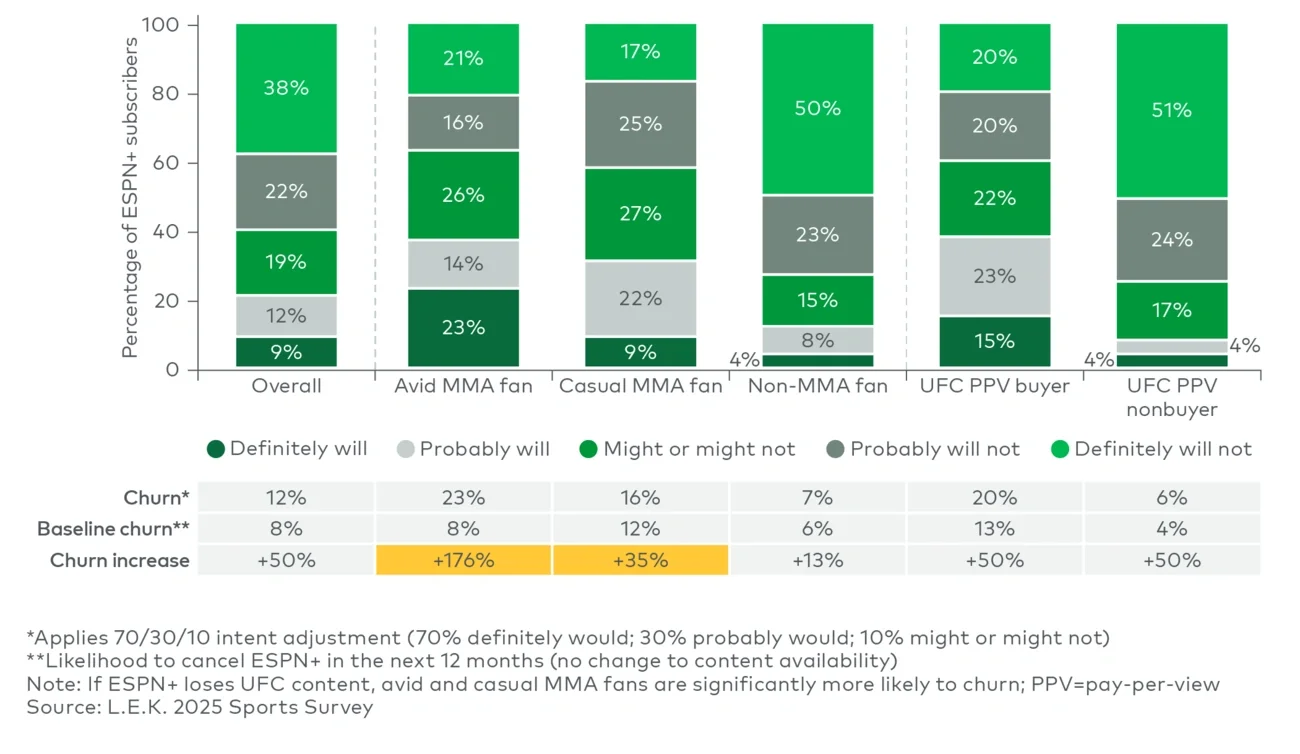
Avid MMA fans — who represent 21% of ESPN+ subscribers — stated they purchase an average of 3.5 UFC PPV events annually, according to our 2025 Sports Survey. Another 17% of subscribers are casual MMA fans, purchasing around 1.5 events per year, compared to just 0.8 events for nonfans. While ESPN remains the front-runner for UFC’s domestic rights, other potential bidders are in the mix. Warner Bros. Discovery’s B/R Sports is often cited as a contender, though its existing relationship with All Elite Wrestling could complicate a deal with TKO-owned UFC.
Netflix has also been mentioned as a potential partner for UFC, especially given its recent World Wrestling Entertainment agreement. However, in our estimation, the high price tag and PPV distribution model make it unlikely that Netflix will be a serious contender for UFC’s domestic rights. UFC’s international rights, however, are a more natural fit — Netflix’s global footprint makes it an ideal partner to consolidate fragmented rights, improve international monetization and accelerate UFC’s overseas audience growth.
