Australia is a global leader in bauxite mining, alumina refining and primary aluminium production, yet almost all aluminium scrap generated domestically – around 440 kilotons (kt) each year – is exported for remelting offshore. Establishing domestic recycling and remelting capability is therefore not about fixing a waste problem – it is instead about capturing economic value, strengthening sovereign capability, reducing emissions intensity and meeting growing customer demand for lower-carbon, recycled materials.
With coordinated policy action, Australia can transform a lost opportunity into a national advantage. This Special Report, produced in partnership with the Australian Aluminium Council, outlines how this can be achieved.
Download the full analysis now.
L.E.K. Consulting’s Global Education Practice examines the key trends shaping education investment in the APAC region. This webinar provides insights into major transactions from 2025 and explores the themes expected to drive activity in 2026.
Watch the recording to stay informed on the evolving market dynamics and opportunities in the sector.
Loading transcript...
Background and challenge
Global wealth continues to expand, with assets increasingly concentrated among high- and ultrahigh-net-worth individuals. This shift is creating new opportunities in private banking, where clients expect highly personalized services, differentiated advice, and access to alternative investments. Growth, however, is uneven: While wealth expands rapidly in Asia-Pacific and the Americas, European markets remain relatively stagnant. In this environment, smaller private banks can compete through agility and specialization, while mid-scale institutions often struggle to balance growth ambitions with structural cost constraints.
A boutique private bank with a strong institutional heritage and differentiated client proposition faced increasing pressure to translate its positioning into sustainable financial performance. While client loyalty remained high, growth initiatives were fragmented and leadership lacked a clear strategic roadmap to capture emerging wealth opportunities across regions and products.
The bank engaged L.E.K. Consulting to help define its long-term strategic direction, align the leadership team around a clear growth vision, and develop a value-creation roadmap grounded in quantified financial outcomes. Leadership sought clarity on which growth initiatives would drive sustainable revenue expansion while strengthening profitability and operational scalability.
Approach and recommendations
L.E.K. conducted a structured assessment of the bank’s strategy, business model, performance and operating structure to establish the foundation for a clear value-creation roadmap.
The review surfaced two critical findings. First, the bank’'s cost base reflected legacy operating assumptions rather than forward-looking revenue realities. Staffing levels, governance, processes, technology, and service models were calibrated for historical growth, resulting in a complex operating structure that limited the bank’s ability to pivot with market conditions and drive profitability. Second, high complexity and an inefficient operating model led to an increasing cost-to-income ratio (CIR), which in turn limited growth. The bank was working harder just to maintain the status quo. Additionally, with capital invested in an inefficient operating model, profitability declined, and return on equity (ROE) fell. The operating model also became less resilient as complexity increased.
L.E.K. worked with the executive leadership team to refine the bank’s strategic vision and mission, aligning leadership around a differentiated positioning in the global private banking landscape and clarifying the strategic priorities required to support long-term growth. Additionally, the team developed a focused go-to-market strategy across key regions. This included market sizing and growth analysis in priority wealth markets, identifying target client segments, and evaluating regional expansion pathways, including licensing requirements and operational setup considerations.
To develop an effective growth plan, L.E.K. helped the bank evaluate how current initiatives could deliver stronger financial outcomes within its existing operating model. This included developing a segmented profitability view across client groups and products, identifying structural cost drivers in legacy servicing and delivery models, and highlighting additional revenue levers such as Lombard lending, structured financing solutions, and pricing discipline to strengthen client wallet share and revenue growth.
Results
L.E.K.’s analysis helped the bank prioritize initiatives that could deliver the greatest measurable impact, while streamlining its current cost structure. By identifying areas of complexity and excess in the legacy model and pinpointing initiatives with the highest potential returns, we helped the client develop a value-creation plan that:
- Aligned the operational cost base with forward-looking growth scenarios;
- Qualified execution milestones tied to CIR and ROE improvement, thereby embedding financial discipline into decision-making
Implementation of this roadmap delivered:
- Achievement of a 13% ROE target
- 10% compound annual growth in assets under management
- A 20 percentage point improvement in CIR
Key takeaways
The U.S. optometry market is entering a phase of structural growth, with value-driven expansion becoming increasingly important alongside continued volume growth.
At the same time, fragmentation at the practice level is colliding with rising operational complexity, creating strong demand for scaled, trusted partners across supply, fulfillment, reimbursement and care delivery workflows.
Direct-to-patient fulfillment now accounts for approximately 50% of U.S. contact lens sales, with continued share gains expected over time.
Ultimately, the U.S. market is not being disrupted so much as it is being structurally rewired.
Introduction
The U.S. optometry market is entering a phase of structural growth, with value-driven expansion becoming increasingly important alongside continued volume growth. While underlying demand for vision correction remains resilient, the sources of growth are shifting toward premiumization, digital enablement, medicalization and workflow efficiency. At the same time, fragmentation at the practice level is colliding with rising operational complexity, creating strong demand for scaled, trusted partners across supply, fulfillment, reimbursement and care delivery workflows.
Key trends
U.S. vision-care demand remains resilient, with growth increasingly driven by mix shift alongside volume
Underlying demand for vision care in the U.S. remains strong, supported by durable demographic and lifestyle trends. Rising myopia prevalence, increasing screen exposure and a growing population over age 45 continue to drive steady utilization of eye exams and corrective products. However, market growth is no longer driven primarily by rising exam volumes alone.
Volume growth remains supported by demographic and behavioral tailwinds, but an increasing share of growth is coming from shifts in modality and pricing mix, as patients migrate toward higher-value products such as daily disposable contact lenses and premium ophthalmic lenses with specialty coatings and materials. As a result, the U.S. market for refractive eye care and optical products is expected to continue expanding in value terms over the coming years, broadly in line with recent historical performance, with unit growth continuing at a consistent pace.
The focus of growth is shifting from exam volume to the delivery of more sophisticated and clinically tailored solutions. Practices that can incorporate evolving products and care pathways into routine care are better positioned to participate in this shift. One example is the growing attention to myopia management, especially among pediatric and adolescent patients. Rather than simply correcting vision, some practices are beginning to offer structured myopia management programs designed to slow progression over time through specialized lenses, pharmaceuticals and ongoing monitoring.
Adoption remains early stage across much of the U.S., especially relative to certain international markets. Because these programs are intended to moderate progression rather than eliminate the need for correction altogether, their impact is more likely to reshape the timing and intensity of care rather than to materially reduce long-term demand.
The U.S. optometry ecosystem remains fragmented at the point of care but more consolidated upstream
There is ongoing expansion by private equity-backed groups and national retail optical chains, both of which are expected to continue playing a durable role in access and scale. But, optometry in the U.S. remains fundamentally fragmented at the point of care. More than 70% of U.S. optometrists practice in independent or small-group settings, retaining significant autonomy over clinical and operational decisions.
This fragmentation contrasts sharply with increasing scale upstream across contact lens distribution, ophthalmic lab services and digital enablement platforms. Independent practices face accelerating operational complexity, including managing multiple manufacturers and labs, navigating managed vision care reimbursement and coping with persistent staffing shortages — all while meeting patient expectations for faster turnaround and home delivery. In response, eye-care providers (ECPs) are relying more on partners that can centralize and simplify these functions without constraining clinical choice or clinical autonomy.
Market behavior reflects this reliance. Relationships between U.S. ECPs and their primary distributors or lab partners are typically long-term, with most ECPs reporting relationships lasting four years or more. Switching remains relatively rare and is usually triggered by breakdowns in reliability, availability or economics rather than incremental price differences. Hence, influence in the ecosystem is accruing to platforms that are embedded in daily workflows and operational decision-making rather than those competing on transactional terms.
Direct-to-patient fulfillment is growing but reinforcing, not replacing, U.S. optometrists
Patient expectations in the U.S. have shifted decisively toward convenience. Home delivery, online reordering and automated reminders are becoming table stakes across contact lenses and, increasingly, prescription eyewear. Direct-to-patient fulfillment now accounts for approximately 50% of U.S. contact lens sales, with continued share gains expected over time.
Importantly, this shift has not resulted in widespread disintermediation of optometrists. U.S. patients place significantly greater trust in their eye-care provider than they do in manufacturers or online retailers when selecting contact lens brands. Pure-play online retailers, although meaningful, have seen their market share largely stabilize since the pandemic rather than continuing to expand aggressively.
Instead, momentum is becoming more concentrated in provider-enabled omnichannel models, where digital platforms manage ordering and fulfillment while the provider remains the clinical and relational anchor. On the contact lens side, the ECP-enabled direct-to-patient channel is larger than pure online retail and continues to outpace it in growth. A similar dynamic is emerging in eyewear: Patients value the ability to combine in-office fitting and clinical guidance with digital reordering and fulfillment. These models allow practices to retain patient relationships and still meet rising expectations for convenience, reinforcing the optometrist’s central role in the U.S. vision-care system.
The shift toward provider-enabled omnichannel models is also supported by incremental advances in digital tools that reduce friction in product selection and replenishment, including improved virtual try-on capabilities and streamlined prescription workflows. At the same time, ongoing innovation in lens materials and wearable technologies continues to support premiumization while keeping the optometrist central to product recommendation and care decisions.
U.S. optometry is becoming more medical and more operationally complex
In the U.S., optometry continues to expand beyond routine vision correction into higher-acuity, reimbursement-driven care. Many practices are now managing chronic eye conditions, providing medical treatments and participating more actively in post-operative and specialty care pathways. This expansion reflects both evolving clinical capability and capacity constraints on the ophthalmology side, where limited specialist availability and growing demand for higher-acuity care are pushing more routine medical eye care into optometric settings. As a result, the historical boundary between product-driven vision correction and medical eye care is blurring, with retail-focused platforms introducing medical services to enhance patient lifetime value and visit frequency, while medical-first groups integrate optical capabilities to capture downstream spend and improve continuity of care.
This expansion is being supported by continued advances in in-office diagnostic imaging and clinical technologies, which enhance detection, monitoring and longitudinal management within the optometrist’s scope. Highest-acuity interventions remain within ophthalmology, but improved diagnostics and workflow tools are enabling optometrists to manage a broader range of conditions with greater confidence and consistency.
This vertically integrated evolution supports higher revenue per patient and more diversified reimbursement. It also changes the operating model required to deliver care effectively. Managing a broader mix of retail and medical visits increases demands around documentation, billing accuracy, scheduling and care coordination, and it raises expectations for clinical quality and turnaround times. Success in these integrated models is thus becoming more dependent on disciplined operating models and supporting infrastructure that can handle greater complexity without disrupting the patient experience.
