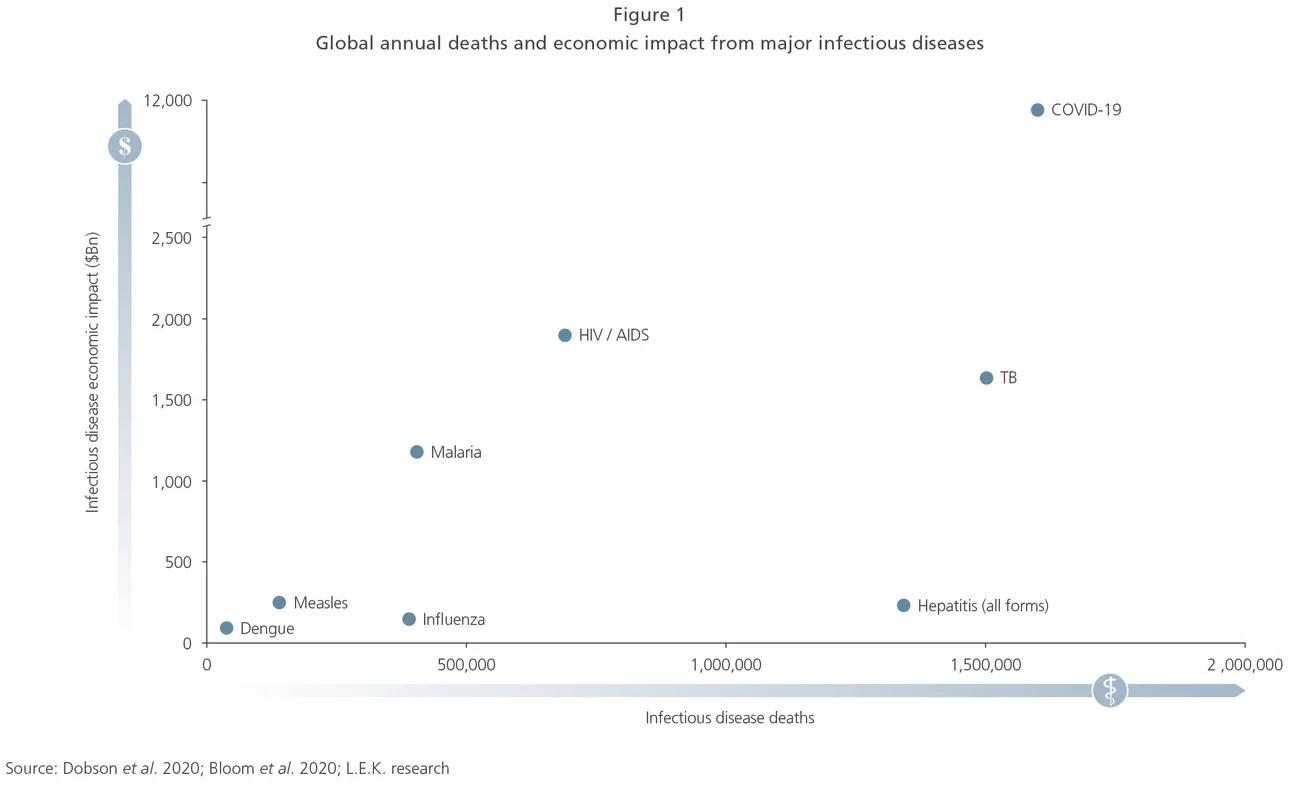
According to a recent analysis by the National Bureau of Economic Research, HIV, tuberculosis and malaria cause millions of deaths and an estimated $4.5trn in economic damage per year, greater than Germany’s total annual economic output. According to data from a 2019 Harvard analysis, seemingly innocuous disease such as seasonal influenza kills 390,000 people per year on average, including 27,000 to 130,000 individuals in the US and Europe, whilst an estimated 20 to 40 countries experience significant new disease outbreaks each year, a trend that has been increasing since 2010.
Furthermore, of the 20 neglected tropical diseases prioritized for intervention by WHO, 19 are bacterial, parasitic, viral and fungal infections such as Chagas disease and leishmaniasis, affecting over one billion people worldwide, causing countless millions of deaths a year and contributing to enormous social and economic burden from social stigma, physical disabilities, disfigurement, blindness, discrimination, loss of social status, malnutrition, growth failure and impaired cognitive development.
