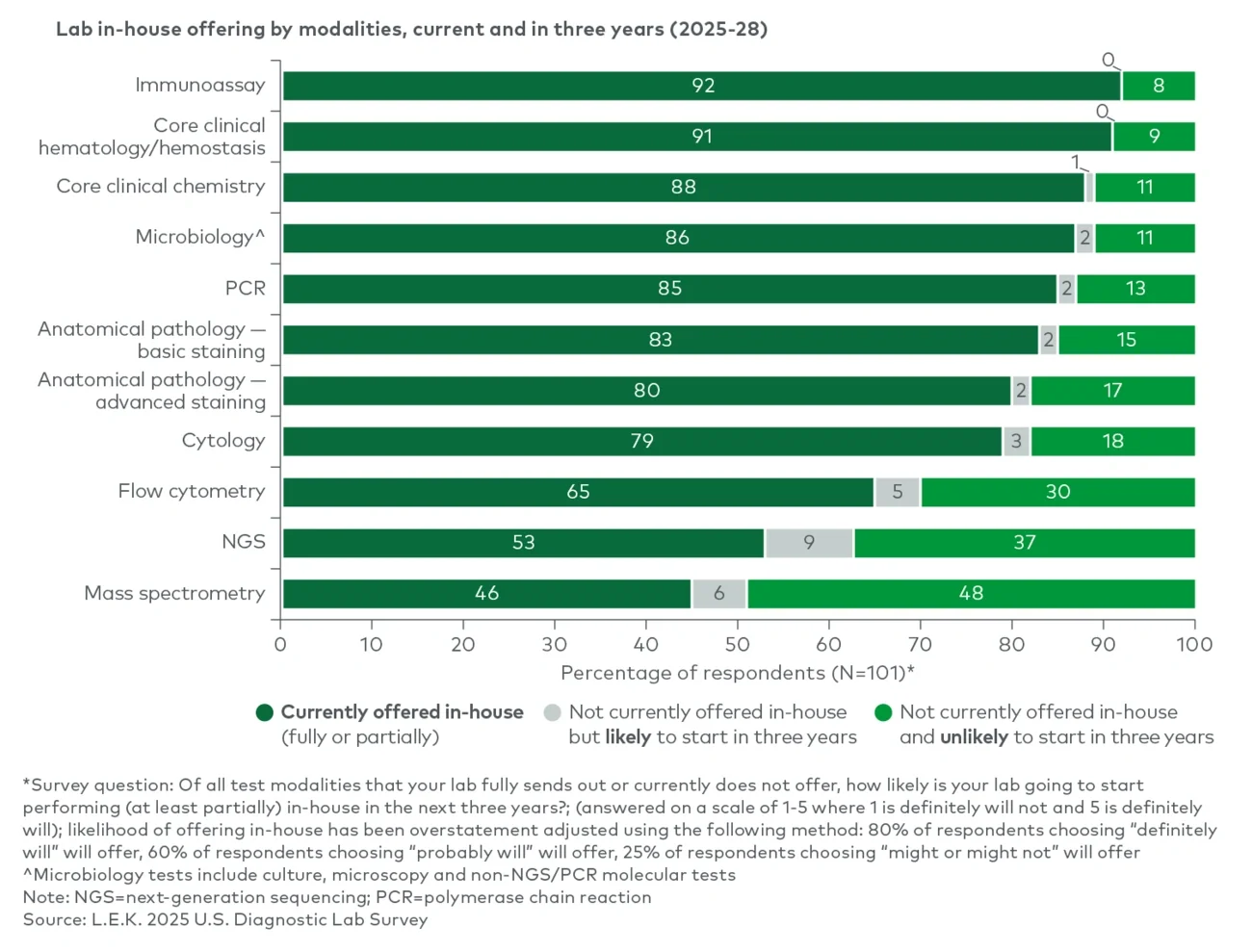
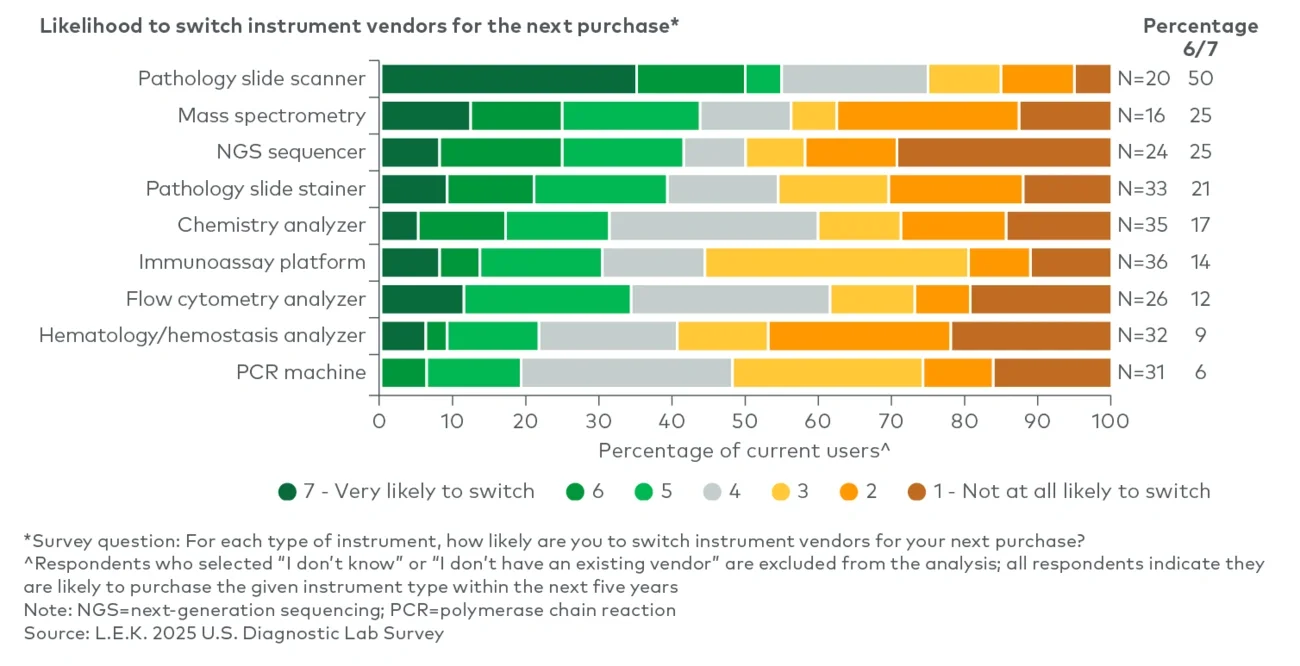
Which diagnostic modalities are poised to see the highest volume growth? Which modalities are labs most likely to bring in-house versus continue to outsource? And where is vendor loyalty most likely to shift as the next refresh cycle approaches?
To answer these questions, L.E.K. Consulting surveyed 100-plus executives and directors across hospital and reference labs in our 2025 U.S. Diagnostic Lab Survey. This edition of Executive Insights summarizes