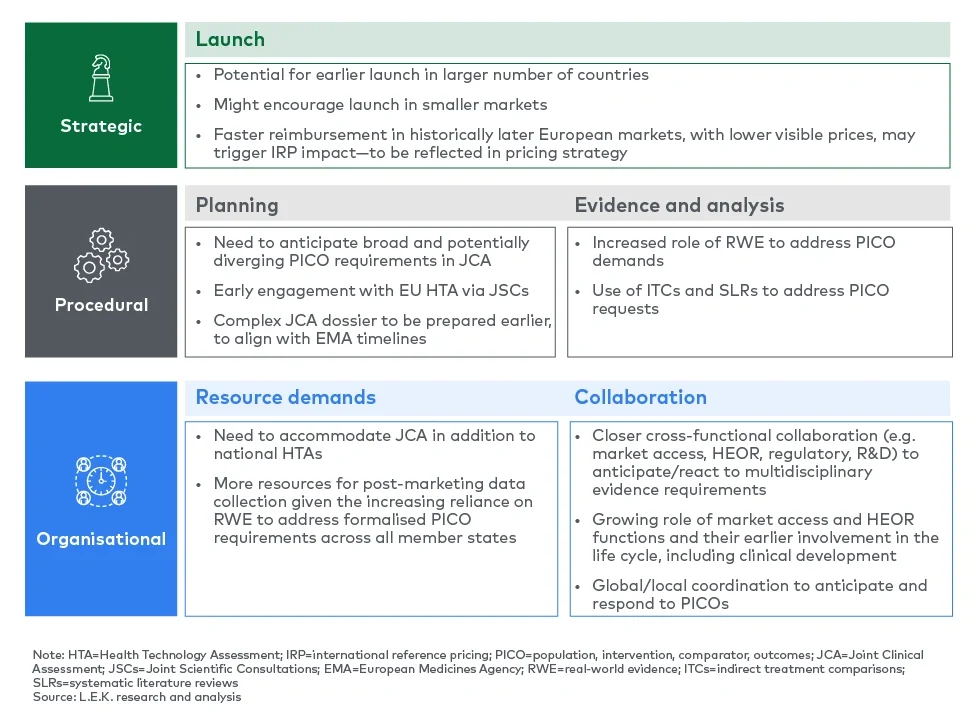
Pharma companies need to keep these accelerated timelines in mind in their launch decision-making as this, together with the coverage of all member states in a joint assessment, will facilitate earlier reimbursed access in a larger number of countries, including smaller markets which have traditionally been deprioritised for launch preparations. Launch readiness and tracking will be more important than ever, given the shortened time frame from regulatory submission to market access.
In this context, pharma companies will have to review their EU launch cadence, with the potential for earlier launch waves in a greater number of markets. However, the opportunity for earlier access needs to be balanced with associated launch costs and pricing implications, as faster reimbursement in historically later European markets, with often lower visible prices, may accelerate price erosion driven by international reference pricing.
“Most companies have traditional launch waves, going by size of market, but I think [the JCA] will expedite the waves and make midsized markets more attractive. … It will help to prioritise and sharpen the launch sequencing efforts within Europe. I expect that midsized markets will get pulled higher up the launch sequence because a certain amount of the market access work will already be done [via the JCA].”
— Global head of market access, midsized pharma company
While the JCA might bring smaller markets into closer reach, interviewed experts fear that it will add complexity, which will be challenging, particularly for smaller companies, and might prompt these to reconsider their European launch approaches altogether. This might include deferring European launches or seeking an experienced licensing partner familiar with regional market access requirements.
“The biggest challenge is to ensure that the process does not hold back smaller pharma/biotech companies from coming to Europe. It could turn out to be so cumbersome and costly that they may just decide not to come.”
— Head of European market access, midsized pharma company
Implications for pharma: Procedural changes will impact evidence generation and analysis
Submission process and timelines
The JCA process will trigger procedural changes in multiple dimensions, including in the way evidence requirements are specified and submitted, as well as in the HTA timeline.