Business solutions are becoming core infrastructure for U.S. practices
Digital business solutions — spanning benefits verification, patient communication and practice analytics — are playing a larger role in how U.S. optometry practices manage administrative complexity and patient engagement. Adoption is being driven by rising participation in managed vision care, including the expansion of large plans such as VSP, as well as staffing constraints that make automation more valuable. In particular, shortages among technicians and administrative staff are accelerating interest in tools that reduce manual effort per patient and enable greater delegation across workflows.
Adoption and spend vary meaningfully by practice size and sophistication. Larger and more complex practices tend to invest more heavily; smaller practices adopt more selectively based on perceived return on effort. Interest in these tools is strongest where workflow pain points are most acute, with growing preference for integrated platforms that reduce system fragmentation and simplify staff training, rather than stand-alone point solutions.
Conclusion
The U.S. optometry market is not being disrupted so much as it is being structurally rewired. Growth is accruing to models that reduce complexity for a fragmented provider base, preserve optometrist trust while enabling digital convenience, and embed deeply into daily clinical and operational workflows. In this market — where trust, reliability and continuity of care remain paramount — platforms that simplify the process rather than bypass the clinician are best positioned to capture long-term value.
For more information, please contact us.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2026 L.E.K. Consulting LLC
L.E.K. Consulting’s latest analysis finds that after a megadeal-driven spike in 2024, North America’s education M&A market is normalizing in 2025 with broader activity across small and mid-sized deals. Meanwhile, long-term investment themes are taking shape for 2026. Growth is fueled by K–12 systems consolidating fragmented edtech tools, continued expansion of school choice programs, and rising demand for vocational and skilled trades education supported by policy and employer needs.
At the same time, AI adoption in education is still early but steadily increasing, suggesting significant future upside. Overall, the sector is shifting from volatility toward more balanced, theme-driven growth opportunities across K-12, higher education and workforce training.
Loading transcript...
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2026 L.E.K. Consulting LLC
Sustaining Breakthrough Science Through Discipline
Key takeaways
In life sciences tools and diagnostics, sustained value creation depends as much on the innovation-led operating model as it does on breakthrough science, given tightly integrated hardware, consumables and software systems.
As companies scale, they must transition from founder-led invention to a repeatable innovation engine anchored in structured market validation and explicit decision discipline.
Building that engine requires seven reinforcing disciplines that connect broad opportunity sourcing and rigorous early screening to evidence-based governance, capital allocation and active portfolio management.
Long-term outperformance demands continuous self-assessment and deliberate evolution of the operating model to maintain focus, adaptability and durable growth.
Innovation in life sciences (LS) tools and diagnostics is difficult because value is created by scaled applications across the triad of hardware, consumables and software. Because performance, reliability and outcomes emerge at the system level, innovation and growth quickly increase coordination demands across the entire value chain. In this context, the innovation-forward operating model becomes a primary determinant of sustained value creation.
Early-growth tools and diagnostics companies frequently stall as they move beyond initial product-market fit. Initial success is often driven by founder-led invention — a breakthrough technology, instrument, assay or data layer. But commercial scaling requires a shift from a founder-led cadence to a repeatable innovation engine that can manage system interdependencies and long capital-intensive paths to return on investment. Without that shift, organizations drift toward fragmented roadmaps and slow launches, where incrementalism crowds out material improvements.
These challenges cannot be solved by importing operating models from adjacent sectors. Biopharma models are asset-centric and optimized for products that are managed relatively independently, while software models assume modularity and asset-light quick iteration. Tools and diagnostics sit between these extremes: They require continuous innovation, but within physically integrated systems where changes propagate across hardware, reagents/kits, software and workflows.
In this environment, operating-model misalignment shows up quickly in cost creep, manufacturability difficulties, customer issues and inconsistent commercial performance.
As platforms scale, R&D intensity typically moderates but remains structurally elevated: mature category leaders such as Illumina still invest approximately 20% of revenue in R&D, and scaling companies even higher (e.g., Oxford Nanopore 44%, 10X Genomics at 37% in 2025) compared to around 23% U.S. public commercial biopharma R&D average or 10%-20% of scaled software companies (e.g., Microsoft, Adobe).
When 25%-50% of revenue is reinvested into R&D, the operating model becomes a first-order determinant of runway, launch cadence and margin trajectory — and must be deliberately designed to sustain system-level innovation while progressively focusing R&D intensity with scale.
In this article, L.E.K. Consulting outlines seven steps to build a scalable, repeatable and commercially successful innovation engine for emerging and growing tools and diagnostics companies (Figure 1).
Figure 1
Summary of seven steps
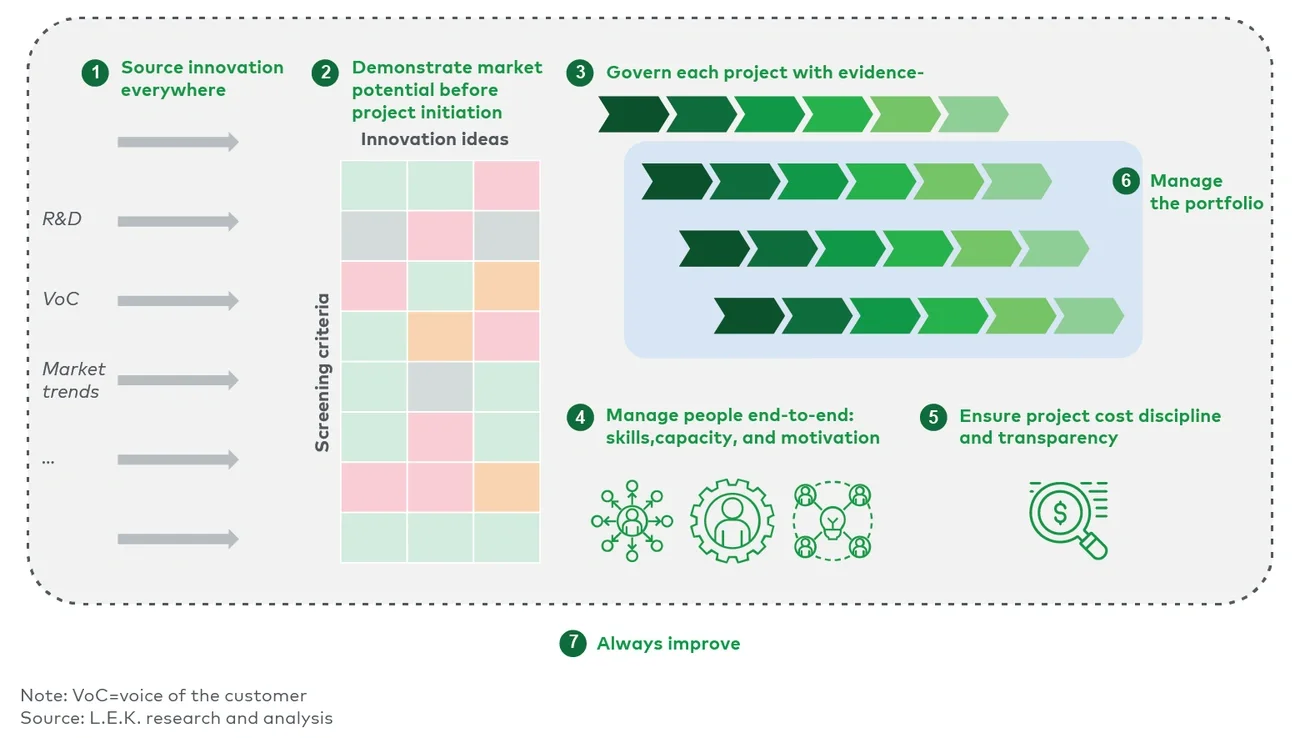
Seven steps to drive innovation-led growth
1. Source innovation everywhere
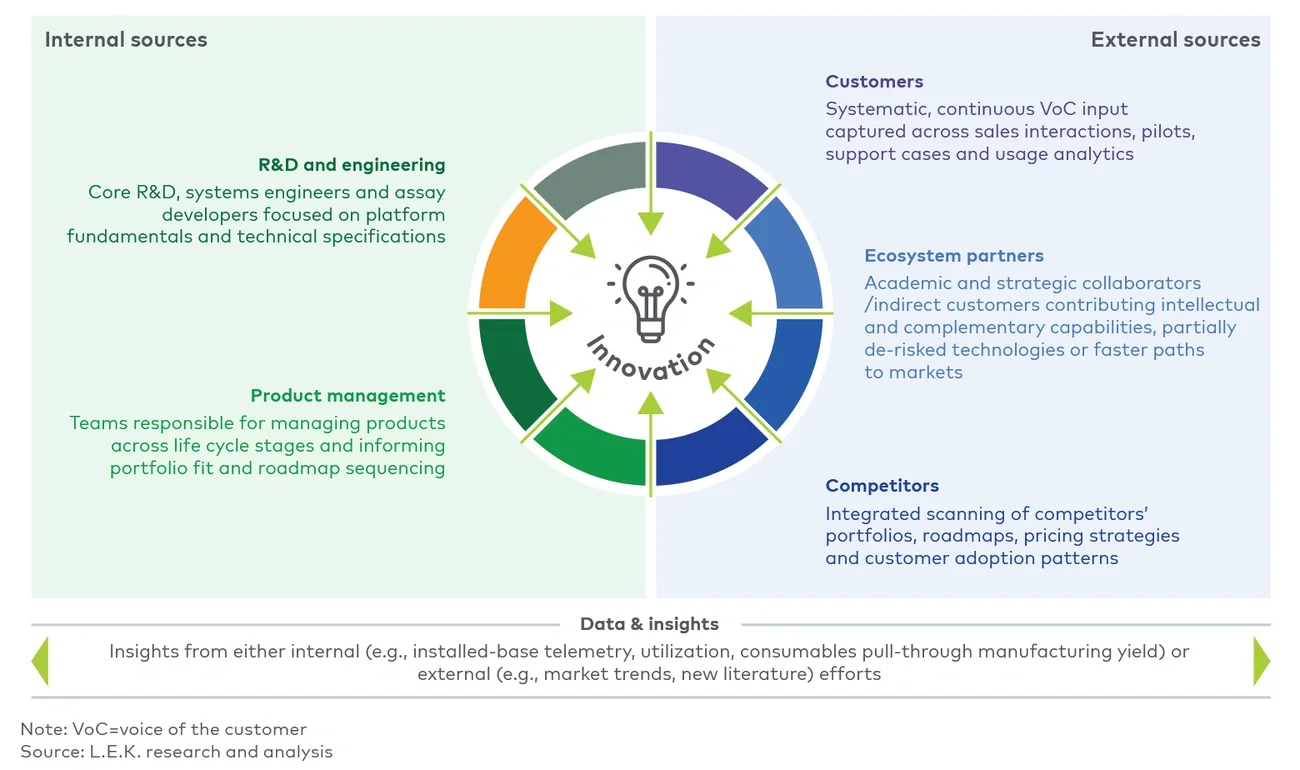
Innovation in tools and diagnostics should be sourced through a wide-aperture system that reflects the multidisciplinary, workflow-driven nature of the business (Figure 2). Breakthrough and incremental opportunities emerge when inputs from external technology trends, customers and end users, customer-facing teams, and internal scientific and engineering expertise are deliberately integrated — not when ideas flow from a single function.
