Success in indirect procurement demands both expertise and execution, delivering immediate value while building lasting capabilities. Unlike direct procurement, which is a core capability for manufacturers, indirect procurement spans diverse categories with unique stakeholders, cost drivers and complexities. Without dedicated resources and strategy, companies across industries often overspend and miss opportunities for value creation.
A leading global manufacturer faced this challenge firsthand. After successfully implementing a global sourcing function for direct materials, the company sought to replicate that success in indirect services and materials — an entirely different challenge.
Though the company had a strong leader in place, unlocking the full potential of indirect procurement required specialized expertise, a clear strategy and hands-on execution. To drive this transformation, the company leveraged L.E.K. Consulting’s experience in navigating complex procurement challenges to build a structured, scalable indirect procurement function — equipping the organization with the processes, tools and expertise to drive ongoing efficiency and cost savings.
Building a foundation for indirect procurement
While the manufacturer excelled in direct procurement, indirect spend was left to functional leaders, driving up costs, increasing risk and consuming valuable time for their teams.
L.E.K. took a structured approach to building the company’s indirect procurement capability from the ground up. Using a comprehensive strategic sourcing process (see Figure 1), the team ensured consistent execution across categories while building the client’s capabilities.
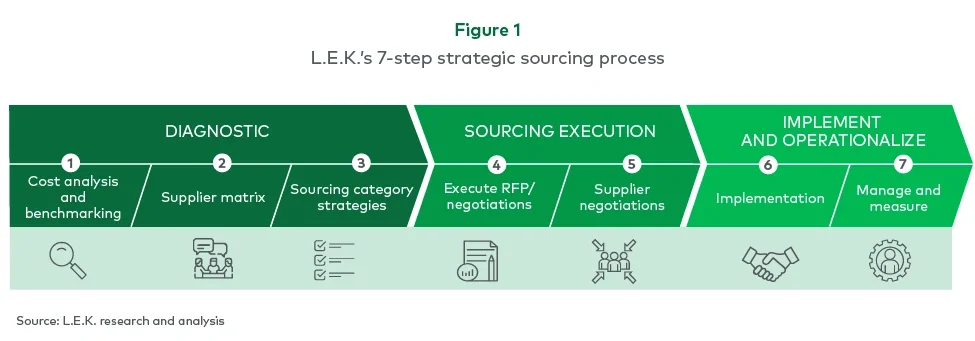
Executing for impact
With a structured approach in place, the focus shifted to execution. To drive immediate results while strengthening long-term sourcing capabilities, we embedded a senior procurement leader to work alongside the internal team. This working group tackled eight projects across logistics, packaging, facilities services and corporate services, leveraging a range of strategic sourcing techniques:
- Competitive RFPs to introduce new suppliers and optimize costs
- Incumbent negotiations informed by benchmarking and should-cost modeling
- Global sourcing to expand supplier options and drive efficiency
- Group purchasing strategies to unlock volume discounts and improve terms
- Demand management to identify services that could be discontinued or have service levels reduced to meet the need from the business
- Cost modelling to develop a perspective on cost drivers and cost savings potential in support of a key supplier relationship
These efforts delivered significant results, including 15% annual cost savings across freight, packaging, maintenance, repair and HR services. Additionally, the team developed a roadmap for 17 future projects, expected to generate 5%-8% savings on addressable indirect spending over the next two years.
In addition to cost savings, this structured approach resulted in category strategies, improved supply resiliency, supplier diversity, fit-for-purpose service levels and a path to value across key indirect categories.
Indirect procurement touches every layer of an organization, from corporate services to manufacturing, distribution and logistics — as illustrated below.
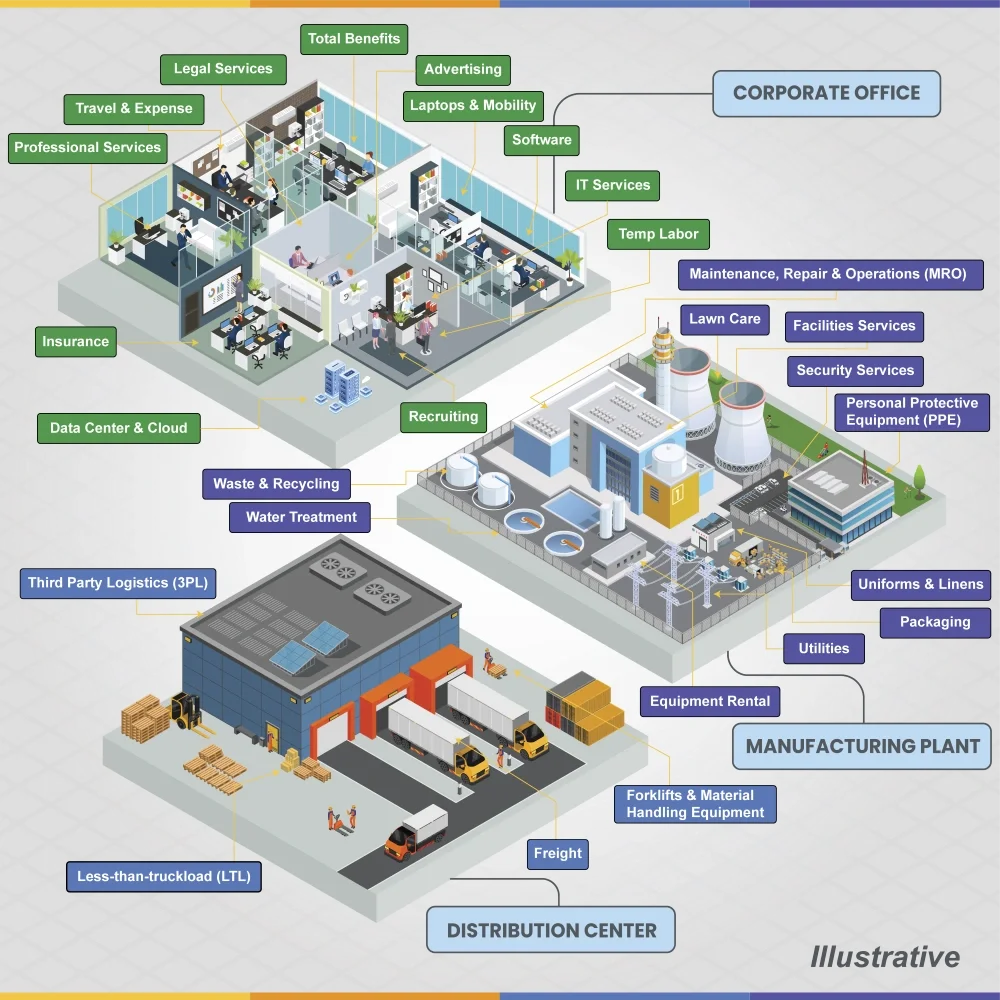
Creating an engine for value
Beyond immediate cost savings, we built a procurement function designed for long-term success. The capability roadmap developed helps establish key infrastructure, processes and expertise that will support the manufacturer’s objectives to drive ongoing efficiency and strategic sourcing excellence.
- Tools and templates: A tailored set of resources standardized RFPs, supplier evaluations and contract negotiations, making best practices repeatable.
- Master data integration: Collaboration with finance and IT to create spend visibility across disparate systems where indirect category spend was managed.
- KPI dashboard: A performance tracking system provided real-time visibility into procurement efficiency, supplier performance and cost savings.
- Contract portfolio review: Evaluation of 50+ agreements uncovered renegotiation opportunities, risk areas and cost-saving adjustments.
- Integrated procurement processes: Workflows mapped across 15 systems uncovered disparate systems, data flows and decision points that provided the foundation for the indirect team to take on management of indirect categories.
Our experts embedded experts within the team, guiding them through live sourcing projects, supplier negotiations and category strategy workshops. This hands-on approach built confidence, strengthened procurement skills and ensured long-term process ownership.
Essential insights for manufacturing leaders
This transformation highlights critical strategies for organizations looking to elevate their indirect procurement capabilities across industries:
- Reframe assumptions: Excellence in direct procurement doesn’t ensure success in indirect categories. Indirect procurement spans a broad range of services and materials, requiring specialized strategies to manage complexity, mitigate risk and drive efficiency.
- Prioritize actionable expertise: Effective transformation combines strategic guidance with hands-on leadership. Partners who work alongside teams to deliver immediate results while building long-term capabilities are essential.
- Focus on enduring value: Sustainable impact comes from embedding new capabilities, improving transparency and aligning procurement strategies with broader business goals.
Regardless of industry, indirect procurement is often considered a tactical cost reduction activity. Organizations that take a strategic approach to indirect spend can unlock hidden efficiencies and gain a lasting competitive advantage.
To learn how L.E.K. can help transform your indirect procurement function and capture significant value, contact us today.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting LLC
Creating an engine for value
Beyond immediate cost savings, we built a procurement function designed for long-term success. The capability roadmap developed helps establish key infrastructure, processes and expertise that will support the manufacturer’s objectives to drive ongoing efficiency and strategic sourcing excellence.
- Tools and templates: A tailored set of resources standardized RFPs, supplier evaluations and contract negotiations, making best practices repeatable.
- Master data integration: Collaboration with finance and IT to create spend visibility across disparate systems where indirect category spend was managed.
- KPI dashboard: A performance tracking system provided real-time visibility into procurement efficiency, supplier performance and cost savings.
- Contract portfolio review: Evaluation of 50+ agreements uncovered renegotiation opportunities, risk areas and cost-saving adjustments.
- Integrated procurement processes: Workflows mapped across 15 systems uncovered disparate systems, data flows and decision points that provided the foundation for the indirect team to take on management of indirect categories.
Our experts embedded experts within the team, guiding them through live sourcing projects, supplier negotiations and category strategy workshops. This hands-on approach built confidence, strengthened procurement skills and ensured long-term process ownership.
Essential insights for manufacturing leaders
This transformation highlights critical strategies for organizations looking to elevate their indirect procurement capabilities across industries:
- Reframe assumptions: Excellence in direct procurement doesn’t ensure success in indirect categories. Indirect procurement spans a broad range of services and materials, requiring specialized strategies to manage complexity, mitigate risk and drive efficiency.