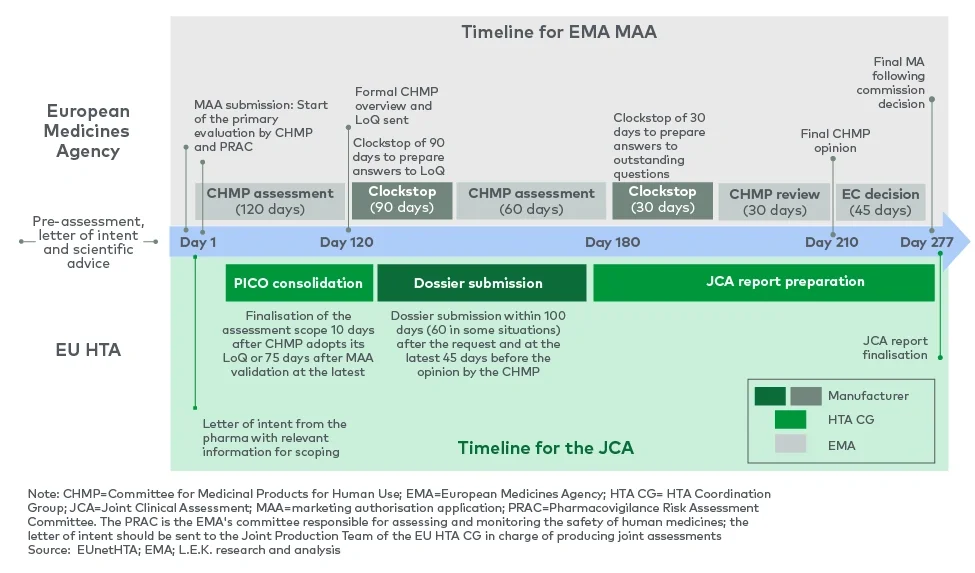
The JCA starts with the scoping process. To this purpose, a population, intervention, comparator, outcomes (PICO)2 survey is set up to provide each member state with the opportunity to communicate PICO parameters. Disparities between countries in terms of expected endpoints and comparators may initially result in a high number of PICO parameters, but answers will be consolidated and condensed as much as possible.
The consolidated PICO requirements will be transmitted to pharma companies to allow them to gather the necessary data. Assessors and co-assessors will then evaluate the evidence provided and draft the JCA for publication by the European Commission.
A challenge of the PICO scoping process is that it occurs after the pharma company has applied for EMA approval, meaning that requests from the survey cannot be reflected in the clinical trial design. Further, the need to amalgamate PICO requests from 27 member states will bring complexity, particularly in therapeutic areas with significant differences in clinical practice across markets, and could result in requests for analyses which were not specified as part of the trial design.
Pharma will need to anticipate PICO needs and seek guidance on PICO requirements as early as possible. This highlights the importance of joint scientific consultations (JSCs), which can occur ahead of clinical trials to make sure that inputs from national HTA bodies are considered during the design process.
Companies must be prepared to prove eligibility for the JSC in the selection phase. This includes demonstrating unmet medical need, highlighting the expected positive impact on patients and healthcare systems, aligning with major EU clinical priorities, and applying for the JSC while phase II and III studies are still in the planning stage.
However, concerns remain about meeting the growing demand for scientific consultations given the limited availability of slots. To address this, simulating PICO requirements through other routes — such as early engagement with national HTA agencies, medical organisations and patient associations — will be critical.
“The JSC in itself is not enough. The JCA isn’t binding, and it is on the local HTA body to interpret the data in the JCA as it sees fit and also to request any extra data to meet the national requirements. So I think that even if you get a JSC slot you will still have to take local early advice as well. For example, the German G-BA recently highlighted that a separate consultation at [the] national level is recommended, in addition to the JSC.”
— Head of European market access, midsized pharma company
Pharma companies will need to plan their submission strategies and processes carefully to align with both EMA and HTA timelines, taking into account that the new JCA timeline creates a short window of time from receipt of the scoping document to develop a comprehensive dossier.
“The JCA is running concurrently with the regulatory process and hence comes way earlier than the national HTA submissions. This means that market access teams need to start their planning much earlier.”
— Head of European market access, midsized pharma company
Evidence collection and analysis
The multitude of scoping requests from member states may create evidence-generation challenges, particularly considering the timing of the scoping process after the initiation of clinical trials. This might strengthen the role of real-world evidence in supplementing the JCA evidence base and meeting PICO demands (e.g. as an external comparator group for single-arm trials).
Further, it is expected that indirect treatment comparisons (ITCs) will play a major role in addressing the evidence needs specified by member states, supported by the methodological guidance on ITCs provided by the Member State Coordination Group on HTA (HTA CG).3 Planning the necessary systematic literature reviews to feed into the ITCs will be key to addressing diverse PICO requirements across member states.
Implications for pharma: Organisational demands on resourcing and collaboration
Increased resource demands
Pharma companies must allocate resources to meet the EU HTA regulation requirements in the context of tight timelines. This will increase operational demands, particularly for those preparing for assessments in 2025. Companies need to consider how to secure and deploy resources in a cost-efficient manner. If external expertise and/or additional internal resources are used, the budget for this needs to be secured early to avoid delays in accessing support ahead of critical inflexion points.
“Having the JCA run in parallel with the EMA submission is the biggest change. For local dossiers, the pharma company decides when to submit and where. The JCA is mandatory; you need to prepare it in parallel with the MAA and there are very clear timelines for the submission, so there is less control. This has the potential to increase resource needs at least during peak times.”
— Head of European market access, small pharma company
For smaller companies building up European infrastructure, this means having affiliate market access resources in place earlier to accommodate the JCA timeline.