Continuous voice-of-customer (VoC) feedback, captured across external research, sales, applications, service and support interactions, should be treated as a persistent input, grounding innovation in real-world workflows, customer needs and current constraints.
Over-reliance on central R&D can bias portfolios toward technically elegant solutions for problems of limited customer value; under-leveraging field, service and applications teams leaves a critical insight engine untapped. Leading companies therefore create multiple visible pathways for ideas to enter and be evaluated, ensuring innovation is informed by scientific possibility and by structured external research of adoption dynamics, unmet needs and evolving customer constraints.
Figure 2
Overview of internal and external innovation sources
2. Demonstrate market potential before project initiation
Tools and diagnostics organizations push the frontier of what is scientifically and technically feasible. That capability underpins category creation, but it also creates risk: Without concept discipline, teams can pursue technically impressive solutions with weak product-market fit, unclear economics or limited relevance to priority segments. As ideas are triaged into viable concepts, companies should explicitly link the market problem to the target workflow, downstream economic value and required solution architecture.
We recommend an adaptation of the Heilmeier Catechism — originally developed by George Heilmeier during his tenure as the Defense Advanced Research Projects Agency director — as a structured early screen. The catechism should be applied across the entire hardware-consumables-software ecosystem and its role within the broader platform.
LS tools and diagnostics — adapted Heilmeier screen:
- What problem are you solving and for which workflow?
Articulate the objective in plain language, grounded in a specific workflow or use case. - How is this workflow addressed today, and where does it break down and create meaningful pain points?
Describe current solutions and their technical and economic limits — and why those limits matter to buyers. - What is new in the proposed solution, and which component of the triad creates primary value?
Clarify what is differentiated across hardware, consumables and/or software, and which triad component is the primary source of value versus an enabler. - Who cares, and why does it matter economically?
Define target customers and decision-makers, the value proposition and how value is captured (e.g., expansion of installed bases, increased consumable pull-through, adoption for scaled applied markets). - What are the technical, market, commercial and execution risks?
Assess feasibility and performance risk, competitive and demand uncertainty, and operational execution risk (e.g., manufacturability, cost, speed, scalability). - How does this fit within the existing and future workflows and across the triad system?
Assess adoption and disruption risk alongside backward compatibility and future platform alignment. - What will it cost, and how long will it take?
Estimate development effort, ongoing capital and talent requirement, and time to market. - What are the intermediate and final proof points?
Define measurable milestones that demonstrate technical feasibility, customer value and commercial viability.
Applying this adapted catechism early forces teams to confront workflow relevance, system coherence and economic logic before committing significant product development resources. In practice, it helps prevent spec chasing, concept inflation and ideas that are technically sound but commercially fragile.
3. Govern each project with evidence-based stage gates
Pursuing step-change innovation requires organizations to make hard choices. Teams cannot take bold technical or market risks if career progression and organizational norms implicitly reward only success. Companies that consistently outperform on innovation understand opportunity cost and reinforce a culture that creates space by stopping many initiatives early to free capital, talent and leadership attention for the initiatives that truly matter.
Yet many tools and diagnostics organizations struggle to do so, underestimating opportunity cost and allowing unattractive efforts to persist as “zombie” projects that drain scarce resources.
To counter this dynamic, companies must institutionalize clear shifting/stopping rules and recognize teams for early, evidence-based decisions to change course or disengage. Each project should explicitly define its key potential failure points (e.g., technical feasibility, workflow relevance or economic value) and track evidence against these uncertainties throughout development.
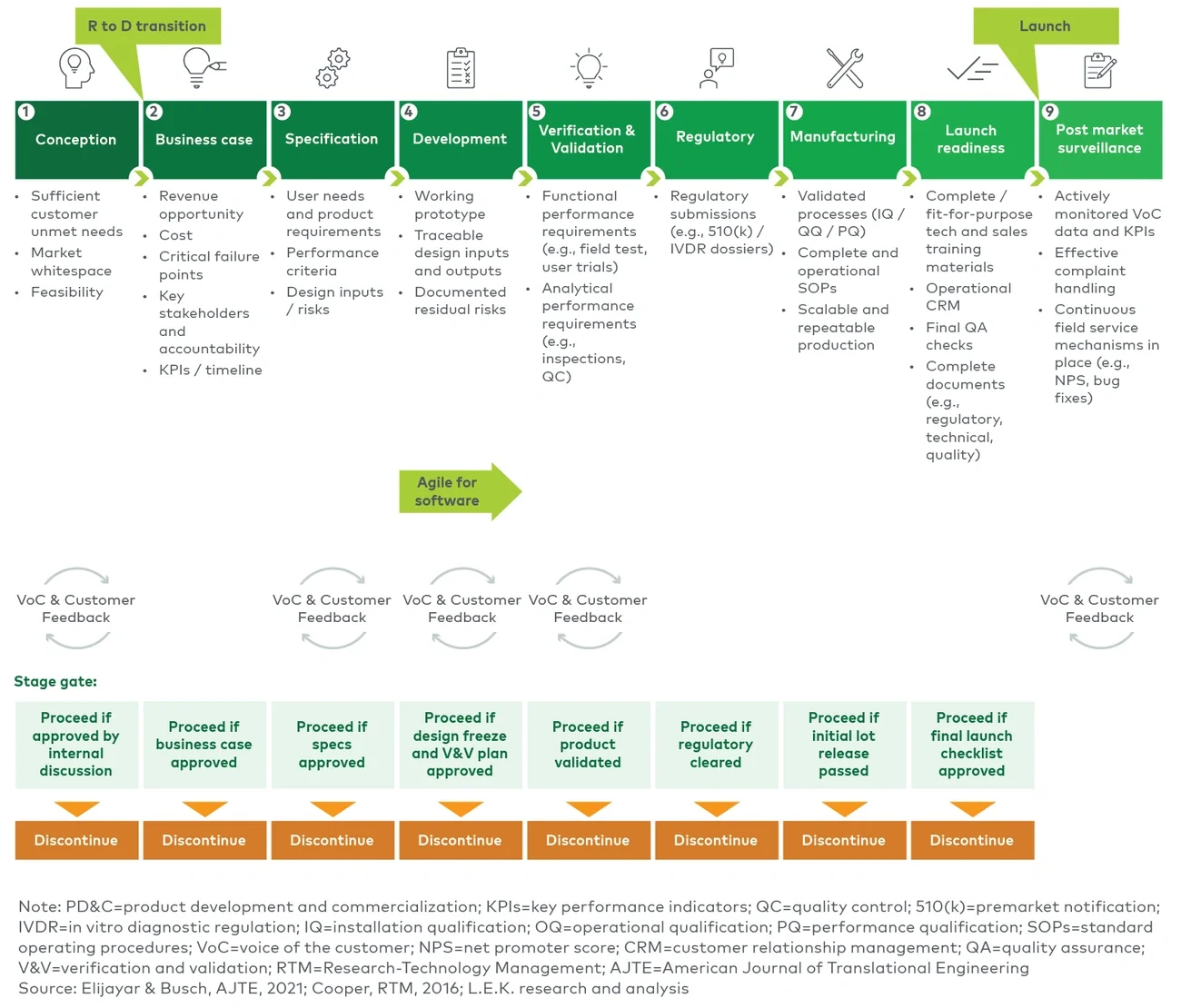
Such discipline requires a robust stage-gated product development and commercialization (PD&C) process, the end-to-end governance mechanism that oversees an idea’s progression from early research through development, scale-up, launch and post-launch life cycle management. At defined transition points, PD&C governance requires explicit, evidence-based decisions to sustain, shift or stop investment based on the status of core assumptions, workflow relevance and economic value.
• Sustain: Core assumptions remain intact and evidence supports continued investment
• Shift: Targeted course correction is required to address specific risks or learnings
• Stop: Feasibility, differentiation or market value has eroded beyond recovery
Embedding this decision logic within PD&C governance normalizes stopping as a sign of rigor — not failure (see Figure 3). Done well, the PD&C process increases speed and quality, sustains motivation across programs and reduces rework as the organization scales.
Figure 3
Example stage-gated PD&C activities
4. Manage people end-to-end: capabilities, capacity and motivation
Tools and diagnostics companies rely on highly specialized talent across systems engineering, assay development, software, manufacturing, quality, field/tech support and regulatory functions. As portfolios scale and multiple programs run in parallel, execution risk increasingly stems from mismatches between capabilities (what skills exist) and capacity (where time and attention are available). Managing people effectively requires addressing both while maintaining culture.
Company leaders should plan and manage capabilities across the full innovation and life cycle continuum — anticipate when specialized expertise (e.g., systems engineering, QA/RA) is needed, invest in upskilling to reduce single points of failure and ensure critical roles are staffed at the right inflection points. In parallel, manage capacity and flow by tracking real availability (not theoretical headcount), surfacing hidden queues and informal workstreams and adjusting ways of working to relieve bottlenecks.
5. Enforce project cost discipline and transparency
The greatest risk is rarely underinvesting in innovation. Instead it is allowing diffuse, low-visibility spending to quietly erode cash runway and crowd out high-value opportunities. Financial discipline requires allocating direct and indirect costs to projects with enough granularity to reflect true spending across the triad. Leaders should be able to track burn by project, understand how spend evolves by stage and distinguish value-creating investment from sustaining or rework-driven cost.
Without this transparency, many organizations drift into the “spreading peanut butter” resourcing model: funding too many initiatives in parallel and diluting capital across them. The result is slow progress, rising burn and high-potential efforts that never receive sustained investment. Avoiding this outcome requires making financial trade-offs explicit so that capital is deliberately concentrated rather than implicitly diluted.
To maintain rigorous discipline, governance should be proportional to financial exposure and uncertainty. Early-stage and exploratory work can draw from a defined “innovation pool” with lighter tracking to enable rapid learning. As initiatives mature, increase in capital intensity or demonstrate product-market traction, they should transition to dedicated cost lines with clearer accountability for development spend, capital requirements and design-to-value trade-offs.