- Prioritize actionable expertise: Effective transformation combines strategic guidance with hands-on leadership. Partners who work alongside teams to deliver immediate results while building long-term capabilities are essential.
- Focus on enduring value: Sustainable impact comes from embedding new capabilities, improving transparency and aligning procurement strategies with broader business goals.
Regardless of industry, indirect procurement is often considered a tactical cost reduction activity. Organizations that take a strategic approach to indirect spend can unlock hidden efficiencies and gain a lasting competitive advantage.
To learn how L.E.K. can help transform your indirect procurement function and capture significant value, contact us today.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting LLC
Launching Novel CDx for Oncology: 7 Strategies for Biopharma Companies
Key takeaways
Biopharma companies should integrate companion diagnostics early by adopting an opt-out approach and planning in preclinical stages.
Effective launches require navigating unique operational hurdles, building distinct Dx strategies and leveraging the right partners.
Failing to align diagnostic and therapeutic timelines can lead to lost revenue, slower adoption and reduced market impact.
To succeed, companies must invest in internal Dx expertise, align cross-functional teams and plan proactively for long-term diagnostic evolution.
Early genetic screening, targeted therapies and other precision medicine (PM) offerings in recent years have transformed care and significantly improved outcomes for oncology patients while delivering substantial value creation that drives increased pharma investment. PM leverages biomarker (BM) strategies to successfully develop, commercialize and differentiate therapeutics by improving R&D efficiency and optionality, supporting regulatory filings, and enabling smaller and more productive clinical trials.
To achieve commercial success for an oncology PM therapeutic, however, biopharma companies must also accomplish the effective launch of a companion diagnostic (CDx) that identifies eligible patients and informs ongoing treatment decisions.
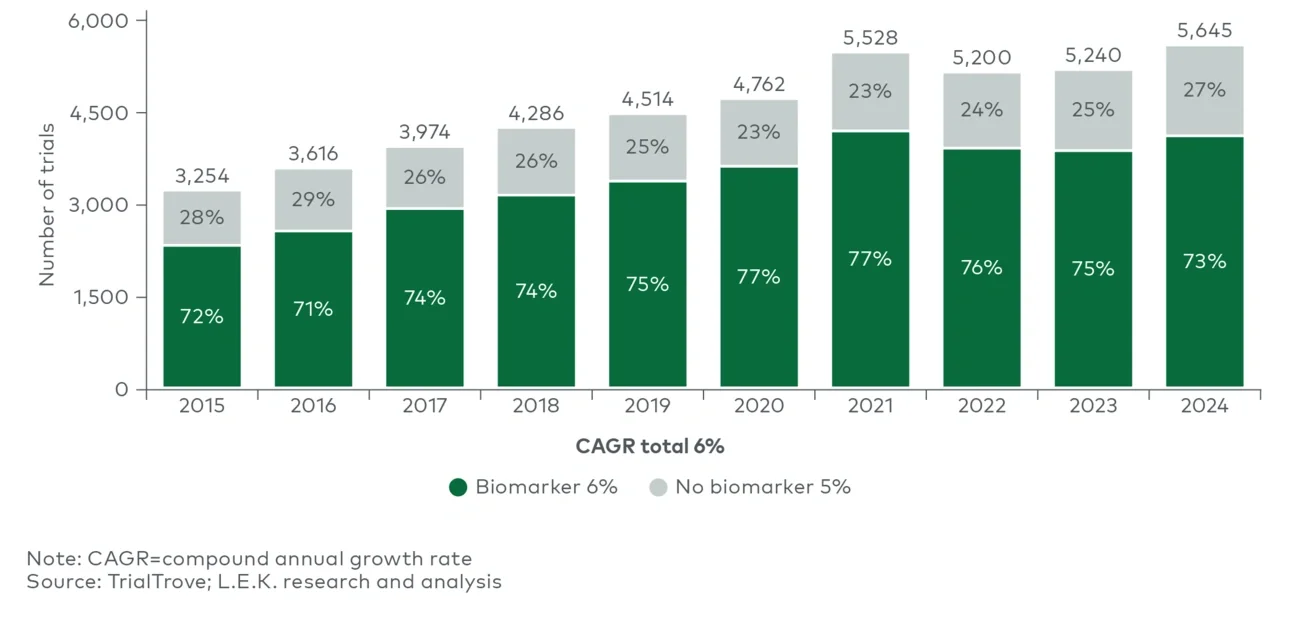
Over the past decade, the proportion of oncology trials using BMs has steadily tracked overall trial growth except for a slight post-pandemic decline amid tough U.S. and Chinese macroeconomic conditions. In 2024, three-fourths of all oncology clinical trials included the use of a BM (see Figure 1).
Figure 1
Biomarker use in oncology trials, by year (2015-24)
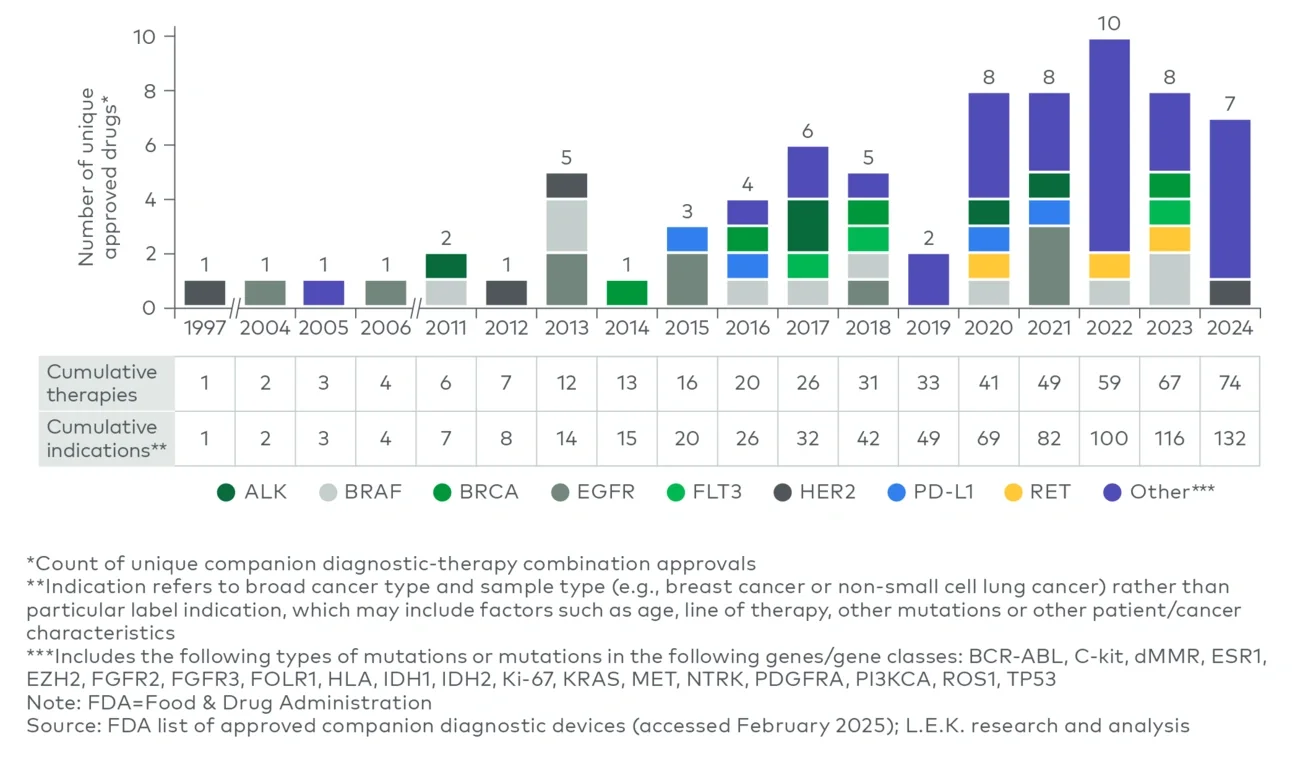
Rising BM use in trials has predictably had an impact on product launches, with the U.S. Food and Drug Administration (FDA) approving seven to 10 oncology therapeutics with CDx annually since 2020 — and with an increasing focus on novel biomarkers rather than traditional ones (see Figure 2).
Figure 2
FDA-approved therapeutics with CDx, including novel oncological therapies (1997-2024)
Given the advantages of launching a diagnostic (Dx) — and the many complexities involved — preparing to launch novel CDx in concert with the therapy itself is imperative. In working with biopharma companies to launch novel CDx for oncology therapeutics, L.E.K. Consulting has uncovered seven critical strategies to share.
1. Adopt an ‘opt out’ mentality.
Leaders in PM follow an opt-out approach: All new oncology programs start with a Dx component, consistently assessing needs and planning for them across the development life cycle. This mindset leads PM leaders to integrate Dx and therapeutic development through established Dx resources and capabilities. All-comers therapeutics can still be pursued, but this requires an active decision by leadership supported by clinical evidence.