As Benjamin Perry from the Drugs for Neglected Diseases initiative states: “The human and economic cost of infectious diseases, especially neglected ones, is underrecognized and of a truly astonishing magnitude.”
Given the staggering breadth and toll of these figures, and the likelihood that they will, without substantial intervention, continue to increase, it is important that future, as well as ongoing pandemics, are prioritized in the same way as COVID-19, both in terms of financial funding and in the public consciousness.
To draw out the lessons we should learn from the COVID-19 pandemic, we interviewed a range of infectious disease and public health specialists from around the world, including leading scientists and clinicians, pharmaceutical and diagnostic company industry experts as well as funders and NGOs active in the space. We also spoke to advisors counseling governments’ responses as well as clinicians and managers on the front-line of the fight against COVID-19, influenza, TB, HIV, hepatitis C and neglected diseases. Finally, we covered all aspects of the value chain from early-stage research, clinical development, manufacturing, supply chain and logistics. We have summarized key lessons from the COVID-19 experience for other infectious diseases into three themes: pandemic preparedness; policy; and patients (see Table 1).

Pandemic Preparedness
Lesson 1: Monitor For Emerging Threats And Prepare A Rapid Coordinated Response
Robust surveillance systems are a critical component of pandemic preparedness because they help to identify and define the landscape of potential future disease outbreaks.
According to Professor Charles Bangham, co-director of the Institute of Infection, Imperial College London: “While it is inherently unpredictable which new disease will emerge and when, it is clear where diseases are coming from: areas with zoonotic transfer from birds, animals such as bats, bushmeat hunting, and ecological disturbance.”
The early detection of diseases as they emerge maximizes the chance of an early response and controlling an outbreak before it can spread out of control. It is therefore unfortunate that funding for monitoring programs tasked with identifying viruses which could cause global public health emergencies, such as USAID PREDICT, is often deprioritized once the immediate crisis has passed. Hopefully, the scale of the COVID-19 impact will serve to reinforce the importance of longer-term funding for national and international monitoring systems.
Allied to appropriate monitoring is the need for a rapid, coordinated response within and between nations. At the start of the COVID-19 pandemic, collaboration between agencies within national boundaries as well as between countries and continents was noticeably poor.
A key lesson to be learnt was also the criticality of supra-national agencies in coordinating the pandemic response. Barry Greene, the former managing director, finance and operation at GAVI, said: “I hope that countries recognize the need for transparency, collaboration and a strong WHO as a result of COVID-19.”
Lesson 2: Invest In Disease-Agnostic R&D
The world was relatively poorly prepared for a coronavirus outbreak: prior to COVID-19, R&D spend on coronaviruses totaled just $21m in 2018, while disease-agnostic “Disease X” platforms accounted for $71m, according to data from Policy Cures Research. In comparison, by October 2020 approximately $9bn had been spent on COVID-19 R&D. This represents an almost 100-fold increase over the total of the two previous categories in a period of fewer than 12 months (see Exhibit 2).