“Affiliates need to engage with access stakeholders earlier. They have work to do at a local level much earlier to collect information and feed [it] into the JCA dossier; this is one of the fundamental changes I see. Smaller companies launching in Europe for the first time need to be aware that they can’t postpone the local head counts — they need them earlier, no longer just six months before approval.”
— Head of market access, small pharma company
“The timeline is so crunched for the JCA it means we will need extra staff. Although the JCA and local dossiers should be non-duplicative, I think there is definitely more work, just due to the fact that we need to submit two dossiers — the JCA and then the local one. This means you need local experts who know the country-level HTAs well and then you need a set of experts at the European level. This is almost like adding another large country to your market access organisation.”
— Head of European market access, midsized pharma company
Cross-functional collaboration
The complexity and timing of JCA preparation will require closer coordination between functions, most notably market access, health economics and outcomes research (HEOR), clinical R&D and regulatory. Breaking down silos will be essential for early scientific consultations and for planning the evidence generation, including the anticipation of PICO demands, and in order to develop the dossier.
Companies will need to adapt governance structures to integrate EU HTA regulation capabilities and ensure its strategic importance is clearly communicated across the organisation.
“We need close collaboration across regulatory, access [and] HEOR at a minimum during the entire process. Any changes in regulatory need to immediately feed into access because the processes are happening in tandem.”
— Head of market access, small pharma company
Further, the EU HTA regulation is expected to shift the market access and HEOR functions into focus and emphasise its centrality for successful launch and commercialisation. Closer collaboration between access and other functions, and in particular the increasing need to consider HTA requirements in clinical development planning, will strengthen the role of market access and reinforce the shift towards an integrated value-and-access function for more effective engagement with HTA bodies.
“In general, the idea that clinical development will be tailored more to the requirements of HTA bodies is an old one, but I think [the JCA] will put more emphasis on the importance of HTA demands. You need cross-functional teams — access, regulatory and clinical at a minimum — for discussions on trial design early on as part of the scientific consultation.”
— Head of market access, small pharma company
“I think it will motivate clinical development to involve market access earlier and follow their recommendations more closely to ensure that we don’t have trial designs that don’t meet payer needs.”
— Head of European market access, small pharma company
Conclusions and implications
The EU HTA regulation offers valuable opportunities for harmonising clinical assessments across EU member states, facilitating and accelerating market access in a larger number of countries, and potentially improving HTA outcomes by forcing market access to be a central part of the clinical planning process (via JSCs).
This will come with significant procedural and organisational implications for pharma companies, adding timeline, resource and commercial pressures. Most notably, the alignment of the regulatory and JCA timelines, the need for early scientific consultations and for simulating PICO requirements across member states, and the requirement to accommodate both the JCA and local HTA submissions will add workload and complexity for market access teams and other functions. This requires capacity building, particularly among smaller companies, as well as closer cross-functional collaboration and coordination between local and global teams.
Procedural and organisational changes might present challenges, but they will also create opportunities for a more central, effective and impactful market access function. Earlier and closer integration of market access considerations into the drug development and commercialisation processes might ultimately lead to better reimbursement and pricing outcomes and, hence, improved access for patients.
“The JCA will be difficult in the beginning because companies aren’t used to it and don’t have processes in place, but I think it will be a positive in the mid and long term. Some colleagues who have worked in more siloed companies think that it will give market access more power to input into the process earlier, and other functions are more likely to listen. It should lead to better HTA outcomes, as pharma companies are more likely to deliver what is valued by payers.”
— Head of European market access, midsized pharma company
How L.E.K. Consulting can help
The EU HTA regulation will bring significant changes to EU market access processes. Pharmaceutical companies need to prepare by adapting internal submission processes and templates to meet JCA requirements and timelines, anticipating PICO needs, and engaging in consultations with regional and local stakeholders to embed HTA requirements into evidence generation.
L.E.K. can provide tailored perspectives on the implications of the EU HTA regulation for pharma companies, covering market access strategy, launch preparation and monitoring, and organisational readiness.
To discuss the topic in more detail, please reach out to the authors.
The authors would like to thank Daniel Secker and Charlotte Barthen for their support in writing this Executive Insights.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting
Endnotes
1Health technologies subject to a JCA include those undergoing a centralised marketing authorisation that are medicinal products containing a new active substance and/or biotechnology products, class IIb or III medical devices, and class D in vitro diagnostic devices.
2The PICO parameters include: population: identification of the relevant population(s) for the assessment scope, based on the claimed indication; intervention: a combination of the intervention to be assessed and the indication or intended use; comparator: pharmacotherapies and/or nondrug interventions that are relevant for the HTA for each of the populations of interest and that serve to determine the relative effectiveness of the health technology assessed; and outcomes: the choice of endpoints of interest and relative improvement necessary to demonstrate the relative effectiveness of the health technology assessed.
3HTA CG, “Methodological Guideline for Quantitative Evidence Synthesis: Direct and Indirect Comparisons” (adopted on 8 March 2024).