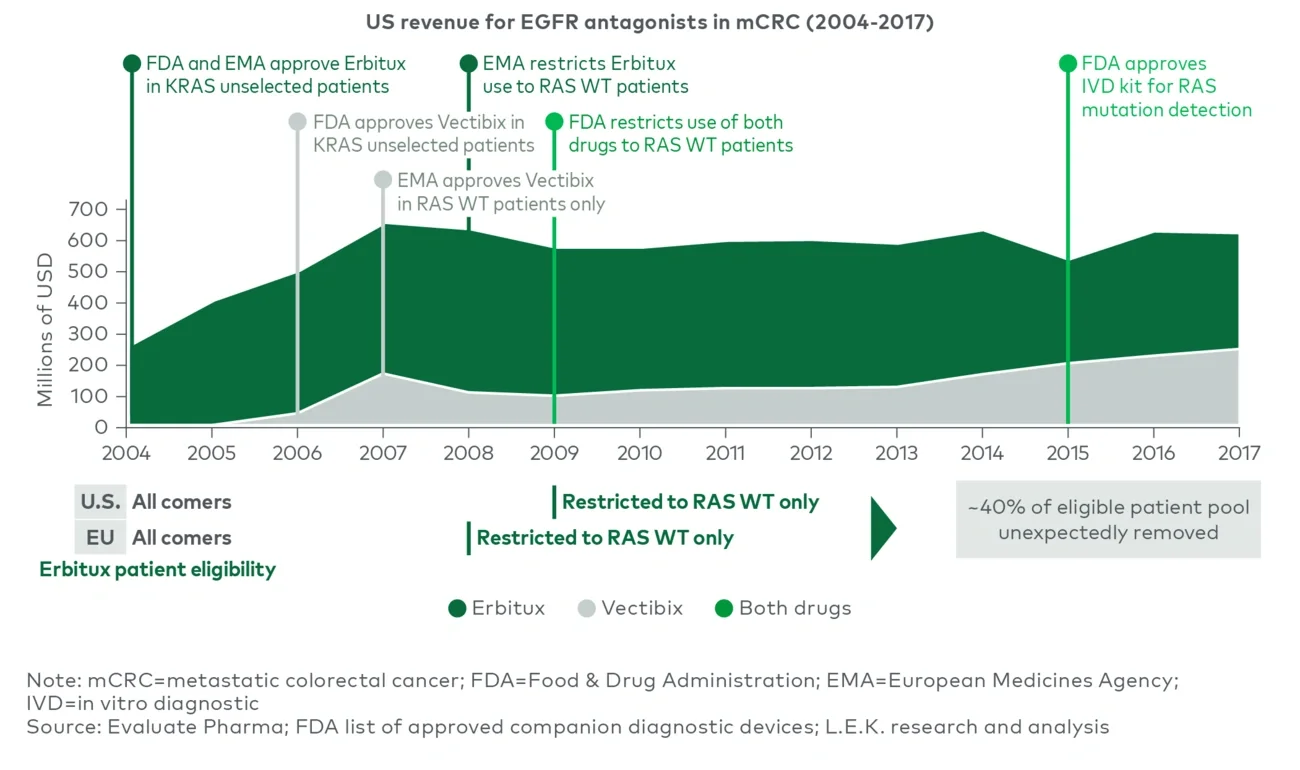
The alternative “opt in” mindset — the assumption that an all-comers approach will work and BM development will follow — limits a company’s ability to build Dx capabilities and processes, and disadvantages PM programs that require early and frequent collaboration between Dx and therapeutic teams. For example, in 2009 (after five years on the market), the FDA restricted Lilly’s EGFR inhibitor Erbitux to KRAS wild-type patients (who comprise approximately 60% of colorectal cancers) based on data from a competitor’s product. U.S. market adoption stagnated after the decision, and the cumulative revenue impact over the next decade reached hundreds of millions of dollars (see Figure 3).
Figure 3
Case study: Erbitux in mCRC
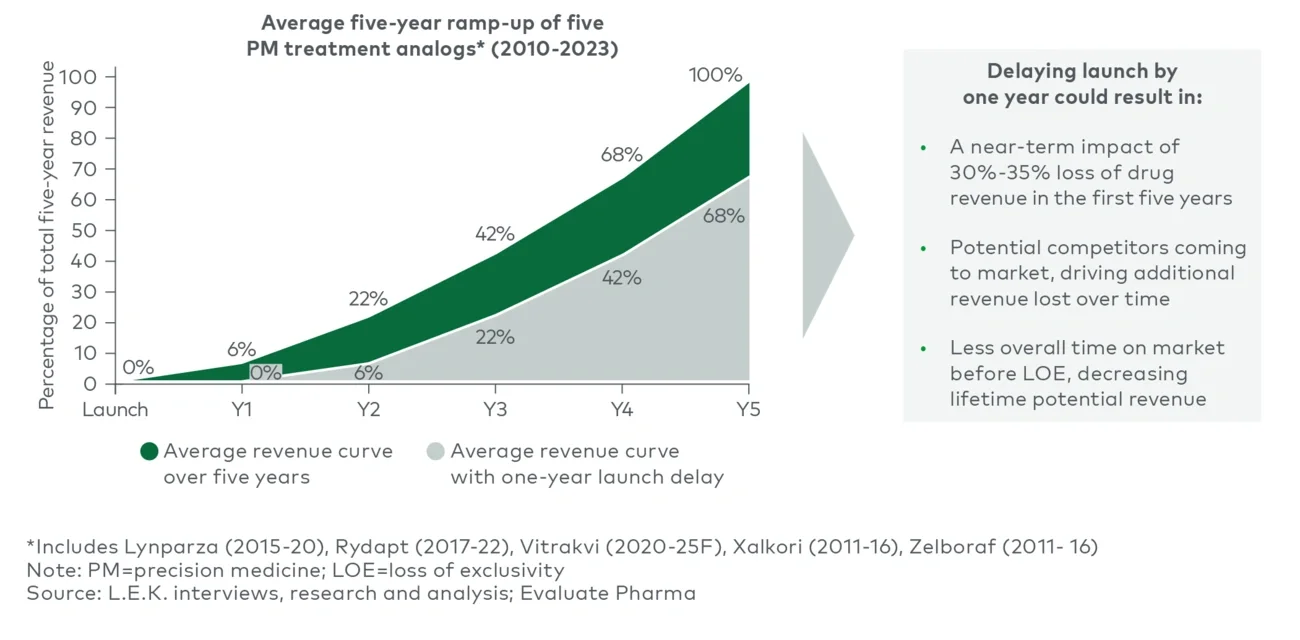
Indeed, historical averages suggest a one-year delay in launching a BM-directed drug could reduce the initial five-year cumulative revenues by 30%-35%, owing to the typical adoption ramp curve (Figure 4).
Figure 4
Revenue loss from launch delays in PM technology
Dx leaders codify the opt-out mentality in their processes, requiring teams to consider Dx needs early and to continually reassess those needs throughout development — whether by adopting a proactive approach to BM discovery through comprehensive patient profiling, banking multiple bio samples and so forth; focusing on post hoc analysis to identify predictors of response; or continually optimizing by, for example, tracking molecular origins of resistance.
Furthermore, they tend to organize personnel in ways that encourage dedicated focus on individual programs while maintaining centralized leadership and integrating functions and programs at the therapeutic area and enterprise levels. Embedding strategic Dx planning throughout the program drives preemptive discussion and collaboration and ensures organization wide sharing of lessons and resources, thus increasing efficiency and institutional knowledge.
2. Start planning for CDx launch in preclinical development.
A successful Dx launch requires multifunctional support across the value chain, and companies should start planning as early as the preclinical stage. Dx development occurs parallel to therapeutic development, with key Dx launch readiness activities stage-gated by both therapeutic and Dx milestones (see Figure 5).
Figure 5
Key Dx activities by function throughout the value chain
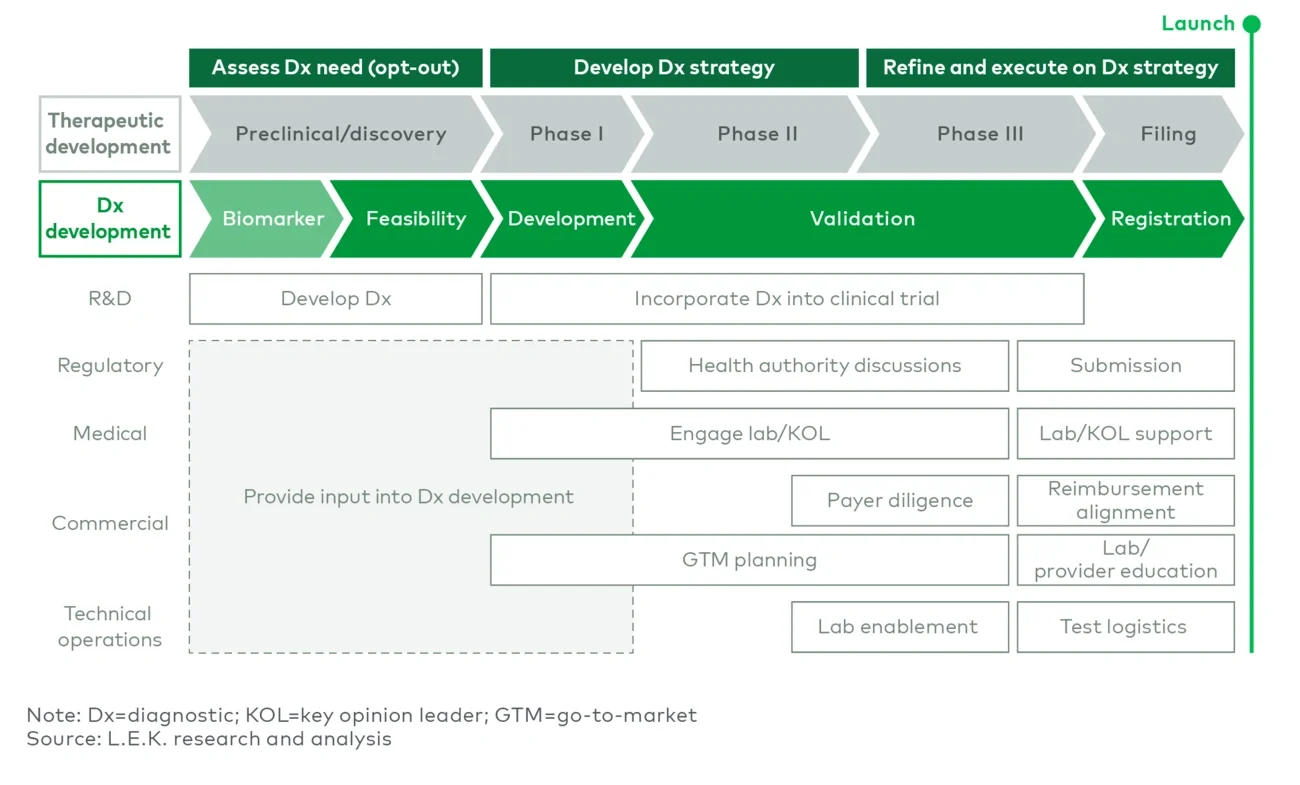
To drive efficiencies, R&D must incorporate cross-functional input from commercial and medical functions during preclinical development. This approach ensures that Dx addresses patient needs and that clinical endpoints support its commercialization. Commercial and medical readiness activities should focus on understanding and educating the market, developing a Dx-specific strategy and preparing the organization for Dx launch.