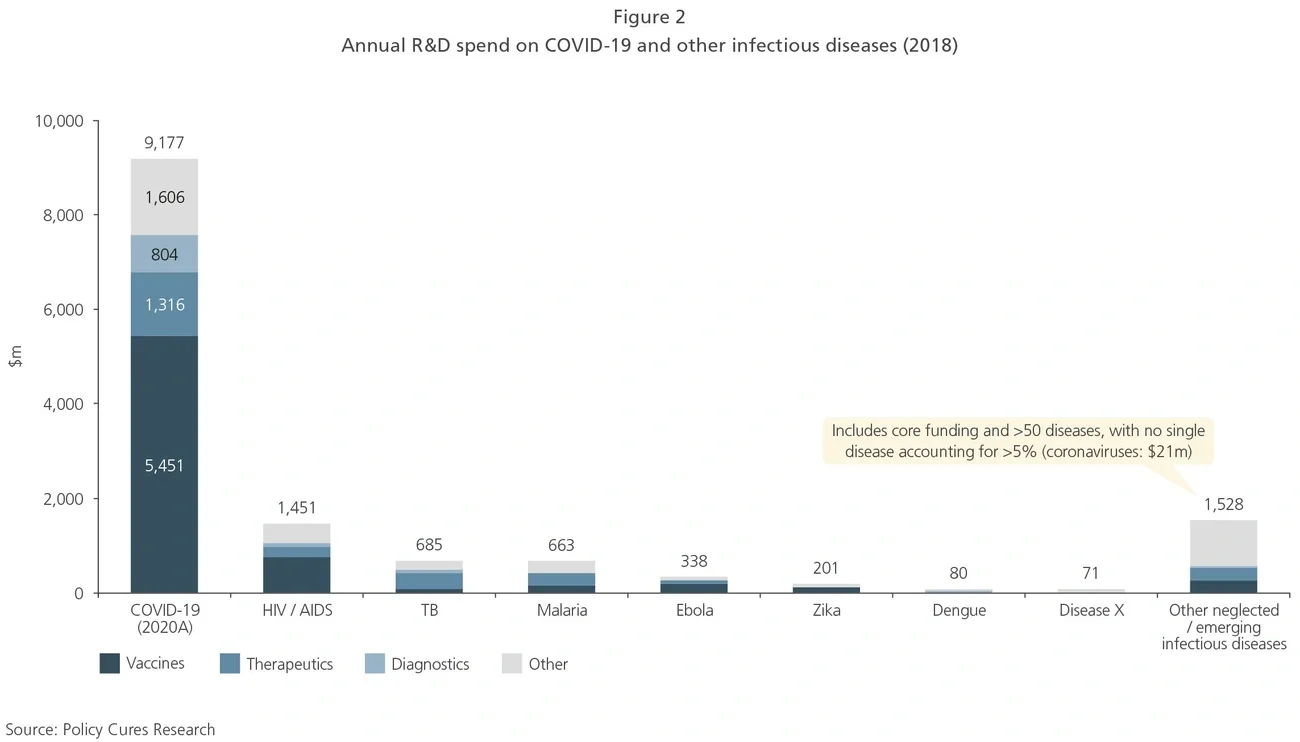
Disease-agnostic R&D to develop “Disease X” vaccine platforms will hopefully receive a welcome increase in funding given their now well-understood importance.
Beyond vaccines, there remains significant untapped potential for “tool-boxes” of molecules against potential pandemic pathogens, as highlighted by Mat Todd, professor of drug discovery at University College London. He said: “Molecules need to be developed in advance. That’s possible if you target so-called “conserved” targets that are relatively constant over time. Viruses rely on host machinery to work properly, so you can target that machinery before the virus exists.”
Lesson 3: Rapidly Redirect The Focus Of The Biopharmaceutical Industry
The biopharmaceutical industry and its collaborators have demonstrated exceptional agility and speed to focus rapidly on an emerging global crisis. Whether in the rapid repurposing of existing patented molecules (e.g. remdesivir from Gilead Sciences, Inc.), expediting clinical development timelines or in bringing to bear its enormous manufacturing strength and cold chain logistical know-how, the industry has firmly established its credentials in aligning purpose with profit when in the face of a global catastrophe.
For Gilead SVP Jacopo Andreose, there are now two important areas beyond the current pandemic on which the industry ought to continue to focus. The first is the new technologies that have already proven effective, for instance the breakthrough in mRNA vaccinology. “What other new technologies are out there for other infectious diseases that might work?” asks Andreose. The second is the massive effort already undertaken to identify drugs that could work against COVID-19. “Can we say that we have done this as impeccably yet for other infectious diseases?” he questions.
Lesson 4: Invest In Health System Resilience
Initiatives are urgently required to make health systems more resilient against future disruption caused by infectious disease outbreaks to minimize avoidable indirect mortality and morbidity.
This is as true in the more developed health care systems as it is among the developing nations. For instance, an estimated additional 33,890 cancer deaths are predicted to occur in the US in 2021 due to delayed diagnoses, whilst the Global Fund has estimated that up to 1.4 million additional people could die as a result of disruption in the prevention and treatment of HIV, TB and malaria. There is good reason to be worried: during the 2014 Ebola epidemic in Guinea, more additional deaths occurred from disrupted malaria treatments than from Ebola itself.