The goal is not bureaucracy but instead tracking early warning signals that preserve runway and keep flexibility to invest where it matters most.
6. Manage the portfolio
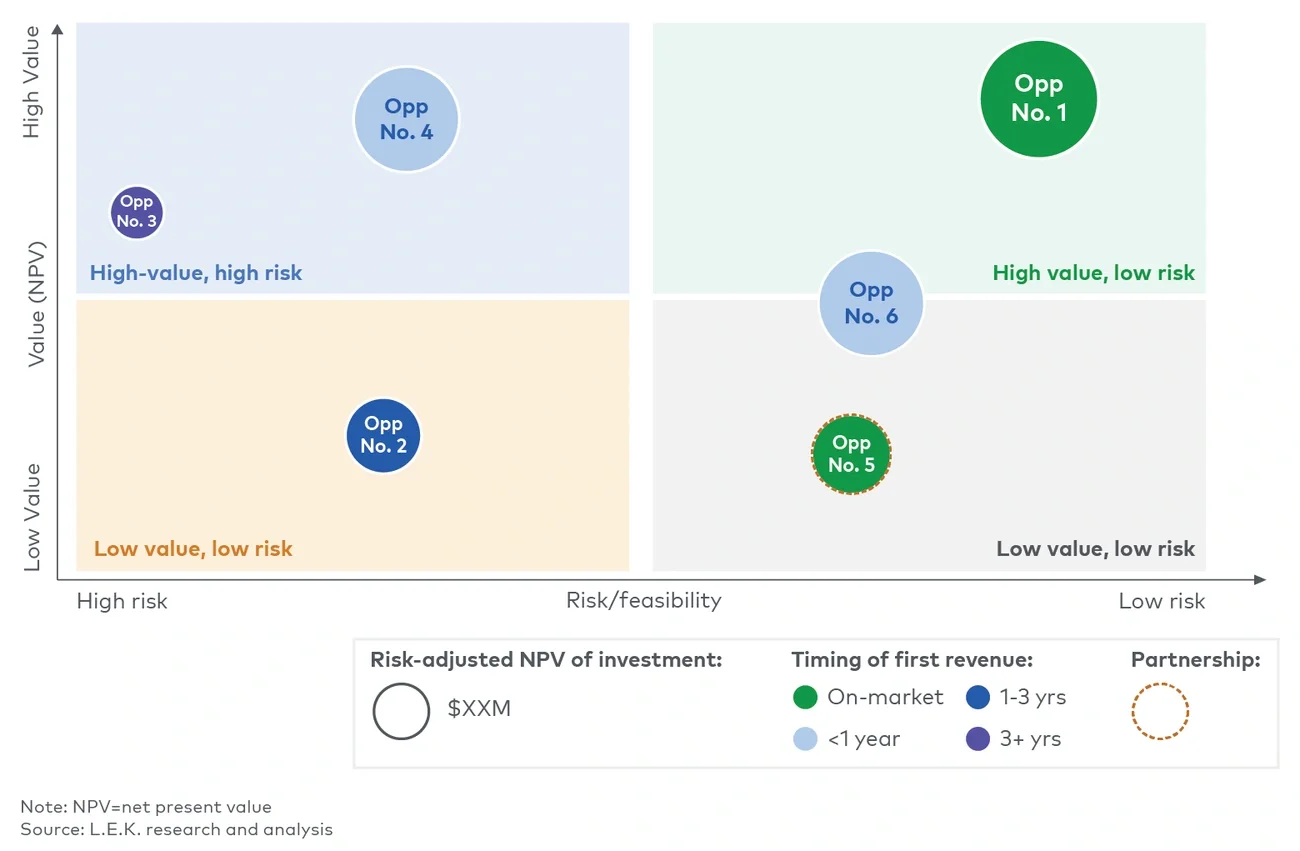
Portfolio complexity compounds quickly as products expand across triad scope, workflows, market segments, geographies, regulatory regimes and more. As a result, portfolio stewardship cannot be episodic, reactive or intuition-driven; it requires proactive evidence-based reassessment of every product and concept across the full life cycle (see Figure 4).
Effective portfolio management enables organizations to continuously optimize value by balancing risk, opportunity and cost. This requires that organizations:
- Actively rationalize the commercialized portfolio as new offerings launch, streamlining overlapping products, variants and use cases to reduce customer confusion, supply-chain complexity and service burden
- Maintain a thoughtfully balanced pipeline, spanning high-risk/high-reward innovations, moderate risk enhancements that extend or reposition current product, and lower-risk life cycle updates that sustain and refresh the product
- Align triad life cycle governance, with explicit plans for backward compatibility, forward integration into future platforms and disciplined product and feature retirement
- Drive reuse and platforming across shared elements of the offering to reduce complexity, accelerate development and lower sustaining costs
When applied consistently, portfolio management becomes a value-optimization engine, not just a project list review process.
Figure 4
Example portfolio prioritization matrix
7. Always improve
Leading tools and diagnostics companies do not stand still. They cultivate a culture of continuous challenge by systematically questioning processes, assumptions and ways of working across the organization. The objective is not change for its own sake, but for sustained value creation — better products, higher reliability, greater operational effectiveness and shorter cycle times as complexity and scale accumulate.
Digital tools and artificial intelligence (AI) are increasingly central to this effort, but they should be viewed as enablers of continuous improvement, not ends in themselves. The opportunity is not to layer technology onto existing workflows or pursue an “AI first” posture divorced from customer value. Rather, advances in automation, data and AI should prompt organizations to rethink workflows, decision rights, skill requirements and performance metrics to improve outcomes.
Technologies and partners should be adopted deliberately, only where they deliver clear, measurable gains; accumulating tools or partnerships for novelty or perceived sophistication often increases complexity without commensurate benefit while diverting resources away from critical projects.
Based on the steps outlined, we encourage leaders to consider a small set of self-assessment questions to diagnose their operating model — an approach we often use with leadership teams to translate innovation ambition into scalable, system-level execution:
- Innovation sourcing: Are we exploring enough sources of innovation?
- Market relevance: Have we conducted a structured assessment of the market opportunity and how our new programs fit?
- Stage gates: Are our stage gates designed to force explicit stop, pivot, or scale decisions — or do initiatives advance by default?
- Talent and capacity management: Do we understand and actively manage our people?
- Cost transparency and focus: Can leaders see the true cost of each initiative and actively allocate resources on the few that matter most?
- Portfolio discipline: Have we proactively reviewed our portfolio against defined prioritization criteria?
- Continuous improvement: Are we getting faster, more reliable and less wasteful over time?
If these questions highlight opportunities to strengthen your innovation operating model, please reach out to L.E.K. to further assess your current state and define a more disciplined, scalable path forward.
Note: We thank our external readers Jenny Mackey, Adam Siebert and Thomas Bell for their input and support.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2026 L.E.K. Consulting LLC
Royalty Financing Comes of Age: Why This Once-Niche Source of Capital Is Gaining Momentum
Key takeaways
Beyond major pharmaceutical companies, a broad range of organizations possess valuable, often underrecognized, royalty positions.
However, royalty-related financing remains an often misunderstood source of capital in the biopharmaceutical industry.
As the market has matured, the range of royalty deal structures and execution pathways has expanded significantly.
Over the past 24 months, market dynamics have reshaped how biopharma companies think about their capital formation strategies.
Executive summary
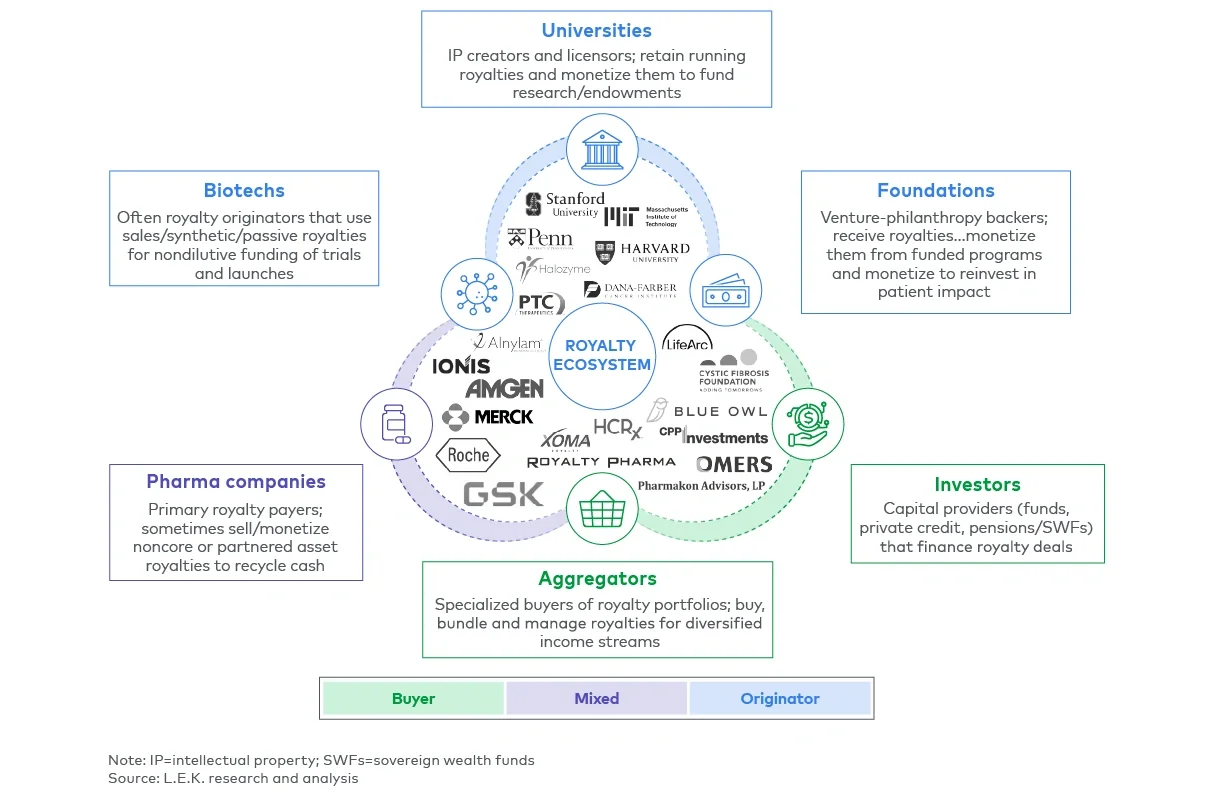
Royalty interests are more widely held than many executives realize. Beyond major pharmaceutical companies, a broad range of organizations possess valuable, often underrecognized, royalty positions. Emerging biotechs may hold royalties from outlicensed products or codevelopment arrangements. Universities and research hospitals often retain the rights to discoveries licensed to commercial partners.