3. Address the unique operational challenges of adding CDx.
Companies must consider how the specific complexities of a Dx test should inform the commercial and go-to-market strategy. During development, an individual Dx faces specific commercial obstacles that differ from challenges with therapeutics — surrounding the analyte, such as protein or DNA; the testing technology, e.g., PCR or NGS; validated instrumentation such as 510(k) clearance; and the testing format, whether an in vitro diagnostic (IVD) or a laboratory-developed test (LDT) (see Figure 6). Pharma companies looking to develop a therapeutic with CDx should first understand the BM requirements for their indication. Next steps include determining whether they can support a decentralized testing model and building a robust payer strategy.
Figure 6
Considerations for Dx approach
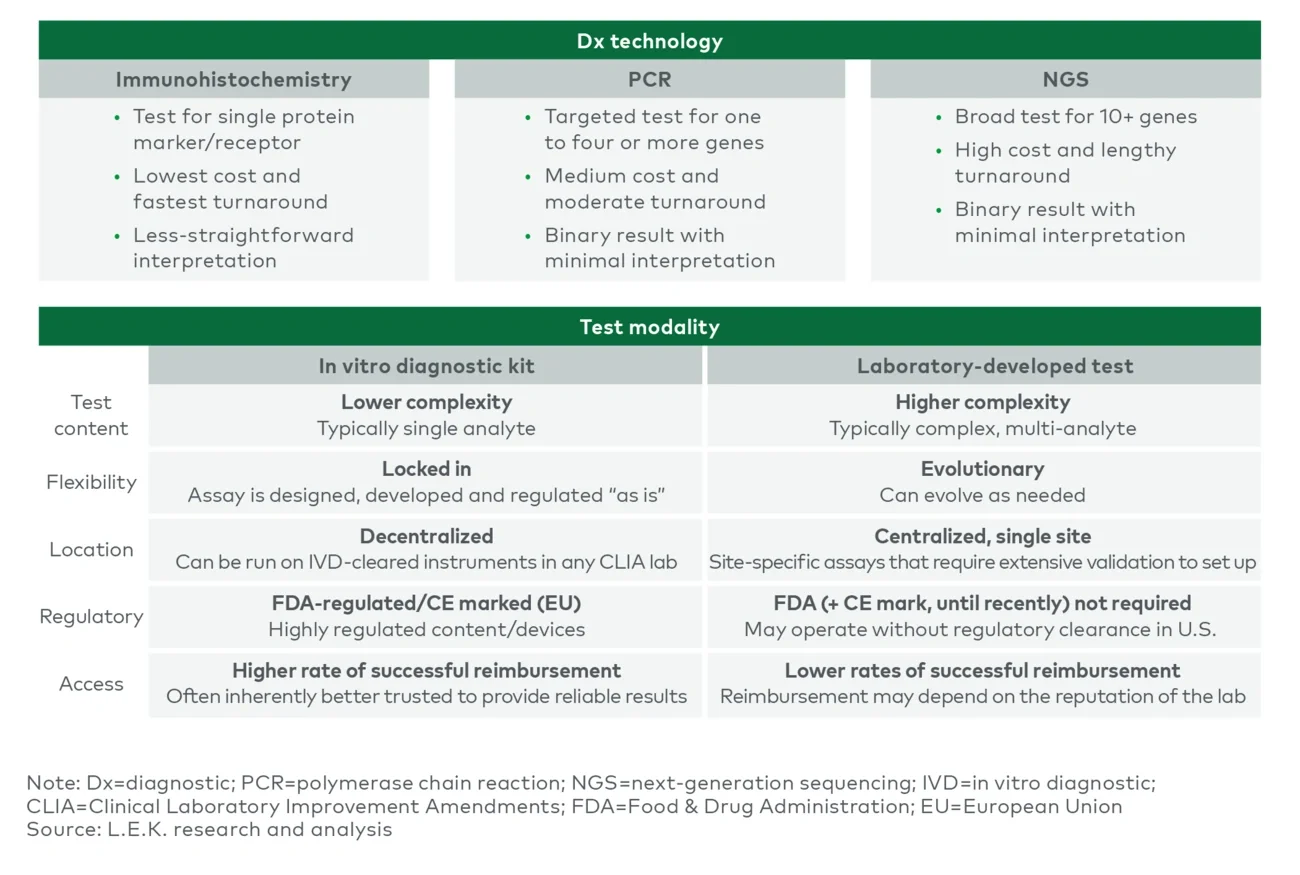
For example, LDTs may face reimbursement issues and require extensive lab validation, yet in the U.S. they often are faster to market and support more numerous and complex BMs because regulatory clearance is not required. Alternatively, IVD kits are FDA regulated, do not support all analytes and face greater competition from other diagnostics, but any CLIA laboratory with the correct instrumentation can run them — and typically enjoy a higher rate of reimbursement.
For some companies, launching and supporting, for example, both LDT and IVD versions of the same Dx adds further complexity and requires additional readiness planning and resources.
4. Build a separate Dx launch strategy.
PM leaders treat Dx launch and therapeutic launch as interconnected yet distinct processes, with different stakeholders and challenges. Because key CDx stakeholders are a diverse group that shares little overlap with therapeutics stakeholders — think pathologists versus prescribing oncologists — targeted outreach is the best way to build awareness and willingness to prescribe. Given the intricacy involved in effective testing (particularly with novel CDx), a launch strategy needs to address the necessary instrumentation or other technology; consider laboratory needs, such as LDT support and sample prep guidance; and take market access into account.
Ideally, companies should consider the interplay between Dx and therapeutic launch strategies when planning for launch. For instance, typical sales incentive structures based on the number of patients on a therapy may be unsuitable in a PM setting, where the number of patients screened for a therapy is potentially a more meaningful measure. Developing a Dx-specific launch strategy can enable widespread adoption and enhance the overall PM opportunity.
5. Leverage partner capabilities purposefully while developing internal expertise.
When empowering critical partners (internal and external) for their expertise in developing, filing and manufacturing Dx tests, biopharma companies should be intentional about expanding specific activities and achieving sufficient oversight. Depending on the organization’s size and capabilities, tasks such as BM selection, test development, study result interpretation or Dx sales may be beyond internal capacity. On the other hand, activities that require close interaction with the therapeutic team (e.g., sample collection and banking) or that are strategic in nature (e.g., market access) may be better managed in-house.
Even when leveraging a partner, launching a Dx requires dedicated internal resources with Dx-specific expertise across the value chain. Specialists who understand both Dx and PM therapeutics are rare and in high demand, requiring early planning and strong retention efforts. Finding the right balance between external expertise and internal foundational knowledge will be crucial to overseeing Dx partners, who may lack the broader in-house context or may not be incentivized to optimize tests or fully invest in launch activities (see Figure 7).
Figure 7
Key development activities ownership: Dx partner vs. pharmacy team
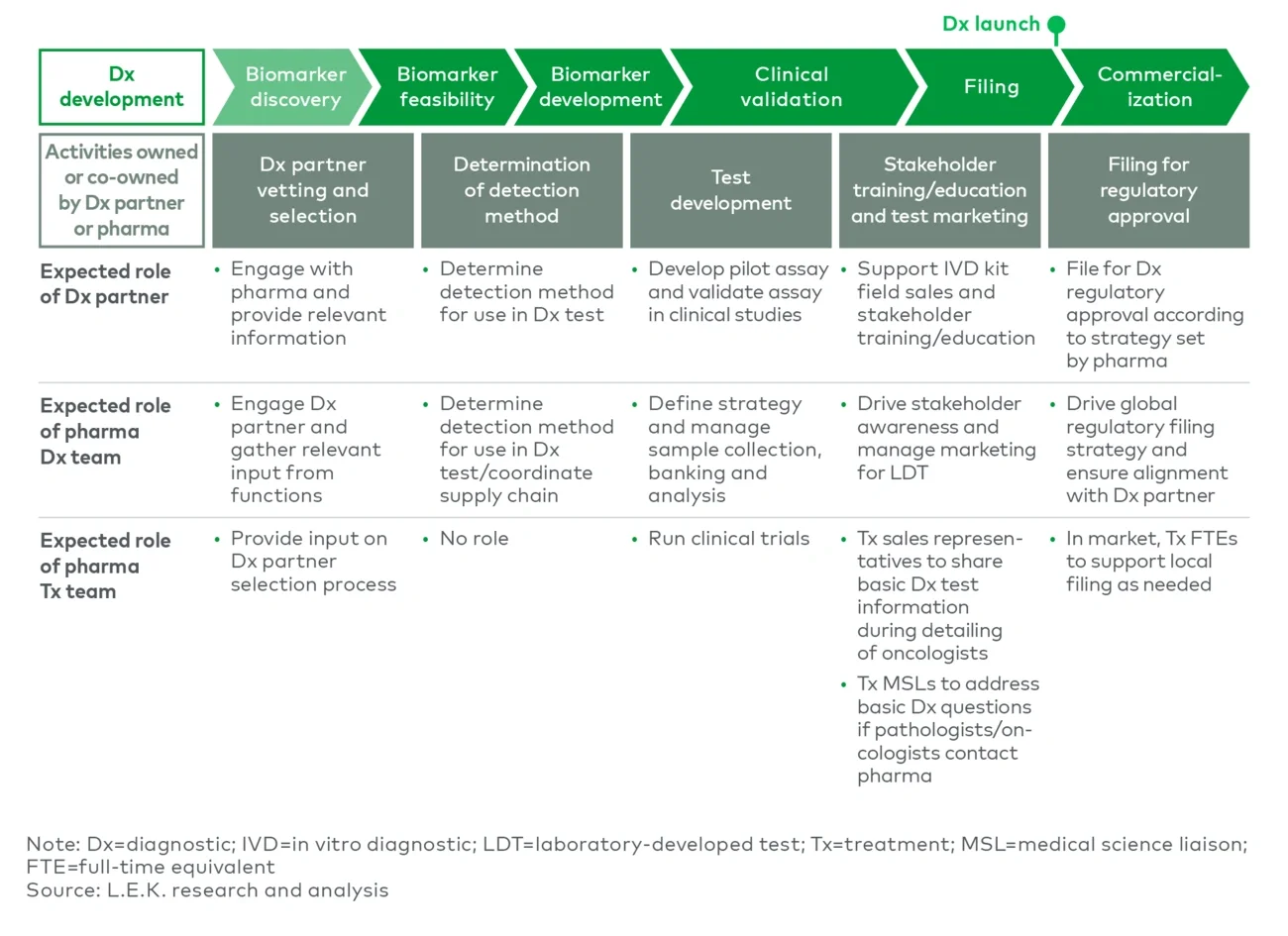
Scaling a Dx ecosystem appropriately can prevent delays in Dx launch planning and execution. Overall, costs incurred when empowering an external partner or developing in-house talent should be viewed as imperative for product success — a strategic investment into that asset franchise rather than just a necessary evil to be minimized.