Potential solutions include increased reliance on telehealth, investment in more robust supply chains, and longer prescriptions, so that patients with daily treatments are less susceptible to health care disruption.
Many of these changes bring benefits outside of a pandemic context as well, as noted by one director of global public health at a large pharmaceutical company who said: “COVID-19 has forced decades’ worth of telehealth adoption in a span of months, and it’s going to have lasting benefits: reduced person-person risk, lower provider costs, patient travel costs, and even lower carbon costs.”
A recent report in Science Magazine, estimated that preventative measures against future pandemics would cost the world approximately $26bn per year. When compared to estimates of the global economic cost of this pandemic of anywhere between $12trn and $28trn, this is likely to be viewed as a reasonable investment.
Policy
Lesson 5: Increase R&D Funding And Expedite Development Timelines
A COVID-19 vaccine was authorized for use by the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK fewer than 300 days after the commencement of clinical trials, demolishing the previous record of four years (Merck’s mumps vaccine; see Exhibit 3). This is not just a reflection of investment in vaccine development, but also of coordination and reform amongst regulatory agencies.
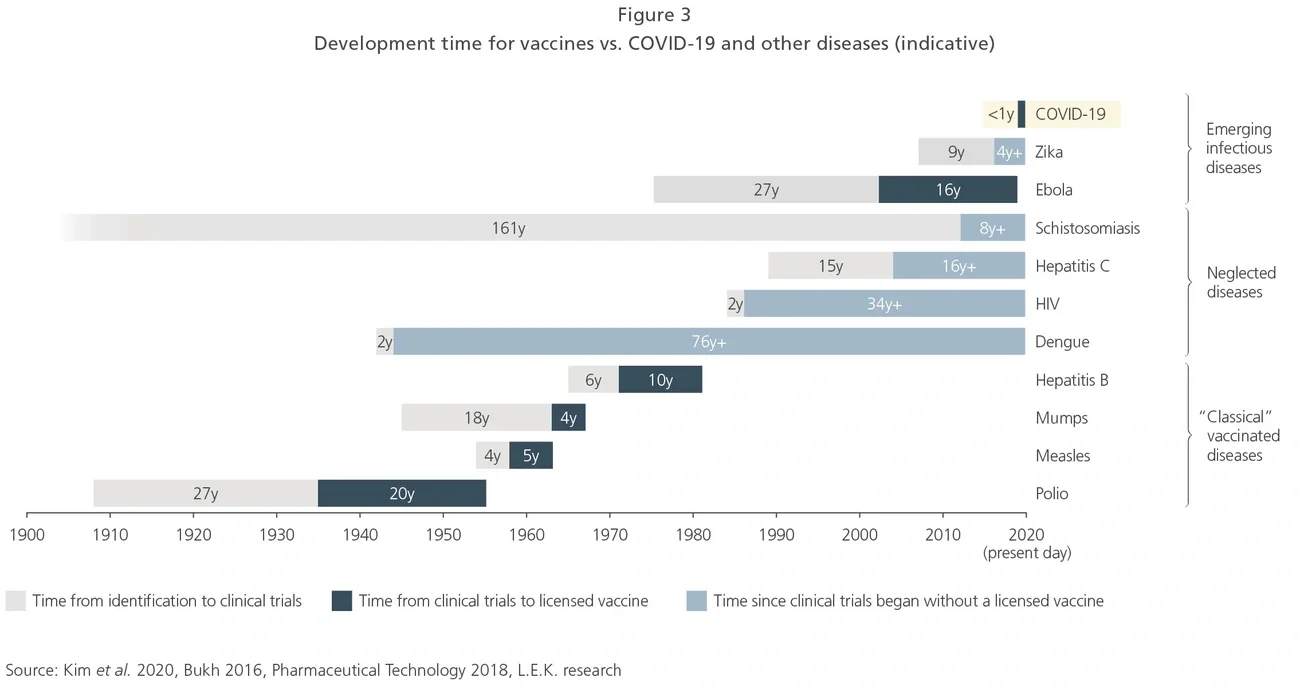
The US Food and Drug Administration for instance has streamlined its communications and turnaround times, as well as making greater use of umbrella trials and emergency use authorizations to dramatically accelerate development timelines for all virus countermeasures.