Foundations such as the Cystic Fibrosis Foundation have monetized royalty streams to reinvest in research and patient programs. In addition, aggregators like XOMA and DRI actively acquire and manage diversified portfolios of royalty assets.
However, royalty-related financing remains an often misunderstood source of capital in the biopharmaceutical industry. Once viewed as an expensive last-resort option, nondilutive royalty funding has matured into a strategically flexible instrument that should be considered alongside more traditional equity and debt options.
As the market has matured, the range of royalty deal structures and execution pathways has expanded significantly. For management teams, understanding the pros and cons of royalty financing as well as how the varied range of royalty-focused structures best align with different strategic objectives has never been more important (see Figure 1).
L.E.K. Consulting has advised on nearly 100 royalty transactions over the past two decades. Our work has helped clients determine the fair value of their royalty assets, evaluate optimal structuring options and engage the right partners and processes to unlock value. In this edition of Executive Insights, we will share key learnings from our experience as well as considerations that should be top of mind for executives considering their capital formation options.
Figure 1
The royalty ecosystem
Why royalty financing is gaining momentum
Over the past 24 months, market dynamics have reshaped how biopharma companies think about their capital formation strategies. High interest rates, relatively closed-off equity markets and a more constrained credit environment have driven up the cost of traditional funding sources. At the same time, return expectations among royalty investors have remained relatively stable, typically in the high single to low double digits, narrowing the cost gap between royalty capital and conventional debt or equity.
In addition, while the supply of traditional capital has tightened, institutional investors such as pension funds, sovereign wealth funds and private credit platforms continue to allocate capital to royalties because these instruments offer steady uncorrelated cash flows that diversify their portfolios. For companies, royalty monetization continues to offer liquidity without dilution as well as freedom from the operational covenants associated with many forms of debt. As a result, what was once viewed as niche or expensive capital has become a more strategic component of the biopharma funding mix.
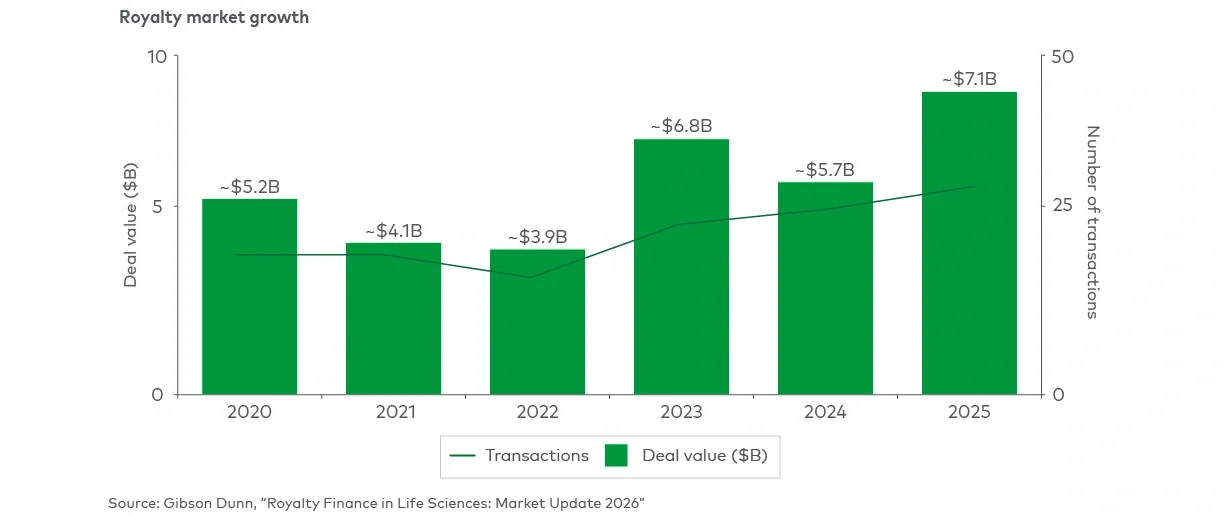
The growth of the royalty financing market underscores its evolution into a mature and competitive asset class. Global royalty deal value has risen from $5.2 billion in 2020 to $7.1 billion in 2025. Royalty financing has been relatively stable and rate-resilient. Greater royalty-related deal volume has been driven not only by the specialized royalty funds that have long dominated the space, but also by new entrants such as private credit and infrastructure investors whose participation has deepened liquidity and spurred innovation in deal structuring.
In addition to higher deal volume, the average royalty deal value has also risen. Some of this increase in average value can be attributed to multiasset portfolios, but it’s also being driven by greater competition. Syndicated transactions, which are often the result of competitive processes, now account for approximately one-third of total royalty market activity.
Collectively, these shifts point to a maturing ecosystem characterized by larger transactions; faster, more-sophisticated processes; and greater competition, all of which favor well-prepared buyers and sellers (see Figure 2).
Figure 2
Total deal value reached $7B by 2025, driven by deeper institutional participation
The expanding range of deal structures
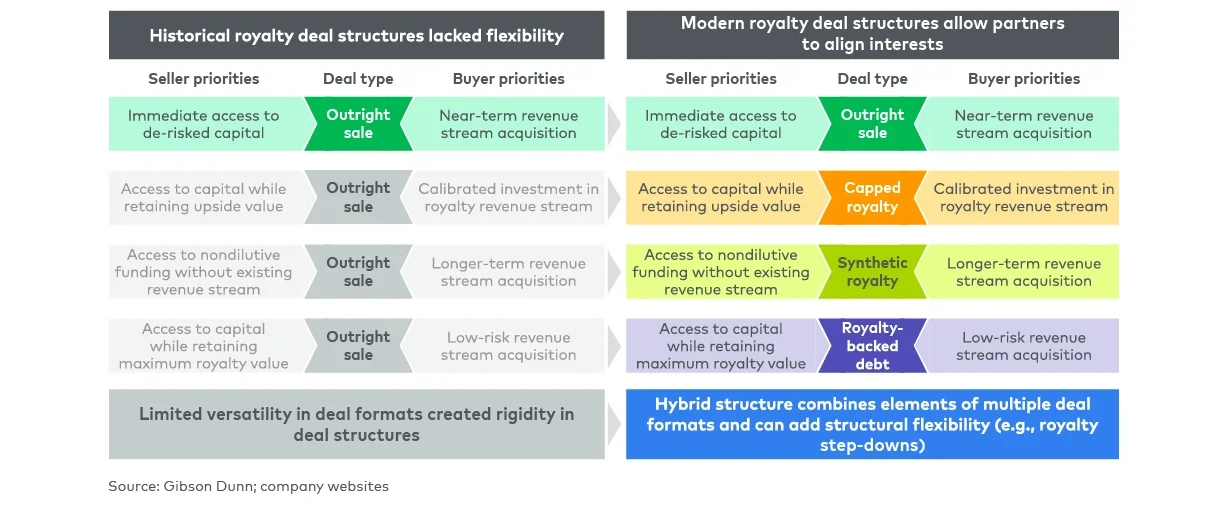
The modern royalty market offers a continuum of structures, each designed to balance risk transfer, upside potential and execution speed in different ways. In an outright sale, the seller transfers an existing royalty stream to the investor in exchange for a lump-sum payment, an approach best suited to mature in-market assets where simplicity and certainty are paramount. Capped royalty structures, in which investor returns cease once a predetermined multiple has been achieved, are particularly effective for assets with potential for life cycle expansion or additional label indications.
Synthetic royalties allow manufacturers to create royalty-like payment streams prior to FDA approval, enabling them to raise nondilutive capital for late-stage development or launch activities. Royalty-backed debt uses anticipated cash flows as collateral, providing flexibility for commercial-stage assets with predictable sales. Finally, hybrid structures combine elements of multiple formats, such as pairing a term loan with a capped royalty sale, which can broaden the number and type of investors that may be interested in participating.
The optimal structure depends on the product’s risk-return profile, the company’s financial objectives and the current market conditions (see Figure 3).
Figure 3
The spectrum of royalty financing structures
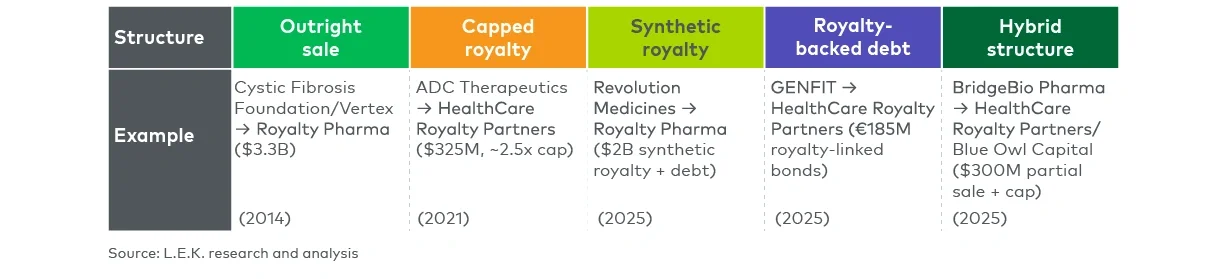
Our experience shows that tailoring structure can often be a way to bridge the divide between two parties’ views of the risk-adjusted value of a particular royalty stream, balance the varying risk tolerance of different investors and increase the appeal of a deal to a broader set of investors (see Figure 4).