6. Infuse dedicated Dx expertise throughout the organization.
Successful Dx launch planning requires an environment where Dx needs are supported, integrated across functions, scaled appropriately and prioritized across the value chain. Essential strategies such as adopting an opt-out Dx mindset and investing in early Dx development and launch planning (as discussed earlier) can be up against an inertial mindset around an all-comers approach.
Overcoming pushback from various levels of the company and other headwinds — such as the high costs associated with Dx development and the relatively low direct revenue from Dx versus therapeutic investment — will require unequivocal and sustained support from leadership. In prioritizing Dx investment, savvy PM leaders must also expedite alignment of activities and incentives across Dx and therapeutic teams to generate the cross-functional collaboration needed for a successful launch.
7. Incorporate a thoughtful LCM strategy.
To become leaders in the PM space, companies must adopt a dedicated life cycle management (LCM) strategy that supports continuous evolution and improvement. Early and proactive planning is crucial for a biopharma company’s ability to create sustained impact of BM oncology therapies, but Dx strategy does not end at launch. A meaningful LCM strategy will empower the organization to anticipate next-generation technologies, expanding indications, real-world evidence planning and continuous engagement with key stakeholders — all of which advances the ultimate goal of maximizing therapeutic potential.
L.E.K. continuously monitors pressing issues throughout the biopharma industry landscape in order to deliver innovative lessons, cutting-edge insights and actionable support and strategies that enhance our clients’ ability to achieve their goals.
For more information, or to explore strategies that can unlock new possibilities for your biopharma business, please contact us.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting LLC
Questions about our latest thinking?
Driving Commercial Growth in Wealth Management Through Consumer Duty and AI
Key takeaways
Consumer Duty raises the bar for wealth management: Firms must justify pricing, ensure fair value and provide transparent communication, making data-driven decision-making essential.
AI and analytics drive smarter engagement: Real-time data insights help firms personalise services, optimise pricing and proactively manage client needs, turning compliance into competitive advantage.
L.E.K.'s three-pillar framework enables compliance and growth: Our structured approach ensures regulatory adherence while unlocking commercial opportunities.
A phased strategy ensures long-term success: Firms that accelerate compliance, embed AI-driven decision-making and scale data-driven insights will future-proof their operations and sustain growth.
Consumer Duty is reshaping wealth management. With full AI deployment offering substantial value gains, firms can no longer rely on legacy pricing models, one-size-fits-all services or opaque customer engagement strategies. The new regulations raise the bar, requiring evidence that clients receive fair value, transparent pricing and appropriate financial products.
By leveraging data and AI, firms can create tailored, efficient and scalable service models that meet both regulatory demands and client expectations. This L.E.K. Executive Insights explores how wealth managers can integrate these changes into their business models to create value and sustain competitive advantage.
The rising bar for wealth managers
Regulators are taking a tougher stance. The days of opaque fee structures and one-size-fits-all service models are fading. Consumer Duty mandates that wealth managers justify their pricing, ensure products meet customer needs and communicate in a way that builds confidence rather than confusion. The implications are profound. Firms must re-evaluate their client segmentation strategies, refine their propositions and invest in data-driven decision-making to stay ahead.
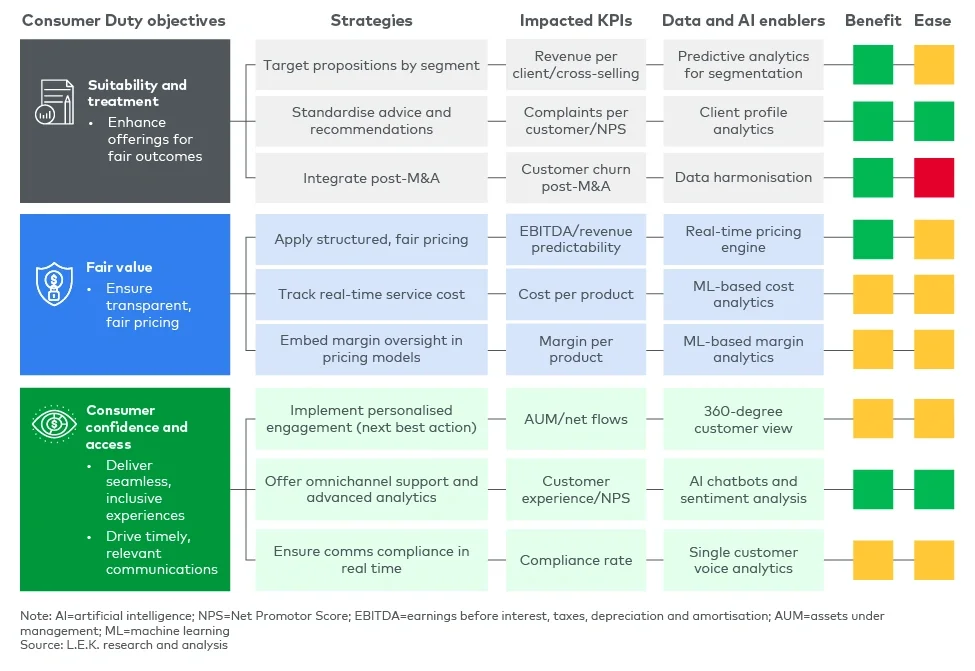
At the heart of compliance is the ability to collect, analyse and act on high-quality client data. Firms that rely on outdated, fragmented data systems will struggle to meet regulatory expectations. L.E.K.’s three-pillar framework — comprising GDPR compliance, a robust data engine, and a structured data dictionary and management approach — provides a foundation for Consumer Duty readiness (see Figure 1).
Figure 1
L.E.K.’s three-pillar framework
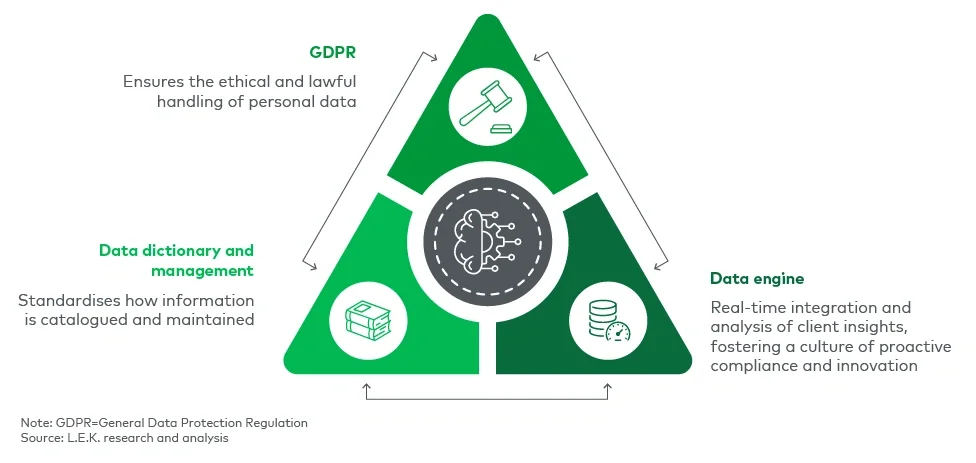
Ensuring adherence to GDPR establishes a compliant baseline, while a well-integrated data engine enables firms to generate actionable insights in real time. Meanwhile, a structured data dictionary and management system ensures consistency and accessibility, reducing the risk of misinterpretation or regulatory lapses.
By embedding this framework, firms can not only meet compliance standards but also unlock commercial growth opportunities through deeper insights into customer behaviour, preferences and risk profiles.
Data-driven compliance and commercial growth
Regulatory adherence and commercial success are not mutually exclusive. Leading firms are already proving that a robust data strategy not only satisfies compliance but also unlocks tangible financial benefits (see Figure 2).
Figure 2
A strong data strategy underpins Consumer Duty readiness
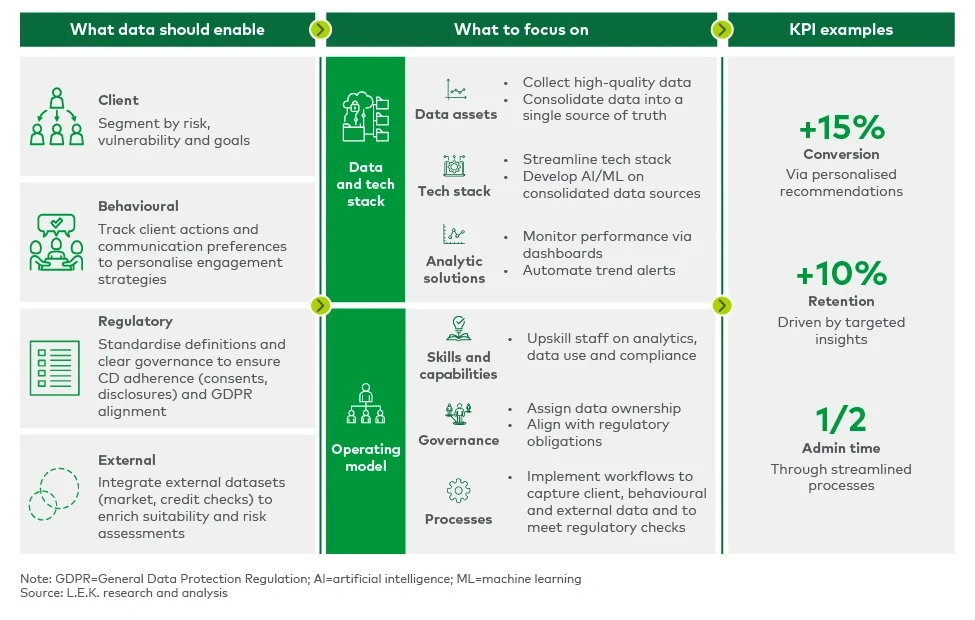
The ability to analyse real-time client data allows firms to adjust pricing dynamically, identify underserved segments and offer more relevant products. AI-driven analytics provide deeper insights into client behaviour, enabling firms to personalise engagement strategies and preempt potential compliance risks. In practical terms, this means:
- Smarter pricing models that optimise revenue without eroding client trust
- Proactive client management, ensuring clients receive appropriate support
- Enhanced service delivery, aligning product offerings with actual customer needs
For comparison, as part of L.E.K.’s ongoing AI Delta work, we provided an exploratory discussion of the potential size-of-the-prize for AI, which aims to help our clients catalyse analysis on the magnitude of the opportunity for their specific businesses (see Figure 3).