MLB: The declining value proposition
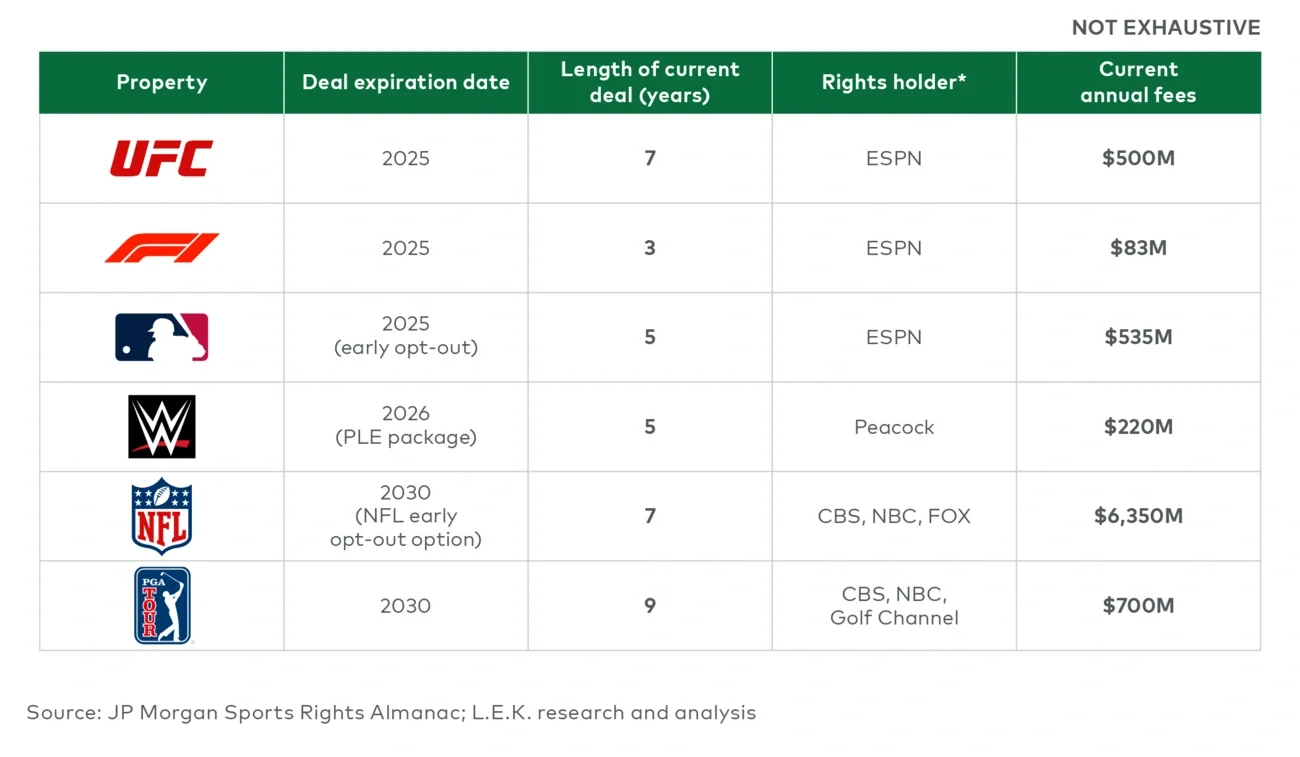
ESPN’s decision to opt out of MLB’s national rights underscores the sport’s challenges at the national level in the streaming era. While interest in local teams and regional broadcasts remains relatively strong, national ratings have declined approximately 3% annually since 2013. At the same time, the ESPN/MLB rights deal was relatively expensive — with an estimated cost per viewer hour of $2.74 — materially higher than other recent agreements.
Meanwhile, newer entrants like Apple TV and Roku are paying significantly less in total for meaningful regular season content. Apple holds rights to Friday Night Baseball, while Roku airs Sunday Leadoff Baseball — both featuring one game per week over roughly 18 weeks from May through early September. These pricing disparities highlight the sport’s uncertain value at a national level, especially as platforms prioritize content that drives engagement and retention.
With the national rights picture in flux and the regional sports network (RSN) model continuing to erode, MLB teams are increasingly exploring alternative distribution strategies, including direct-to-consumer and over-the-air options. These models may improve access and exposure but currently generate far less revenue than legacy RSN deals do.
(Stay tuned for part two of this series, where we’ll explore the collapse of RSNs and the shifting dynamics of the local sports media ecosystem.)
A fragmented sports media landscape ahead
As the sports media ecosystem continues to fragment, several critical implications are emerging for both platforms and leagues:
- Live sports are foundational. Streaming platforms that lack marquee sports content risk losing audience share to competitors that invest in tentpole events and use them to anchor subscriber growth and retention.
- Leagues hold near-term leverage — but it may not last. Fast-growing, globally appealing leagues like UFC and Formula One are well positioned to drive meaningful increases in average annual value during this rights cycle. But as the market shifts fully to streaming, that pricing power may fade — especially with slowing subscriber growth or platform consolidation.
- For fans, fragmentation is a mixed bag. As sports content splits across multiple platforms, access becomes more complicated, and subscription costs may rise. Yet fans also benefit from more flexible viewing options and the potential for richer, more personalized experiences.
These shifts signal a new era in sports broadcasting — one defined not by a single dominant platform, but by a dynamic, multichannel marketplace shaped by shifting economics, evolving fan behavior and high-stakes content decisions.
Coming next
The fragmentation of national rights is only part of the story. The collapse of RSNs presents an even greater challenge, as leagues and teams work to replace billions in lost revenue. In our next article, we’ll examine how the decline of pay TV is reshaping local sports broadcasting — and what it means for the future of regional media rights.
For deeper insights into sports media rights, platform strategy and content monetization, reach out to L.E.K.’s Media & Entertainment practice. Our team specializes in sports and live entertainment, direct-to-consumer models and media rights negotiations — helping clients navigate the evolving sports landscape with data-driven strategies.
For more information, please contact us.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2025 L.E.K. Consulting LLC