The FDA already plans to make aspects of its acceleration efforts around the pandemic permanent, and the potential for multiple disease areas is exciting. This was noted by one senior executive at a large pharmaceutical company, who commented: “The most impressive thing to me has been the regulatory streamlining and use of EUAs to really accelerate development – why can’t we do this in areas with high existing unmet need outside of a pandemic?”
More broadly, the level of R&D investment in COVID-19 over the past year has dwarfed that seen in other infectious diseases, even those with extremely high burdens (see Exhibit 3). Witnessing the success of development programs with such significant financial backing raises the hope that neglected infectious diseases could also see greater and faster progress through larger investment.
Furthermore, Joe Simmonds-Issler, chief of staff of the Coalition for Epidemic Preparedness Innovations (CEPI), believes that technologies that have been validated in response to this pandemic, such as mRNA or adenovirus vaccines, may have direct applications for other diseases. He said: “The University of Oxford are working on other diseases for their adenovirus vaccine platform, and their processes will be more validated and streamlined through this experience.”
Lesson 6: Increase Data Transparency And Collaboration
This pandemic has also demonstrated the need for, and benefits from, data transparency and collaboration. Michele Robbins, a pharmaceutical executive with more than 20 years’ experience working in HIV, said: “We witnessed the importance of sharing scientific and clinical results quickly amongst different organizations and institutions. Sharing the data certainly contributed to finding solutions more quickly and more cost- effectively.”
Countries such as the US, the UK, France and Germany have sometimes suffered from having data that is fragmented and hard to access, while researchers stress the need for centralized and simplified sources to track, for example, epidemiology and patient outcomes.
Collaborative efforts to address this problem have, though, been evident, including those arising from pharmaceutical company alliances (e.g. between GlaxoSmithKline plc and Sanofi) and pharma-academia collaborations (e.g. between AstraZeneca PLC and Oxford University), as observed by Jacopo Andreose, SVP, Gilead, who said: “The infectious disease sector has always been relatively collaborative but this level is new advocates for collaboration within our industry are being really vocal.”
These partnerships have allowed companies to focus on their strengths, pool resources and drive innovation, with great potential for progress in other diseases.
COVID-19 has also brought about increased coordination and collaboration, as well as investment in product commercialization, specifically in manufacturing and distribution. Multiple organizations, most notably CEPI and the US government’s Operation Warp Speed, have spent billions of dollars funding the manufacture of vaccines and therapeutics “at risk,” to minimize waiting times, if they are found to be effective and safe.
The world is set to reap the benefits of this with billions of vaccine doses likely to be available throughout 2021. Paul Barnsley, of the think tank Policy Cures Research, states that the logic of manufacturing at risk to reduce timelines could be extended to other high-burden diseases: “We are manufacturing at risk to get the vaccine out there as quickly as possible and save as many lives as possible. This argument for saving lives by saving time applies equally to diseases like HIV and TB as well.”
Lesson 7: Improve Medical Access For Both Physical And Mental Health
A society’s ability to defeat infection is predicated on comprehensive access for its citizens to vaccines, testing and treatment. This is especially true with diseases where patients are infectious before symptoms arise, or where infection is a source of stigma. This is true for COVID-19 but also diseases such as HIV and hepatitis C. Beatriz García Fidalgo, of Abbott Laboratories Ltd comments: “In the absence of a vaccine, defeating these diseases is contingent upon identifying high-risk groups and then proactively screening them and offering treatment; by the time they show up at the doctor’s office it’s often too late.”
COVID-19 has also highlighted the mental health impacts of disease: mental health impairment from the pandemic is estimated to have cost the US$1.5trn so far, according to a recent report in JAMA, either from direct symptoms (“Long COVID”) or related to stay-at- home measures. Whilst the burden of COVID-19 is deeply regrettable, there are significant mental health implications of other infectious diseases too, and the focus on the psycho-social elements of these diseases particularly ones with chronic or extended duration such as HIV, hepatitis and TB – is likely to be welcome.