Figure 3
The AI Delta in wealth management
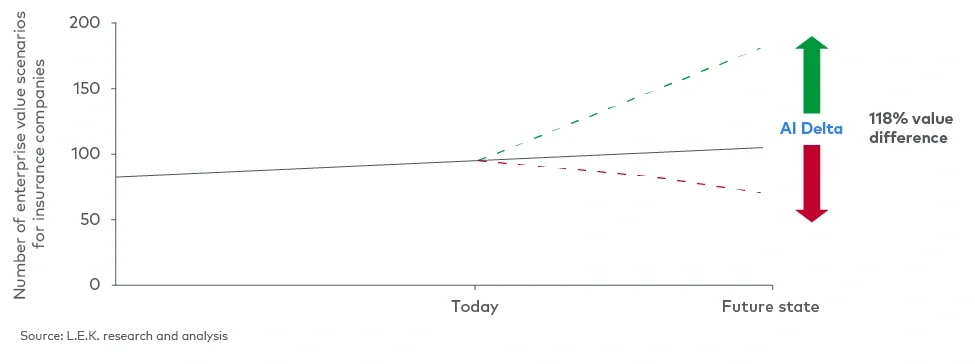
Transforming compliance into competitive advantage
Firms that embed AI and predictive analytics into their operating models will not only meet regulatory expectations but redefine their commercial trajectory. Take, for example, the challenge of pricing. Traditional models rely on broad assumptions, but AI-driven segmentation allows firms to fine-tune their value proposition, ensuring each client receives a service level and product package that reflects their immediate needs and value contribution at the right price point.
Similarly, customer engagement can no longer be reactive. Sentiment analytics can help wealth managers detect when clients are confused or dissatisfied, allowing firms to intervene early. Data-driven decision-making, once a differentiator, is rapidly becoming the minimum standard (see Figure 4). Firms that are slow to evolve will struggle to retain clients who expect personalised, transparent and responsive service.
Figure 4
Using data and AI to boost performance and meet compliance
A three-phase strategy for sustainable growth
To translate Consumer Duty compliance into commercial success, wealth managers must adopt a structured roadmap in which they:
- Accelerate: Assess current data capabilities, address compliance gaps and launch pilot initiatives that demonstrate quick wins.
- Deliver: Deploy AI-driven segmentation, establish real-time monitoring for regulatory adherence and embed compliance tracking into day-to-day decision-making.
- Scale: Expand AI- and data-driven insights across the organisation, integrating compliance seamlessly into business strategy and long-term growth plans.
Firms that follow this trajectory will not only meet the regulatory bar but will also create more resilient, agile and client-centric businesses.
Embracing the future of wealth management
The message from regulators is clear: wealth management must evolve. But firms that see Consumer Duty as a mere compliance hurdle will miss the bigger picture to drive profitable growth and enhanced client relationships. Those that embrace a data-driven, AI-enabled approach will be better positioned to serve their clients, grow their businesses and maintain regulatory confidence.
At L.E.K., we work with firms to navigate these complexities, ensuring compliance while unlocking new commercial opportunities. If you’re considering how to turn Consumer Duty into an advantage, please contact the team.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting
Questions about our latest thinking?
Loading transcript...
Join L.E.K. Consulting’s Education Practice as we explore the defining trends that shaped the U.S. education sector in 2024 and examine the emerging investment opportunities for 2025. In this insightful webinar, Jitin Sethi, Managing Director and Head of L.E.K.’s U.S. Education Practice, and Laura Brookhiser, Managing Director at L.E.K.'s U.S. Education Practice, offer an in-depth look at the evolving U.S. education investment landscape and what lies ahead for investors and operators.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting LLC
Building Resilience in a Commercial-Stage Pharma Company’s Supply Chain
Supply chain resilience is a critical challenge in the life sciences industry, where manufacturing disruptions can take years to resolve and directly impact patient care. The complexity of regulatory compliance, combined with long lead times for scaling production, makes proactive planning and risk management essential.
Success requires deep operational expertise to build resilient supply chains that can consistently deliver critical treatments to patients. A pharmaceutical company engaged L.E.K. Consulting’s Operations and Supply Chain team to help address these challenges.
Tackling the resilience challenge
While all pharmaceutical companies must navigate complex manufacturing and regulatory requirements, the company’s siloed structure — managing drug substance, product and packaging independently — further complicated its situation.
As pharmaceutical pipelines grow more complex, with increasingly specialized treatments and global regulatory requirements, managing supply chains will only become more challenging. Rising costs and shifting market dynamics further underscore the need for proactive resilience strategies. For this company, several structural constraints made it even more difficult to build the necessary resilience into its supply chain:
- Manufacturing complexity: Contract development and manufacturing organizations impose high minimum orders and limited production flexibility.
- Qualification time: Adding production lines or suppliers requires 12-18 months of validation.
- Inventory costs: Safety stock for specialized products can reach tens of thousands of dollars per unit.
- Regulatory burden: Process or facility changes need extensive documentation and approval.
L.E.K.’s approach began with forming cross-functional teams for each product, conducting baseline assessments to align understanding as well as training teams on how changes affect upstream and downstream operations.
Mapping and modeling the supply chain
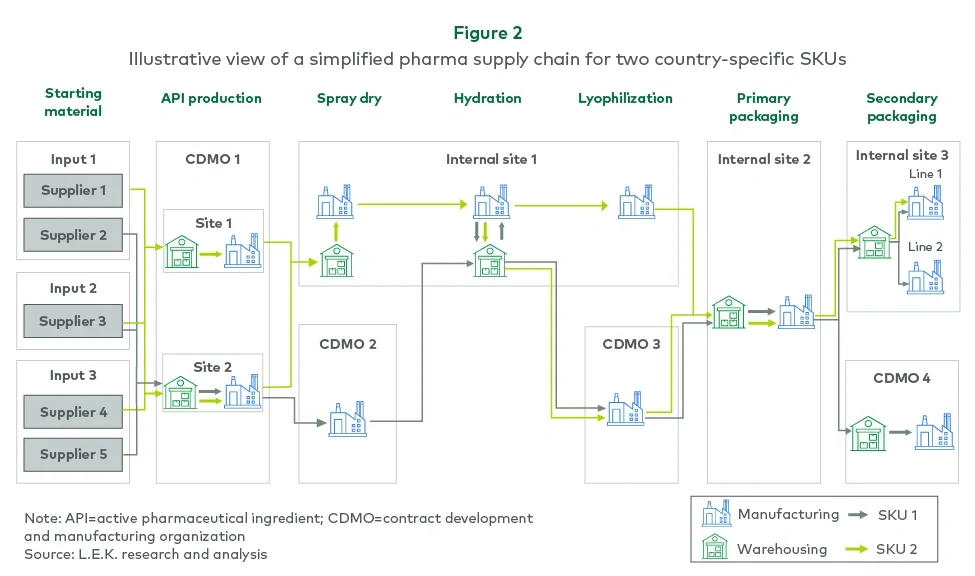
Detailed supply chain mapping revealed hidden vulnerabilities in the company’s siloed operations. Working backward from finished goods, the L.E.K. team created a digital representation showing how demand translated into capacity requirements.
This process uncovered critical constraints — some active pharmaceutical ingredient (API) production suites could only produce certain country-specific SKUs, while certain sterile fill-finish lines were restricted to specific API materials. These limitations showed how overlooked details could compromise resilience during disruptions.
During the engagement, a major disruption at a critical facility provided a real-world test of the supply chain’s resilience. The event threatened to halt production of multiple products, demonstrating how a single point of failure could quickly cascade throughout the network.
This concrete example helped reinforce executive buy-in for implementing more robust contingency measures and extended coverage across the portfolio (see Figure 1).
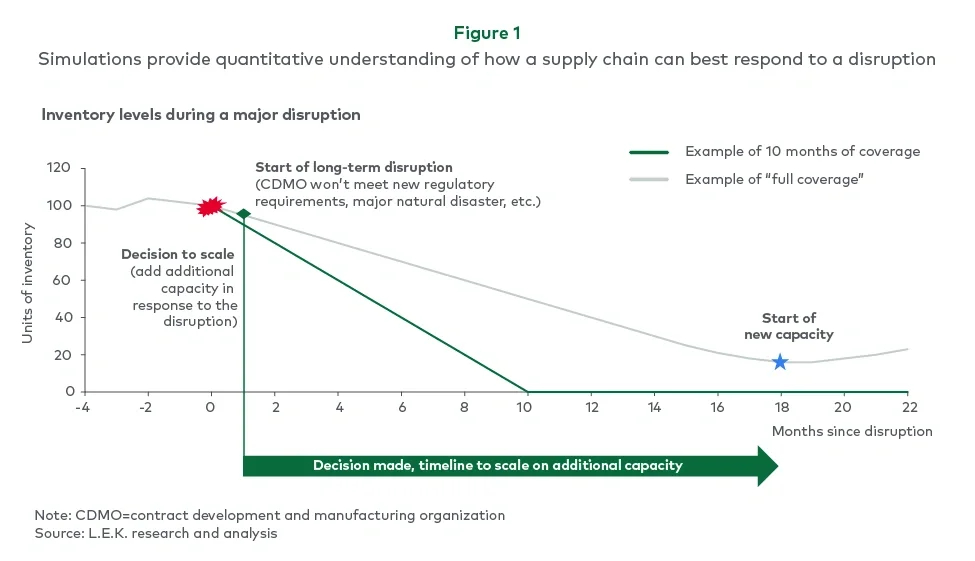
After mapping and verifying the supply chain — including parameters such as batch size, frequency, storage requirements, shelf life and bill of materials — the team built an end-to-end network model using a digital twin. This model replicated complex supply chain interactions over time, allowing L.E.K. to dynamically measure how inventory levels (both work-in-progress and finished goods) responded to shifting demand and potential disruptions.