Figure 4
Royalty deal transaction examples
Aligning objectives and process design
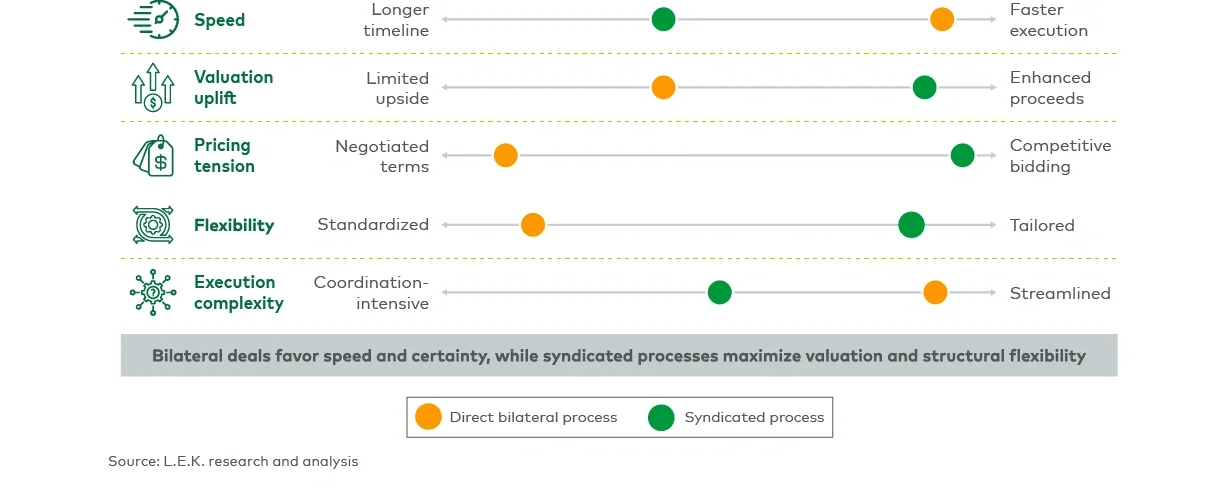
While deal structure defines the economics of a royalty transaction for each party, the process used to execute it often determines its success. Companies can pursue either a direct bilateral transaction or a syndicated process. In a direct deal, the seller engages with a single established royalty fund, such as Royalty Pharma or HealthCare Royalty Partners. These transactions offer speed, confidentiality and the signaling benefit of partnering with a well-known counterparty, but they may limit pricing tension and structural creativity.
Syndicated processes, typically led by an advisor or investment bank, invite multiple royalty funds and private credit investors to participate. This competitive dynamic can lift valuations and allows for more-bespoke multitranche structures, albeit with longer timelines and greater coordination requirements.
In practice, the optimal path depends on a company’s objectives. Organizations seeking speed and certainty may prefer a bilateral deal, whereas those focused on maximizing proceeds or flexibility can benefit from competitive tension. Regardless of approach, establishing a rigorous, data-driven valuation foundation is critical to ensure fairness and confidence in negotiations.
While either approach can be valuable under a different circumstance, L.E.K. has found that introducing even a modest amount of competition into a formal process can yield higher effective proceeds compared with direct deals (see Figure 5).
Figure 5
Royalty deal process trade-offs
What this means for biopharma leaders
For today’s biopharma chief financial officers and business development executives, royalty financing is no longer a tactical fallback; it is a strategic capital formation lever that should be considered. Success begins with understanding the intrinsic value of one’s asset before entering the market. Companies must choose a structure that reflects product risk, life cycle stage and funding need, and they should view process design itself as a negotiating lever. Even a light competitive check can materially shift terms.
Timing also matters: Monetizing when visibility is high enough to support strong valuation, rather than while under duress, allows companies to optimize both proceeds and flexibility. Finally, engaging advisors who combine deep therapeutic understanding with transaction experience ensures that deals balance investor interest with long-term corporate objectives.
The L.E.K. perspective
We bring more than two decades of experience advising across the full spectrum of royalty transactions, from traditional single-asset sales to complex, multi-investor portfolio financings. Our teams combine deep clinical and commercial expertise with real-time insight into investor preferences and deal structuring trends.
For any company holding or able to create a royalty interest, now is the time to reexamine its strategic value. For more information about how to understand, optimize and unlock that value, please feel free to contact us.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2026 L.E.K. Consulting LLC
Education Investment in Transition: 2025 M&A Trends and Opportunities in 2026
L.E.K. Consulting’s latest analysis examines how Education M&A activity in North America is evolving in 2025 and what structural themes are shaping investment priorities for 2026. Drawing on transaction data and sector research, the report assesses momentum across K-12, higher education, professional upskilling and corporate training.
Following COVID-19-driven investment peaks in 2021-22, the volume of Education investments has grown year-over-year since 2023’s low. While 2025 M&A value was lower than 2024, this was driven was by a handful of 2024 mega deals – in fact, disclosed investment value for deals under $1B increased in 2025, reflecting both continued investor confidence in the sector and strategic acquisition activity.
As we look toward 2026, a number of key themes emerge:
- K-12 leaders are consolidating their technology ecosystems in response to rapid tool proliferation. The average number of edtech platforms used per district grew at roughly 23% CAGR in recent years, prompting a shift toward more integrated and interoperable systems, which creates both opportunity and risk depending on where you sit in the ecosystem.
- School choice expansion continues, with 33 state-level programs and OBBB creating tailwinds for private education and the providers that service that ecosystem Skilled trades are an area of investor focus given workforce demand, perceived AI defensibility and increasing focus on alignment of education to employment.
- Professional upskilling and reskilling take center stage as employers and professionals alike respond to changing workforce needs in the age of AI. However, certain business models are better positioned to succeed than others.
- AI adoption across the education ecosystem is advancing steadily but remains at an early stage of maturity. Education represents about 2% of global AI-centric systems spending today, underscoring both measured uptake and meaningful long-term headroom
Download the full analysis to explore the detailed trends and understand how investors and operators are positioning for growth in 2026.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2026 L.E.K. Consulting LLC
Modular Medicines: Combinatorial Modularity as a Strategic Source of Innovation
Key takeaways
Biopharma is moving from molecule-by-molecule “discovery” iteration/optimization toward modular “design-and-assemble” medicines.
Modular medicines recombine validated payloads, functional controllers and targeting/delivery layers.
Patent-cliff urgency, crowded biology and rising chemistry, manufacturing and controls (CMC) complexity are pushing sponsors toward modularity because it can cut stacked technical risk, shorten design-make-test cycles and reuse platform CMC/analytics precedent to accelerate timelines and reduce iteration cost.
There are four key implications: (1) competitive advantage shifts downstream into CMC, analytics and manufacturing responsiveness; (2) “platformized” manufacturing and comparability playbooks become key partner differentiators; (3) precision therapies become more economically viable as platforms enable smaller indications and even “N-of-1” scale-out; and (4) supply chains and capacity planning shift from scale-up economics to cost-per-configuration, rapid changeover and rapid release.
Biopharma has introduced a new design paradigm where success is less about highly iterative “discovery” and more about designing and assembling validated components. Rather than focusing on entirely novel targets or uniquely developed molecules for each design-make-test cycle, innovators are recombining proven building blocks to accelerate development, reduce biological risk and improve capital efficiency. The growth of the building-block tool kit, in turn, becomes an additional source of novelty and innovation for the modular medicines approach.
A vivid proof point was the rapid deployment of a personalized gene-editing therapy at Children’s Hospital of Philadelphia in spring 2025 (Baby KJ). While the scientific details of each of these cases will be unique, the operating model enabling these breakthroughs will be the highly modular therapeutics design: Leverage validated components, adjust to patient-specific circumstances, move fast as a disciplined quality and leverage the Food and Drug Administration’s emerging regulatory pathways.
This edition of L.E.K. Consulting’s Executive Insights builds on our prior work on advanced modalities to explain why “design modularity” is emerging, where its impact is most pronounced and what it means for manufacturing partners and toolmakers.
Modular medicines defined
Unlike traditional drugs — constructed as individually optimized unique molecules resulting from intensive iteration and screening — modular medicines separate core functions into components that work together as an integrated system. Those components can be reused and recombined to design (rather than to discover de novo) new therapeutics, and the components themselves can improve over time, expanding the scope of design space.
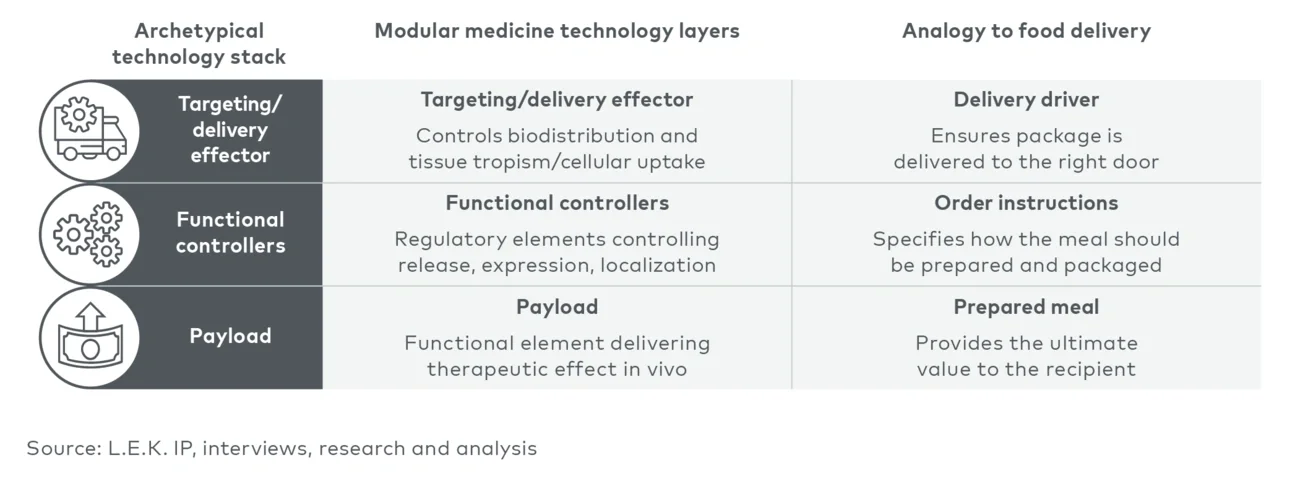
A practical segmentation is three technology layers:
- Payloads: the therapeutic “work” (e.g., genetic payloads, high-potency active pharmaceutical ingredients) that drives the biological effect
- Functional controllers: regulatory/tuning elements (e.g., untranslated regions, promoters, linkers, “kill switches” that turn off expression and mediate safety) that shape potency, pharmacokinetics, localization or expression
- Targeting/delivery effectors: the mechanism (e.g., adeno-associated virus (AAV) capsids, lipid nanoparticles (LNPs), monoclonal antibodies) that governs biodistribution and uptake
Food delivery is a useful analogy: The payload is the meal itself, the controller is the specific order instructions (“leave on the doorstep”) and the targeting/delivery effector is the driver (see Figure 1).
Figure 1
Modular medicine technology stack, by analogy to food delivery service
Why modularity and why now?
Three demand-side pressures are driving biopharma toward modular design, while a growing technology tool kit and supportive regulatory stance are making it more feasible.