Patients
Lesson 8: Boost Public Health Measures
Non-pharmaceutical public health measures themselves can often be the most powerful tools against an infectious disease if they are embraced by individuals and groups. The need for, and importance of, public health awareness has never been clearer: a study published by the New York Times in November 2020 showed a striking association between the stringency of US states’ containment measures and their relative numbers of COVID-19-related hospitalizations.
Blythe Adamson, a leading infectious diseases epidemiologist and affiliate professor at the University of Washington, agrees. She said: “The social determinants of health are often overlooked but I think that the COVID experience has engendered greater empathy for their importance.”
Non-pharmaceutical interventions, such as “social distancing,” undertaken for COVID, also seem to be effective against a broader range of respiratory diseases, including TB and influenza. One senior executive at a large pharmaceutical company said: “If everyone were to follow the same measures for TB as for COVID, we would see astronomical decreases in patient numbers.” In the case of influenza, the southern hemisphere’s winter spike did not materialize in 2020, and it appears to be significantly curtailed in the beginnings of the northern hemisphere’s winter as well.
Lesson 9: Focus On Messaging And Maintenance Of Public Trust
The effectiveness of public health measures, as well as that of vaccines and therapeutics, is contingent on the public’s acceptance of them. The need for clear, consistent, and coordinated messaging has been demonstrated in the struggles of multiple countries, as well as the success of nations such as China, South Korea and New Zealand.
Changing behavior can be incredibly powerful but is also very difficult and requires a lot of focus from governments,” said an infectious diseases pharmacist in a UK hospital. This principle applies to all infectious diseases, from controlling STIs to maximizing vaccine uptake for influenza and previously common childhood infections. The same point can also be made for the importance of combating misinformation, which was already an issue for vaccines and other medicines but has been amplified during this pandemic.
Lesson 10: Invest In Digital Health Technology
Digital health technology has proved an important tool during the COVID-19 pandemic. Rapidly developed digital solutions in various fields including screening, clinical management, planning and tracking, medical supplies, contact tracing and quarantine/self-isolation have all played a role in containing or helping to manage the infection rate. There is emerging evidence for countries such as South Korea that indicate that flatter infection incidence curves are attributable, at least in part, to better adoption of digital technologies.
In particular, the innovation in digital solutions that aid patients’ fight against disease (with due consideration for data protection) has been notable. Contact tracing apps and the first rapid home tests have been successfully rolled out; these advancements may have lasting benefit in the fight against other diseases. One leading infectious diseases physician said “If we think about some infectious diseases like RSV, there are now rapid tests that can give you results within minutes which are as accurate as samples sent to the lab. For infectious diseases where contact tracing is required, digital health technology could be adopted to improve existing systems.”
Lasting Change
The health care, social and economic effects of COVID-19 will continue to be felt for years, if not decades to come. We have already witnessed a transition away from pre-COVID approaches and behaviors, such as large offices or a reliance on face-to-face health care. Similarly, pre-existing trends around remote working and telehealth have been accelerated. There is also the potential for new practices to emerge from this pandemic, such as R&D and regulatory reforms and new, improved public health strategies.
Countries are about to embark on one of the largest and most logistically challenging vaccine roll-outs ever witnessed and, as they do so, one of the biggest lessons may yet be to come: how to overcome the increasing problem of vaccine “hesitancy.” A study published in The Lancet in October 2020 revealed the scale of this challenge in countries such as Russia and France (see Exhibit 4). However, optimistically, in many countries the degree of confidence in a COVID- 19 vaccine was often higher than that generally seen for vaccines overall. We can hope that a successful COVID-19 vaccine roll-out could serve as a reminder of the power and importance of vaccines more generally.