The analysis revealed several critical vulnerabilities:
- Single-source dependencies: Key components relied on suppliers without scalable capacity.
- Site concentration: Redundant capacity often existed within single facilities, creating sitewide risks.
- Overlapping redundancies: Backup production lines serving multiple products created bottlenecks.
- Packaging vulnerabilities: Single-source bulk packaging threatened country-specific SKUs.
- Warehouse risks: Single storage sites created upstream production failure points.
- Limited contingency plans: Most products lacked emergency plans for alternative production.
- Demonstrated capacity gaps: Capacity levels used in production planning were overstated and did not represent the proven output, accounting for production inefficiencies and bottlenecks.
Turning insights into action
Resilience planning often feels nebulous, with generic solutions such as adding backup plans that fail to provide the specificity executives need. As well as investing in modular equipment, the L.E.K. team implemented other specific solutions across different product lines:
- Diversified suppliers: One product team established dual sourcing for critical components.
- Expanded redundancy: Another team invested in a mobile production lab, adding six months of buffer capacity.
- Created an inventory strategy: Targeted strategies brought inventory coverage from partial to full.
- Accepted strategic risk: Teams accepted certain risks where mitigation costs outweighed potential benefits.
The team approached investment decisions by prioritizing the value of protection over the likelihood of disruption. For example, when evaluating a $3 million investment to protect a $20 million revenue drug, the emphasis on ensuring patients never missed a dose justified the cost, underscoring the critical human impact behind the decision.
Delivering impact
The executive team gained clarity and confidence in understanding trade-offs and implementing solutions. During the engagement, the client made structural improvements that allowed the company to take immediate action, such as increasing the months of coverage for its average product, which provided critical breathing room to address disruptions. These early improvements laid the groundwork for implementing key longer-term strategies developed during the engagement:
- Implementing backup production lines and enhanced inventory strategies for high-priority products
- Launching supplier diversification initiatives to minimize single-source vulnerabilities
- Improving network resilience for proactive disruption management
Beyond these immediate actions, the client now has an advanced supply chain resilience model that enables ongoing scenario planning and risk management, ensuring the company can proactively adapt to future challenges. This approach not only protected critical operations and ensured continuous patient access to lifesaving therapies but also created a foundation for long-term supply chain resilience.
L.E.K.’s expertise in pharmaceutical and life sciences supply chains enables clients to achieve:
- Reduced supply chain disruption risk
- Decreased critical drug shortages
- Optimized network costs
- Enhanced supply flexibility
- Accelerated time to market for therapies
- Sustainable competitive advantages through operational excellence
To learn how L.E.K. can help build resilience in your supply chain and protect your ability to deliver critical therapies to patients, contact us today.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting

America’s infrastructure is under pressure — and that’s creating a surge of opportunity. The architectural, engineering and construction (AEC) sector is at the center of transformative shifts driven by federal investment, the clean energy transition, rising regulatory complexity and rapid population movement. With billions in long-term funding being deployed, the market is primed for growth.
But opportunity requires focus. Investors must prioritize understanding where funding is truly durable, identifying operational upside and pursuing growth that aligns with core capabilities. In a highly fragmented landscape, differentiation, local relationships and scalability are critical advantages.
Talent is also a defining factor. Recruiting, retaining and developing the right people is essential in a sector where expertise drives value. Meanwhile, AI is emerging as a powerful lever to improve efficiency, manage risk and unlock new potential — but only with the right strategic and organizational approach.
Download our report for further analysis into this dynamic investment landscape.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting LLC
Removing Barriers to Healthcare Growth: Leveraging Strategic Review to Address Constraints
Key takeaways
An organisation-wide review, encompassing clinical services and support functions, often reveals key inefficiencies and missed opportunities for expansion.
Strong cooperation between clinical leaders and operational/financial stakeholders is key to successfully implementing any necessary changes.
Activity-based costing (ABC) and zero-based budgeting (ZBB) provide the granular insights needed to differentiate between essential and expendable services.
Underperforming services can be restructured through partnerships, consolidation or rightsizing to maintain patient access while reallocating resources for core growth.
Healthcare organisations face increasing pressure to optimise operations, expand access and enhance patient outcomes against a backdrop of changing regulations, tariff pressures and evolving patient needs. For multi-service healthcare providers — often juggling multiple specialties, facilities and care delivery models — identifying and resolving constraints that hinder core service growth is critical to clinical excellence, operational performance and financial sustainability.
This Executive Insights draws on L.E.K. Consulting’s recent project experience to illustrate how a structured strategic review can uncover underperforming or non-contributory services and streamline clinical support functions and overheads.
Beyond merely cutting costs, a strategic review lets healthcare organisations redeploy resources where they are needed most, improve care pathways and deliver a better patient experience.
Context and analysis
For many healthcare providers, constrained resources — clinical workforce shortages, insufficient facility space and tight budgets — can hamper the development of core services. This was especially true for one large healthcare network we advised, where loss-making service lines eroded profitability and impeded much-needed investments in higher-value clinical areas.
A comprehensive strategic review uncovered multiple non-contributory or underperforming service lines and under-optimised support functions. Taken together, these areas diverted resources away from promising growth segments, leading to inefficiencies and hindered patient outcomes.
Approach
A structured methodology — tailored to the complexities of both clinical operations and administrative services — is critical for sustainable improvements. This involves establishing clear financial visibility, identifying underperforming areas and aligning resources with the organisation’s strategic priorities.
Care delivery
A key first step is to establish a financial baseline using activity-based costing (ABC). Shared cost centres — such as pharmacy, imaging, operating theatres and wards — often obscure the financial contribution of individual services. ABC clarifies how each service line contributes to overall margins, helping to identify high-impact areas for strategic reallocation or redesign.
Once financial clarity is established, the next step is to review improvement potential by assessing demand, patient needs, market opportunities and performance gaps. This holistic perspective allows organisations to determine which service lines should be repositioned, reinvested in or discontinued. Making these data-driven decisions ensures that brand equity is preserved while strengthening the hospital’s long-term market position.
Some further steps healthcare providers can take to improve care delivery include:
1. Strengthening revenue streams
- Enhancing scheduling, patient experience and follow-up processes to improve patient throughput
- Addressing inefficiencies in internal and external referrals to reduce patient leakage and improve resource utilisation
- Reviewing physician incentives to align with quality, productivity and patient experience objectives
2. Optimising resource allocation
- Assessing potential volume reallocation, specialisation and efficiency improvements
- Optimising theatre utilisation, reducing average surgical times and shifting minor surgeries to procedure rooms to enhance efficiency.
- Improving inpatient ward organisation through better coordination with operating theatre and admissions teams, aligning staffing levels with demand, and actively managing patient length of stay.
Care support
Beyond clinical operations, optimising administrative and support functions is essential to reducing inefficiencies and reallocating resources effectively. A zero-based budgeting (ZBB) review provides a structured approach to evaluating support services from the ground up. This method helps eliminate redundant processes, redeploy staff and identify opportunities for outsourcing non-core functions.
A ZBB review should address key questions such as:
- Are we doing the right thing?
- Are we using the right resources?
- Do we need to evolve our ways of working and care models?