- Addressing potential biopharma revenue gaps
The upcoming patent cliff increases urgency to refresh pipelines with assets that can be developed efficiently. Investors and acquirers have become more risk aware, often gravitating toward later-stage or “de-risked” programs. Modularity supports a middle ground: generating more shots on goal while presenting fewer unvalidated novel components within any single program. - A growing need for differentiation against a crowded pipeline
When competing programs address the same biological targets, product differentiation comes from delivery, tissue specificity, controllability and durability. Modular design enables sponsors to pair a validated target or payload with a novel targeting/delivery effector or controller element — potentially delivering a differentiated profile by increasing precision, safety and/or efficacy without taking on the technical risk of an entirely novel drug platform. - Ongoing mandates to increase R&D efficiency
Advanced biologic modalities — including complex analytics, long lead times, constrained capacity and inflexible infrastructure — raise the CMC burden. Modularity can mitigate this by enabling comparability strategies, reuse of analytical methods and select preclinical data and process precedents that shorten the path to investigational new drugs (INDs) and reduce preclinical costs.
What is enabling the shift toward modularity?
Growing toolbox of validated components for modular designs
The library of components is expanding (antibody scaffolds, LNPs, AAV capsids, cell therapy backbones, linkers, promoters). As validation accumulates, sponsors can increasingly mix and match with higher development throughput by leveraging precedent, similar to what is done with excipients and other highly validated components of drug formulation.
Increasing traction of artificial intelligence (AI)-enabled design
AI-enabled platforms are emerging as powerful design engines — optimizing sequences (including guide ribonucleic acids (RNAs)), predicting structure/function relationships for proteins and creating next-generation delivery vehicles (capsids, lipid compositions). These workflows compress the design-make-test loop and shift more work upstream into in silico exploration in the dry lab.
Supportive regulatory stance for ‘platforms’
Regulators are evolving alongside the shift. Emerging initiatives explicitly recognize platform reuse, creating pathways to reuse elements of CMC and preclinical learnings across related programs when anchored to a well-characterized platform and a strong comparability strategy.
Real-world proof points for personalized genetic medicine
Personalized genetic medicines are also demonstrating a scale-out operating model where elements of a platform are standardized (e.g., delivery system, unit operations, analytics, quality controls), and the variable layer can be changed quickly and predictably. Programs like Baby KJ (and earlier exemplars such as the personalized antisense oligonucleotide (ASO) drug Milasen) show that individualized therapeutic sequences can leverage validated process know-how and testing frameworks to reach patients on dramatically compressed timelines.
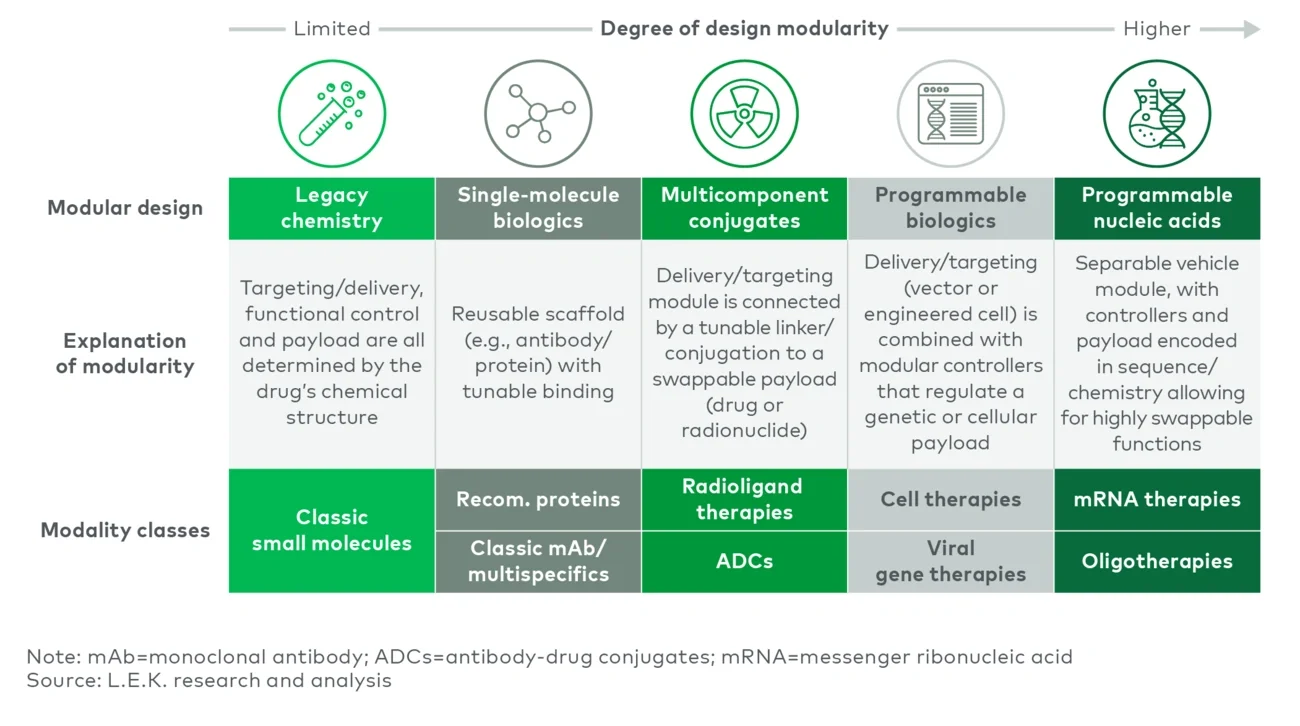
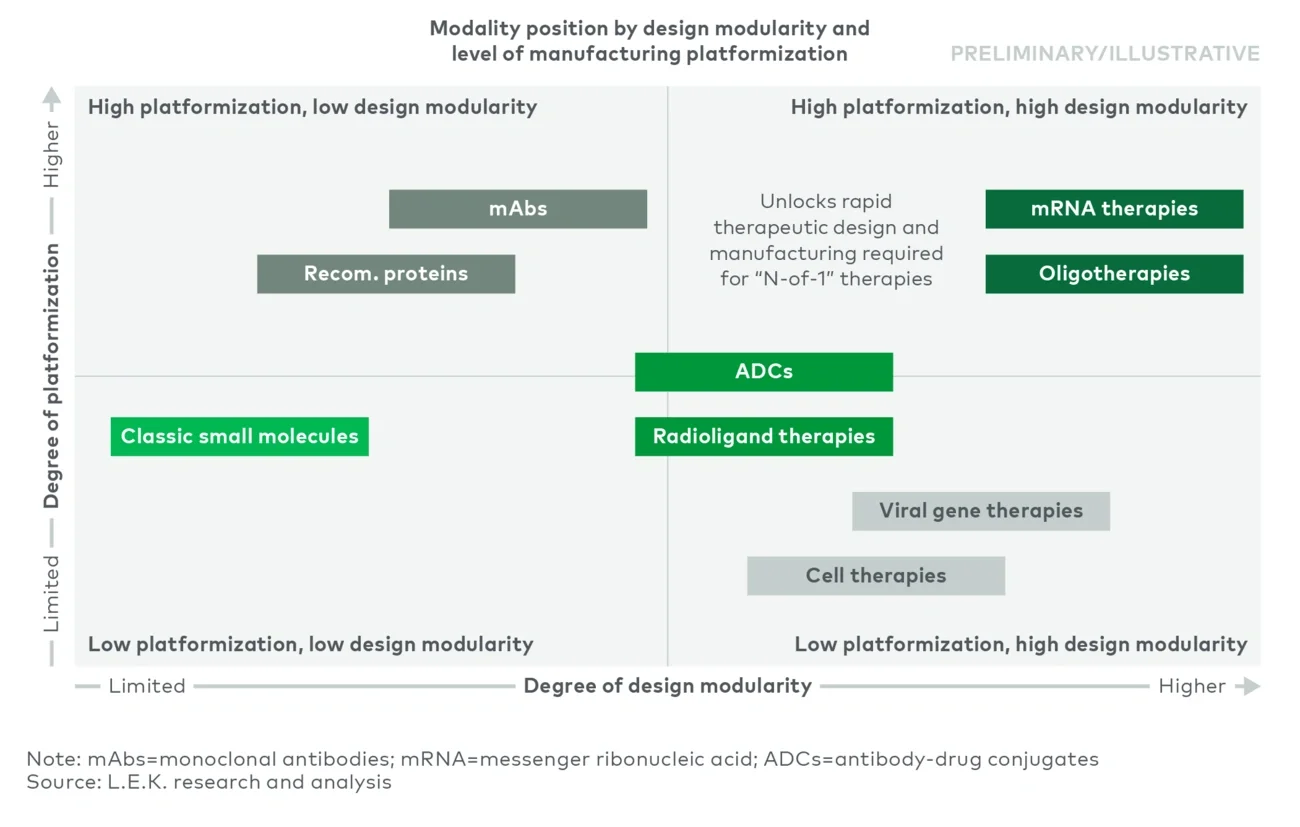
Modularity is a spectrum (and the industry is moving along it)
Design modularity is not binary; it sits on a spectrum (see Figure 2). Legacy small molecules tend to be the least modular because each target requires de novo hit identification and an iterative structure-activity relationship. Single-molecule biologics have tunable domains but remain individually optimized. Multicomponent conjugates (e.g., antibody-drug conjugates (ADCs)) combine known parts to create new functionality.
Programmable biologics (viral vectors, engineered cells) use a chassis as the delivery/targeting layer for swappable payloads and control parts. At the end of the spectrum, with the highest design modularity, are programmable nucleic pair sequence-defined payloads with standardized delivery layers (e.g., LNPs, or bioconjugates such as GalNAc).