expected volume growth, insourcing/outsourcing shifts, and purchasing and switching intent across diagnostic modalities.
Key trends
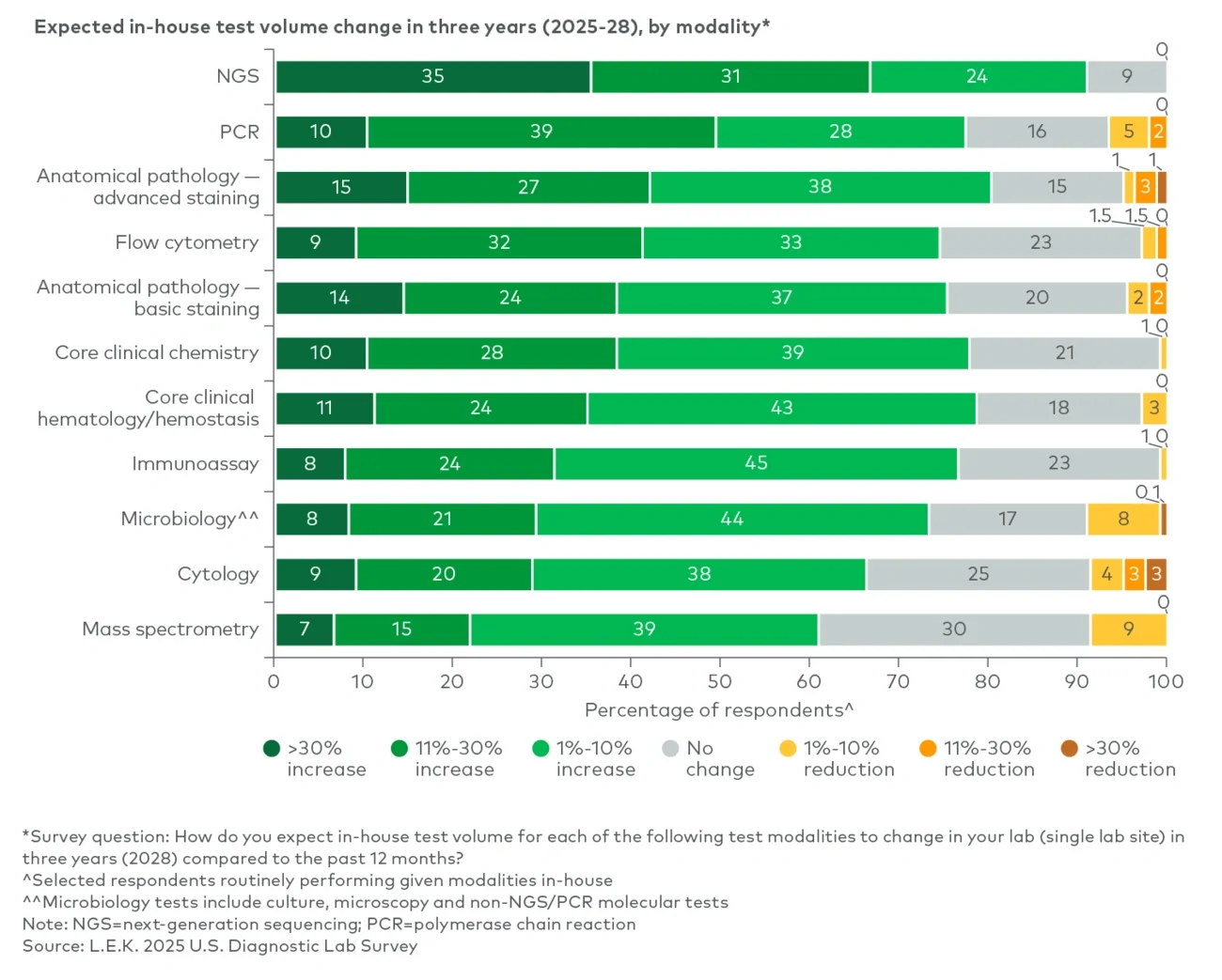
Test volume growth is expected across modalities, led by molecular and pathology advanced staining
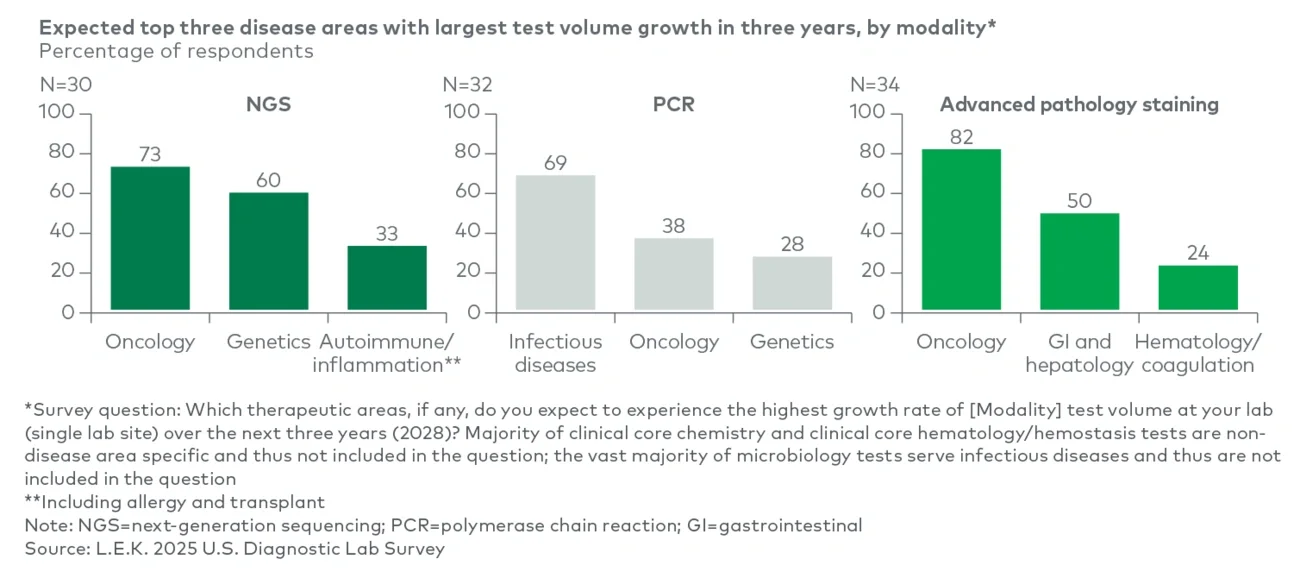
More than 90% of respondents expect in-house test volumes to increase over the next three years, with the strongest momentum in molecular — next-generation sequencing (NGS), polymerase chain reaction (PCR) — and anatomic pathology advanced staining (see Figure 1). Among labs currently running NGS in-house, approximately 65% anticipate double-digit volume growth over the next three years, including 35% projecting growth above 30%, driven by expanding clinical utility in oncology and continued declines in sequencing costs. PCR also shows a strong growth outlook, with around 50% expecting more than 10% growth by 2028, reflecting continued demand and menu expansion in infectious disease (e.g., multiplex gastrointestinal and genitourinary panels).