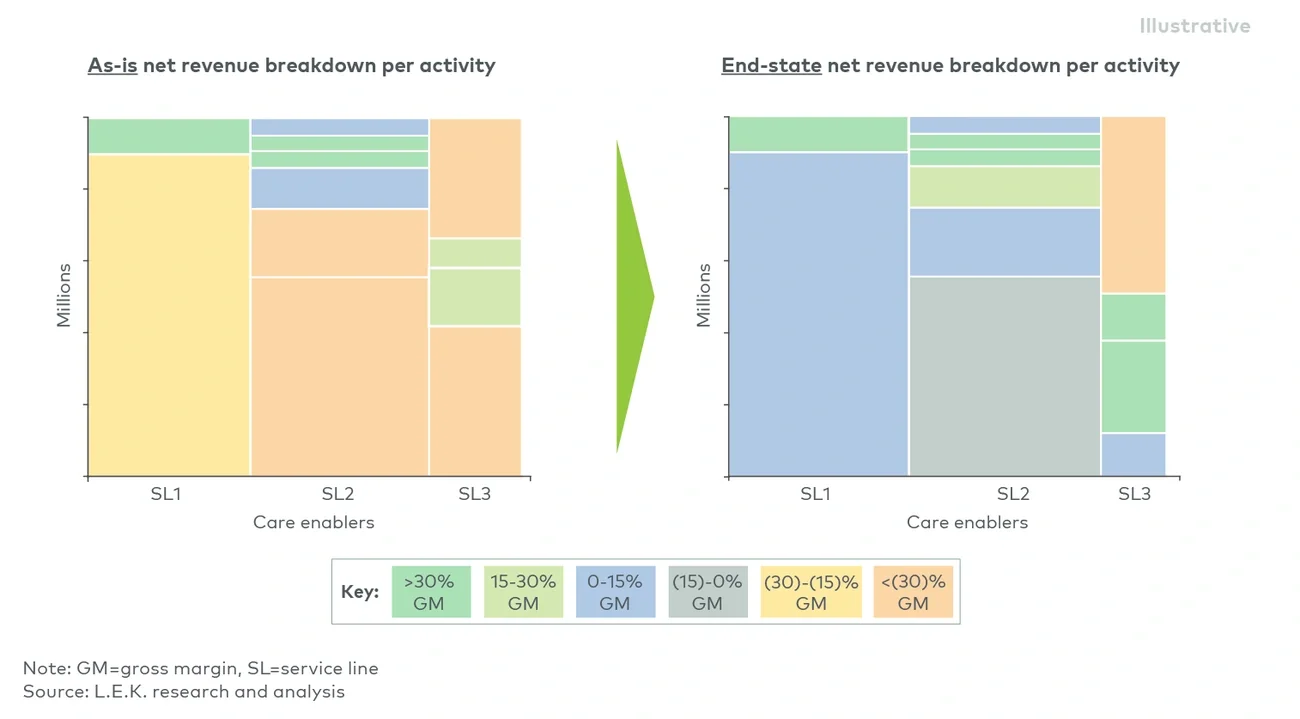
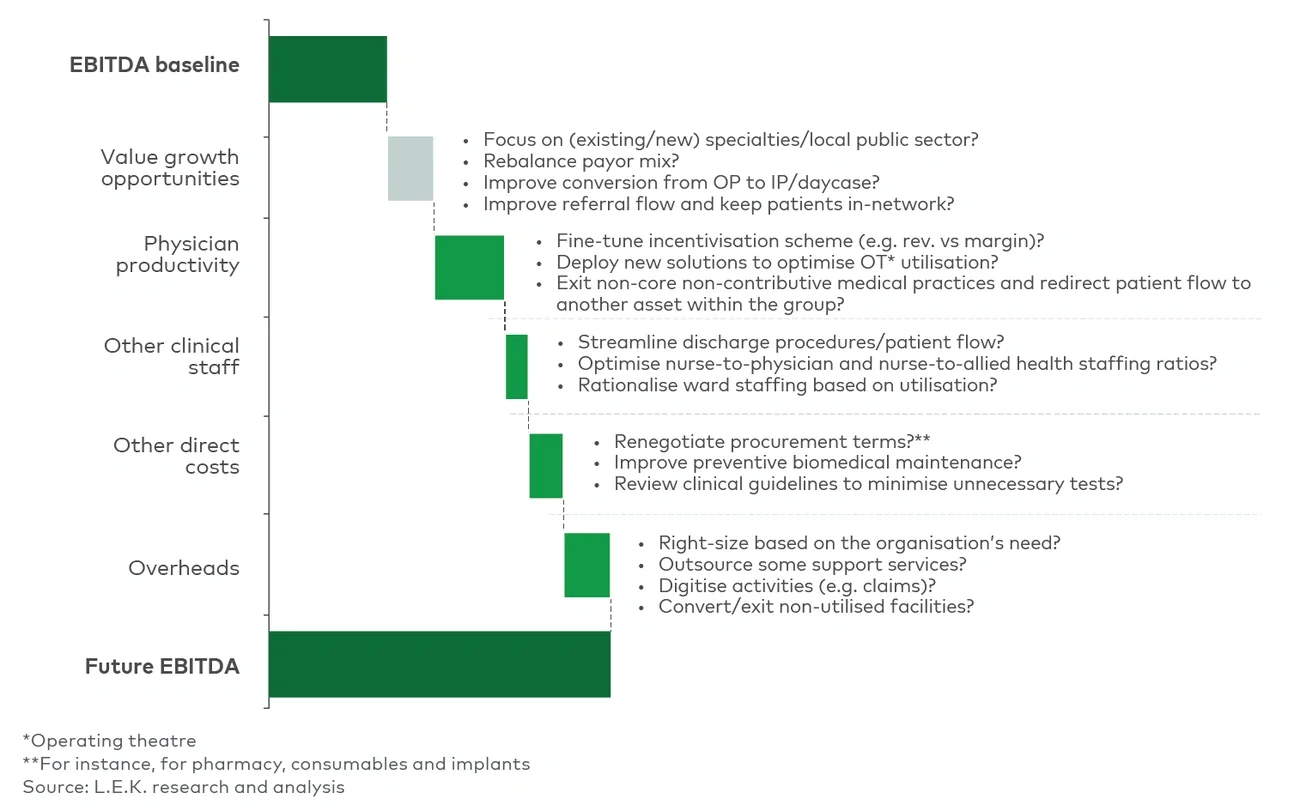
By systematically reviewing these areas, organisations can streamline administrative tasks, increase utilisation of critical staff and reduce non-value-adding expenses (see Figure 1).
Figure 1
Zero-based budgeting enables a clear view of your as-is state and the impact of improvements
Some growth opportunities include:
- Optimising group vs site-level overheads: Centralising support functions such as finance, HR and administrative tasks helps reduce duplication and frees on-site teams to focus on activities that add value.
- Embracing digital and AI-driven solutions: Quick wins include implementing generative AI for call-centre interactions (automating scheduling and routine enquiries), AI-enabled physician scribes to handle documentation and mobile platforms for real-time data capture.
- Supply chain management: Consolidating procurement and centralising stock management can cut costs, reduce on-site storage needs and improve overall resource utilisation.
- Outsourcing non-core functions: Third parties can often handle facilities management, biomedical equipment maintenance and transaction services (e.g. call centres, billing) more cost effectively.
- Simplifying, standardising and digitising medical records: Reducing reliance on paper records minimises workload for administrative teams, consolidates storage needs and improves retrieval times, ultimately enhancing patient care and operational efficiency.
Success factors
Achieving meaningful improvements requires balancing financial, operational and clinical considerations.
A dual approach is essential for aligning with stakeholder priorities. Findings and recommendations should be tailored to different audiences — financial stakeholders, for instance, may focus on ward workforce optimisation, while clinical leaders prioritise nurse-to-bed ratios. Effective communication across these groups fosters acceptance and smoother project execution.
Strong clinical buy-in is also crucial to ensure alignment. While increasing outpatient and inpatient volume may be straightforward, operational changes require deeper collaboration. Additionally, success should be measured not just through financial outcomes but also through improvements in the patient pathway.
Enhancements such as streamlining reception and pre-registration processes, optimising bed discharge procedures and expediting admissions contribute to improved patient experience and higher throughput.
To take a holistic view of each service line, you must consider the following:
- Market potential: Our client had a renowned service line that had recently become loss-making due to regulatory changes. Rather than cutting back, we reviewed market potential and rethought the care model to improve patient throughput and patient mix to preserve core capabilities and enhance profitability.
- Core capabilities: Certain services are essential for maintaining the hospital’s overall appeal and capabilities, even if their standalone profitability is questionable.
- Indirect contributions: Even a non-core loss-making service can still bolster walk-ins or diversify payer exposure, contributing positively to overhead coverage. It can also support fixed-cost structures (e.g. specialty clinics). A thorough evaluation is required to understand the direct and indirect benefits of specific services and their overall contribution.
- Transfers, not closures: If an activity truly cannot be optimised or sustained, consider transferring it (via partnerships or network consolidation) rather than eliminating it altogether.
L.E.K.'s strategic review not only eliminates barriers to healthcare growth but also empowers management teams to thrive (see Figure 2).
Figure 2
Key themes and questions to address at the hospital level
Conclusion
Reviewing and realigning your strategy is more than a cost-cutting exercise, as it enables you to focus on ensuring long-term sustainability and enhancing care delivery. By systematically addressing constraints, healthcare organisations can seize new growth opportunities, optimise operational efficiencies and ultimately deliver superior patient care.
At L.E.K., we specialise in helping healthcare organisations define their optimal service portfolios and implement growth strategies tailored to their unique challenges. Contact us to explore how our structured approach and industry expertise can help you succeed.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting
Questions about our latest thinking?
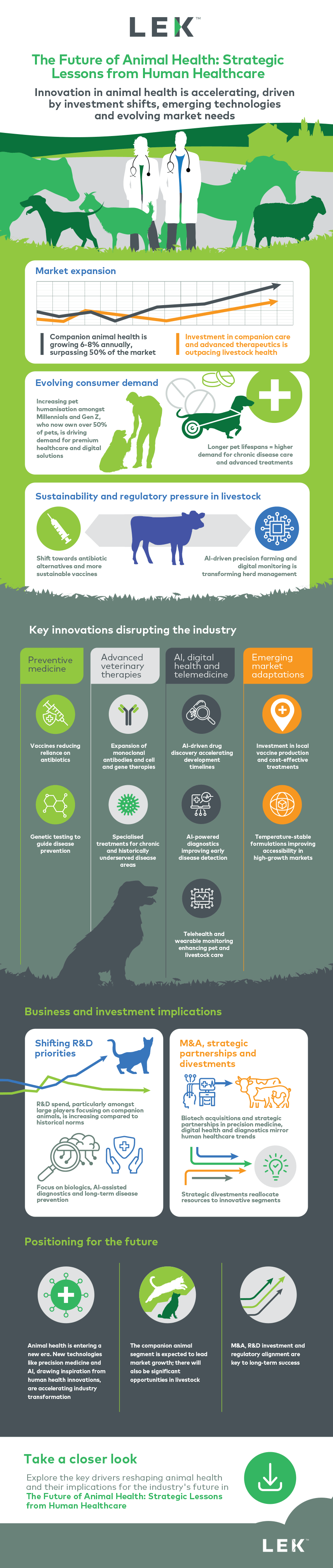
Animal health is entering a period of accelerated transformation. Inspired by developments in human healthcare, the sector is being reshaped by emerging technologies, evolving consumer expectations and shifting investment strategies. Growth is being driven by the companion animal segment, which now accounts for more than half of the market, while demand for advanced therapies, AI-enabled diagnostics and preventive care is rising across both pet and livestock populations.
This infographic highlights the most significant forces disrupting the industry, from innovation in vaccines and monoclonal antibodies to the impact of telemedicine and regulatory change. It also outlines the commercial implications for companies across the value chain, as strategic partnerships, R&D investment and targeted divestments reshape the competitive landscape.
Download the infographic for a visual overview of these changes and access our full Executive Insights for deeper strategic guidance on where the market is heading and how to respond.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting





























