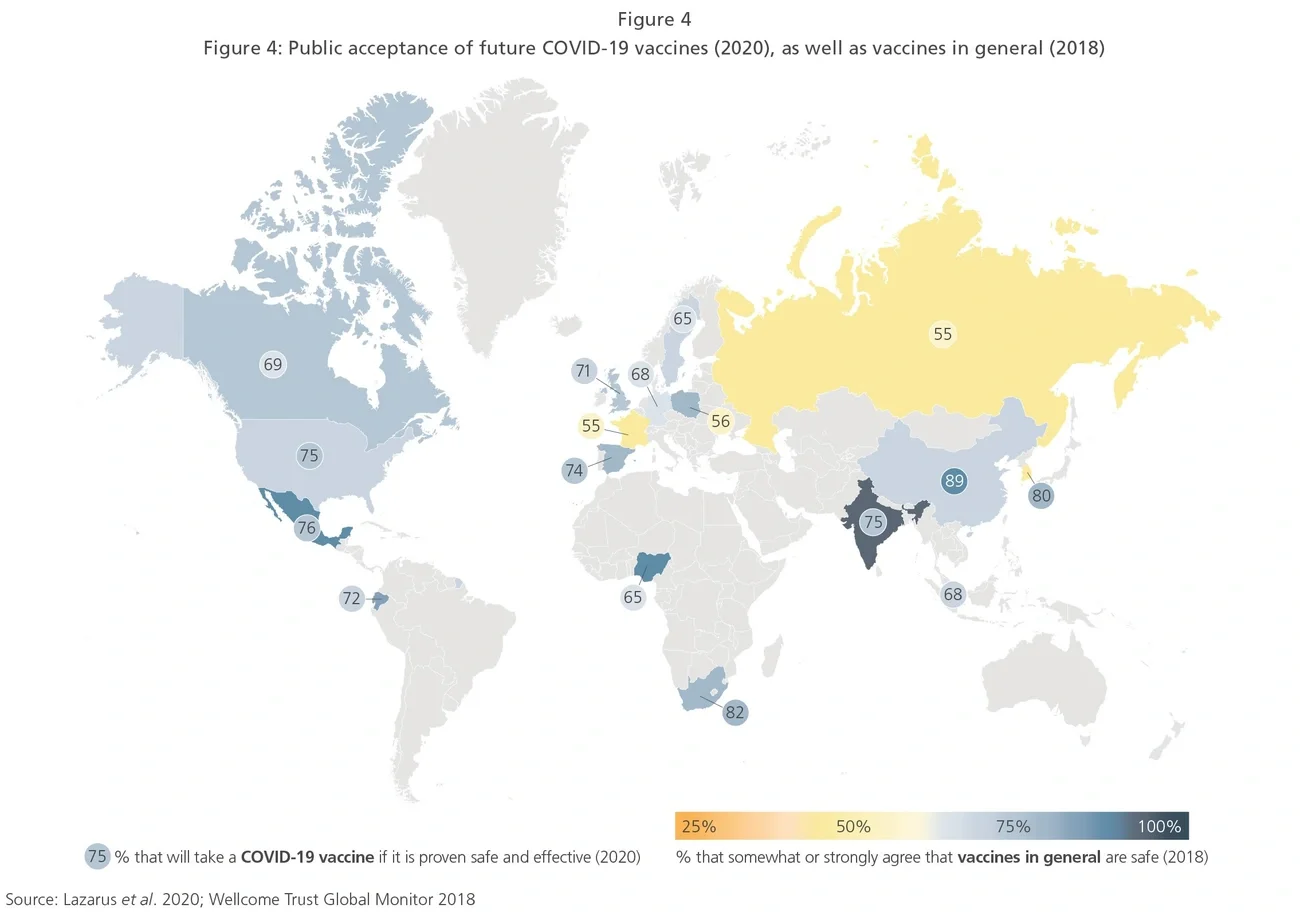
CEPI’s Joe Simmonds-Issler said: “We will emerge from this pandemic with new vaccines and platforms, companies and organizations experienced with developing, manufacturing and distributing vaccines, therapeutics and diagnostics at an unprecedented scale, and a society that has lived through the experience of the disruptive power of disease. We will absolutely have more tools against disease, but we will only go as far as our political will and financial investments can take us.”
Just as important perhaps as any individual new technology or policy is the potential for a greater understanding, both among decision makers and the public, of the disruptive and destructive potential of disease at the societal and personal level. This burden can be felt just as keenly for more familiar infectious diseases such as influenza, HIV or TB, as well as non- communicable diseases such as diabetes and cancer. There are an abundance of lessons and new tools for us to learn from and use as a result of this pandemic; it is then a question of how well we choose to use them.
Editor’s note: This article first appeared in In Vivo.










