Figure 2
Design modularity spectrum
Real-world examples: Modular logic across modalities and historical trends
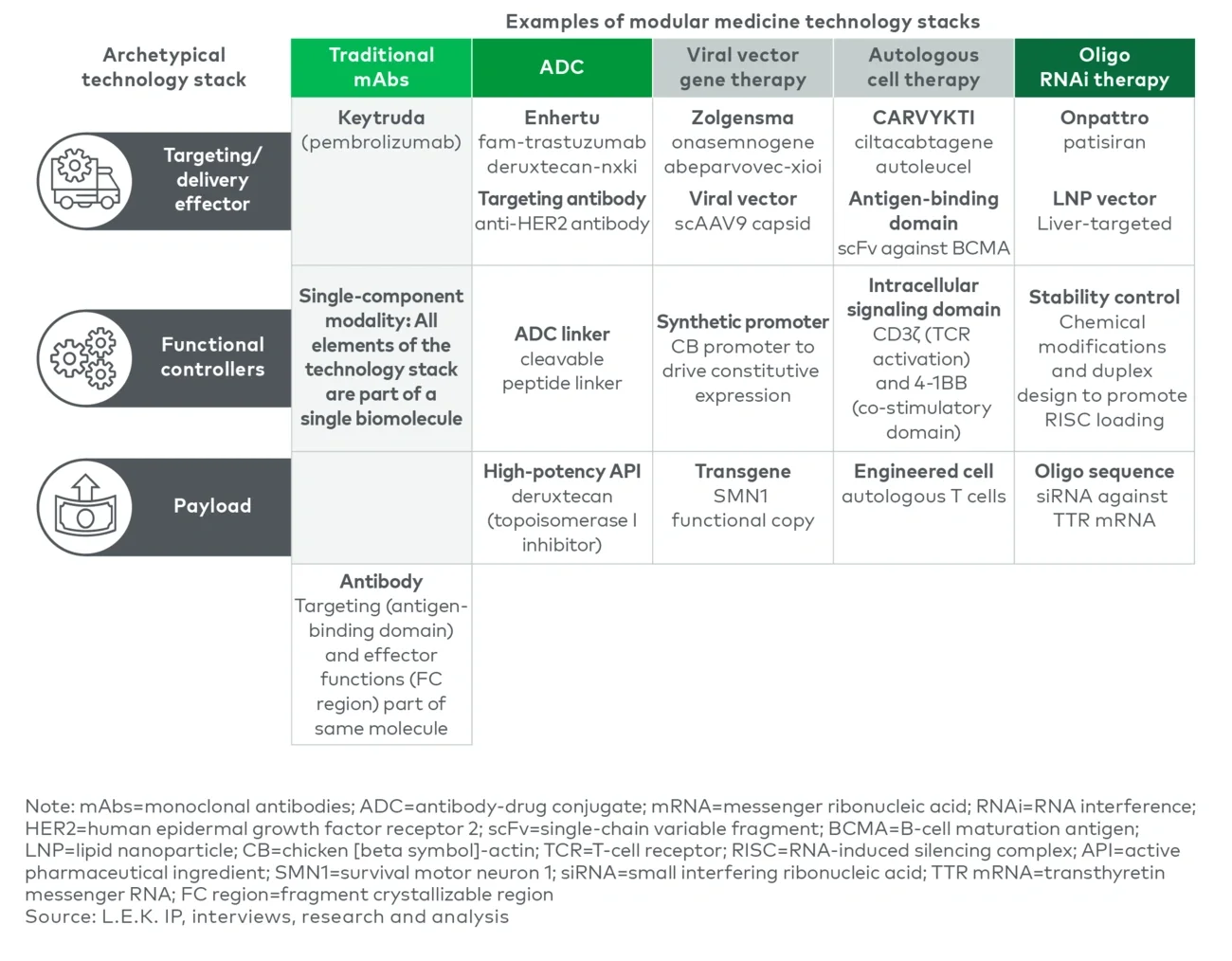
Several approved therapies illustrate modularity as a common innovation pattern (see Figure 3):
- Enhertu (ADC): combines a validated human epidermal growth factor receptor 2 (HER2) antibody scaffold with a linker-toxin system, expanding segmentation to HER2-low
- Zolgensma (AAV gene therapy): pairs a known capsid with a promoter/payload system to treat spinal muscular atrophy
- Carvykti (autologous cell therapy): uses tuned binder and signaling modules on an established chimeric antigen receptor backbone
- Onpattro (small interfering ribonucleic acid (siRNA)/LNP): pairs a sequence-defined payload with a prevalidated delivery approach, de-risking delivery and immunogenicity
Figure 3
Examples of modularity tech stack in approved drugs
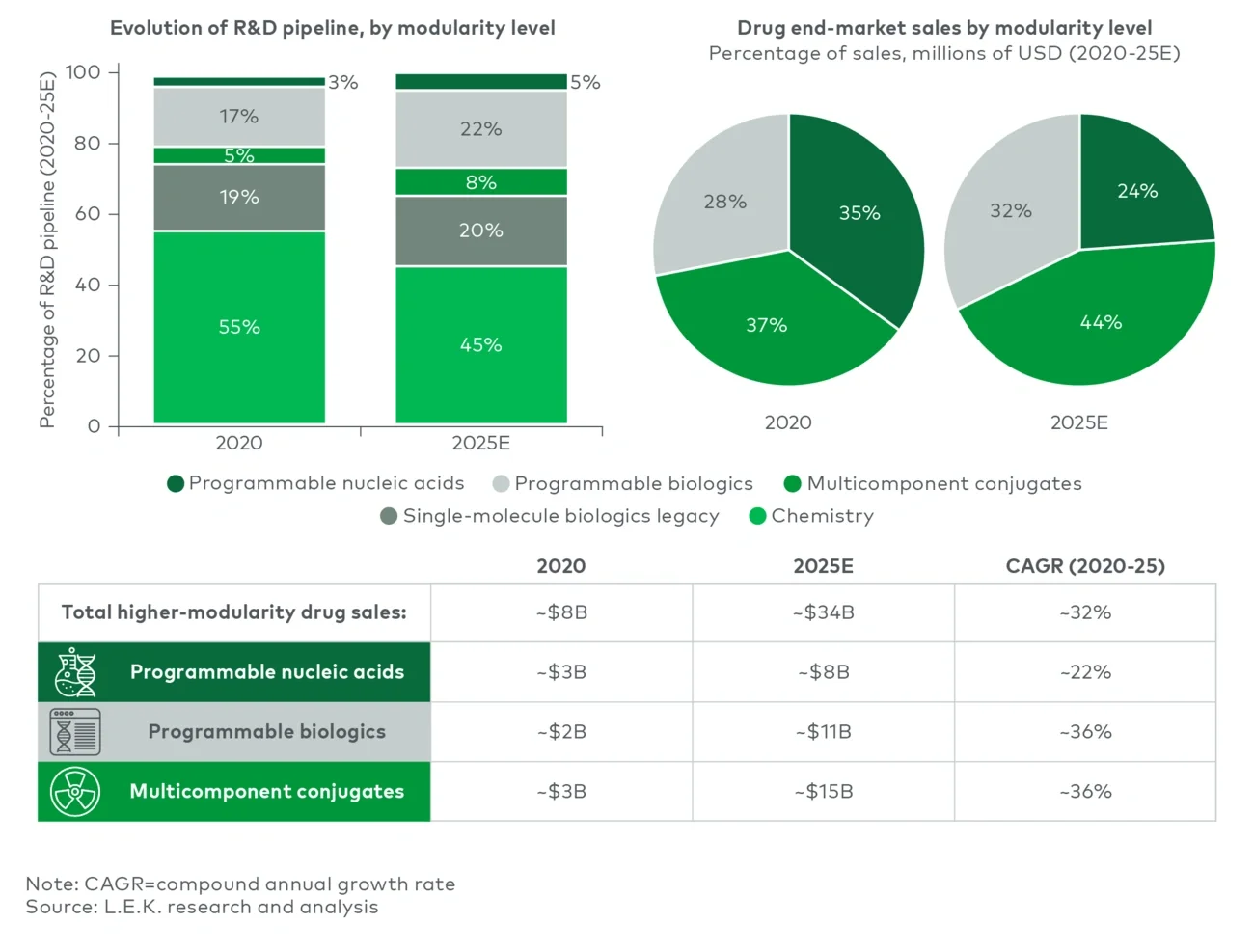
Both the R&D pipeline and drug sales end markets are evolving toward higher levels of design modularity. Increasing along the modularity spectrum yields practical advantages: faster design-make-test cycles, greater leverage of prior CMC and preclinical precedent and (in some cases) access to regulatory pathways that recognize platform reuse.
Historically, the R&D pipeline has shifted from 25% more modular modalities in 2020 to 35% more modular modalities by 2025. Similarly, the end market for these more-modular drug classes has grown dramatically, from approximately $8 billion in 2020 to around $34 billion estimated for 2025, a five-year growth rate of more than 30% (see Figure 4).
Figure 4
Historical trends in pipeline and drug sales, by level of drug design modularity
Combining design modularity with manufacturing 'platformization' to unlock scale
The value of design modularity can be force multiplied when combined with high manufacturing platformization - when a codified set of raw materials and process inputs plugs into an established manufacturing workflow and quality system so each new program is a controlled modification of a base "recipe," not a bespoke process for every product.
In practice, manufacturing platformization means common unit operations, qualified raw materials, shared analytical methods, digital enablers (chain of identity, chain of custody) and comparability playbooks that are widely shared across programs. The result is a smaller CMC lift, faster changeover time and reduced regulatory review friction.
When drugs with high design modularity plug into a workflow with high manufacturing platformization, it becomes possible to successfully address very small indications, as evidenced by successful "N-of-1" projects for ASO- and RNA-based gene editing therapies (see Figure 5). Conversely, low manufacturing platformization can require intensive CMC optimization ahead of launch, as evidenced by the "Process is the product" mantra for early cell and viral gene therapies, as well as the supply challenges that have characterized several early cell and gene launches (e.g., Kymriah, Carvykti, Casgevy).
Figure 5
Matrix of design modularity and manufacturing platformization
Implications for life sciences tools suppliers and contract development and manufacturing organizations (CDMOs)
- Opportunities for innovation and risk move downstream into CMC and supply chain
As drug design becomes more configurable, competitive advantage shifts toward executing fast, high-quality configuration cycles. That elevates CMC design, analytical strategy and manufacturing agility; CDMOs may increasingly help define manufacturable configurations, not just produce batches. - Platform manufacturing becomes a commercial differentiator
Sponsors will increasingly ask how easily new programs can plug into a partner’s validated platform. CDMOs with reusable process/analytics templates, comparability precedent and strong digital traceability can reduce sponsor burden and accelerate timelines. - Precision therapies expand addressable micromarkets
Faster design-to-IND and permission space to reuse platform data support continued indication fragmentation and subsegmentation, unlocking smaller populations that historically would not have been commercially viable under bespoke development models. - Scale-out economics challenge scale-up mindsets and reshape supply chains
Programmable modular therapies imply smaller batches and more-frequent changeovers. Cost advantage shifts from economies of scale to economies of repetition: Suite utilization, parallel closed processing and rapid release become central, while supply chains must support smaller quantities with tighter delivery tolerances and variable demand.
Taken together, modular medicines will reward partners that treat manufacturing and analytics as configurable platforms rather than one-off projects. Tools companies and CDMOs with flexible capacity, standardized component libraries and data-rich quality systems will be best positioned to grow alongside the next generation of modular medicines.
For more information, please contact us.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2026 L.E.K. Consulting LLC


















































