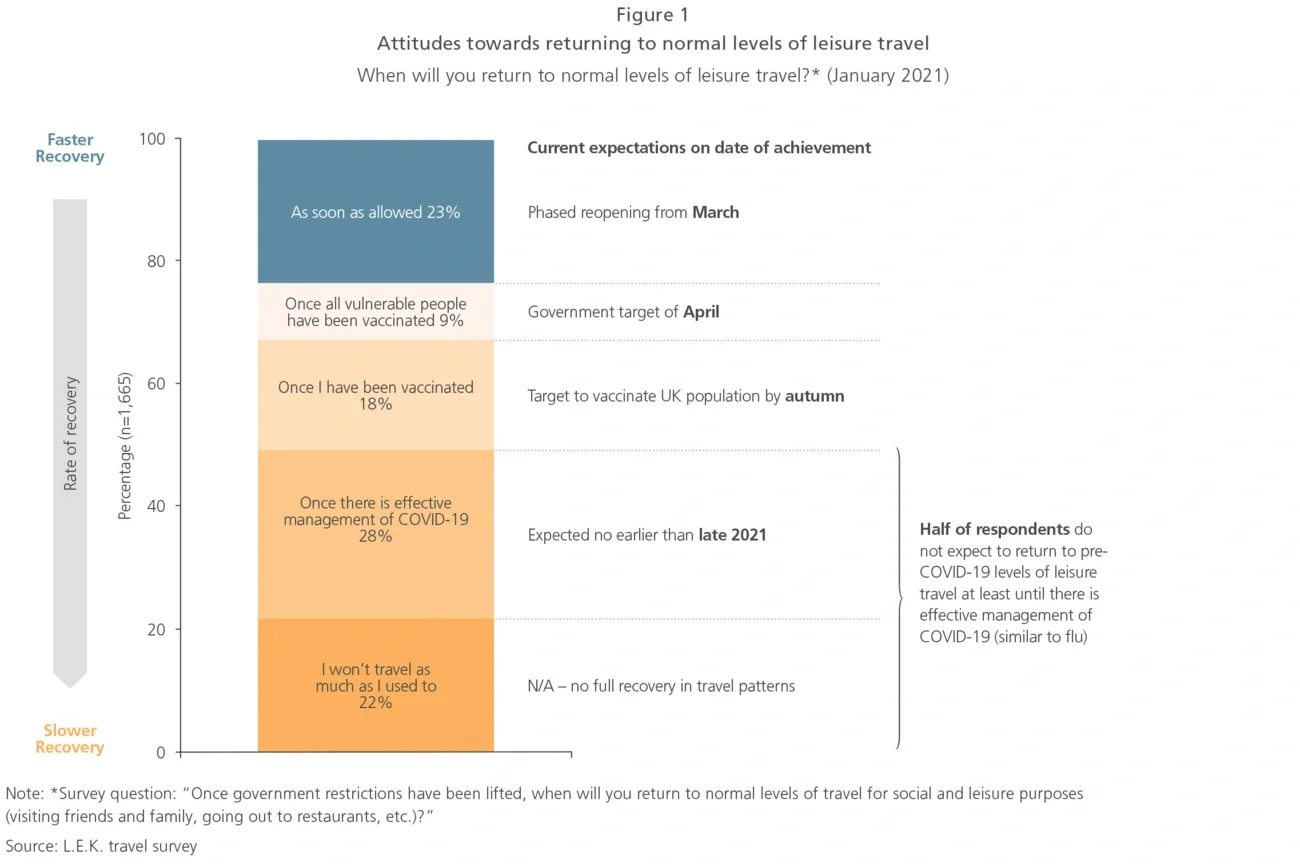
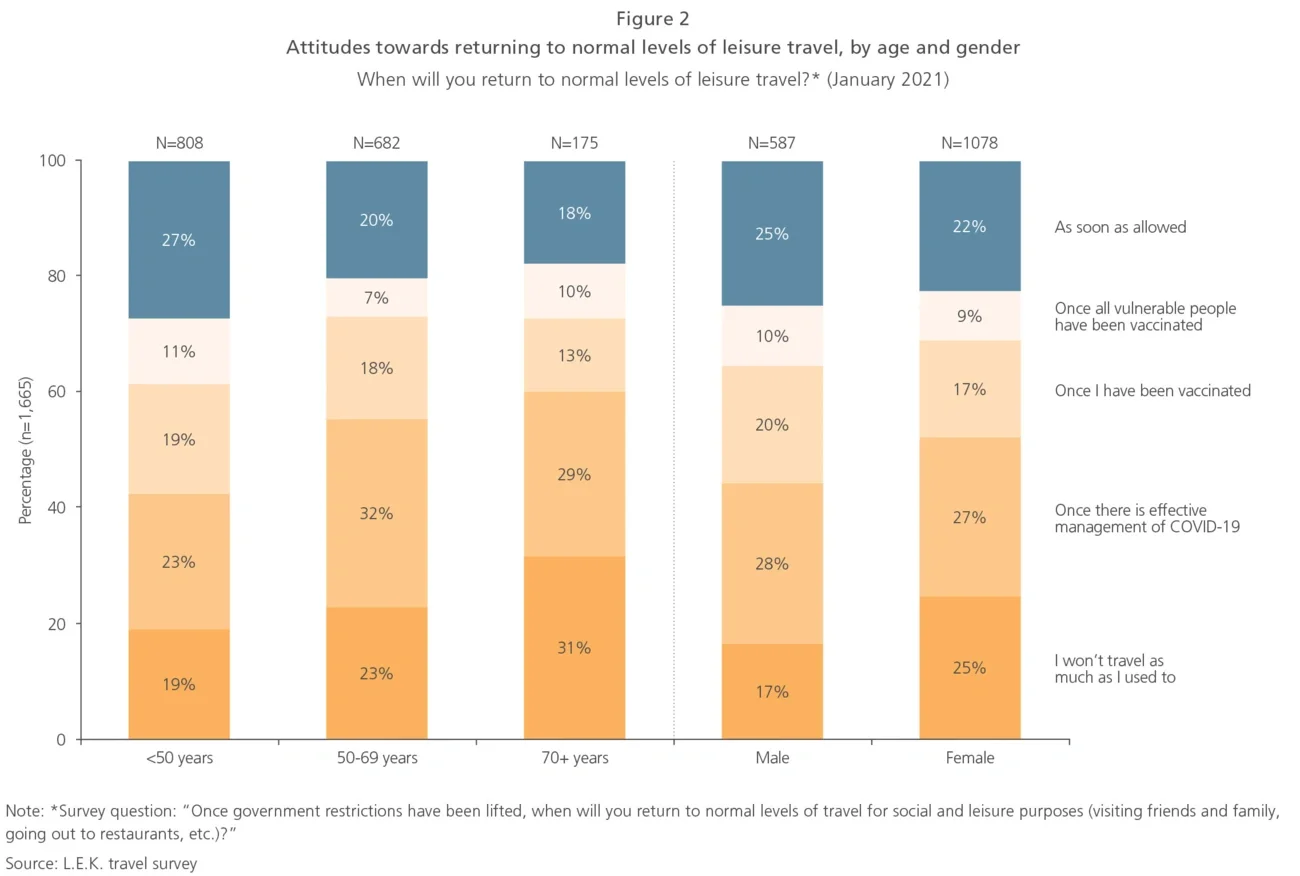
Early signs of success — selected case studies
While efforts to define and implement omnichannel models are ongoing, examples of a successful full-scale omnichannel overhaul in pharma are rare. However, there are a number of early success stories of pharma companies establishing omnichannel approaches for specific segments or built on a small number of channels.
In order to increase its market share for oral contraceptives, a large pharmaceutical company planned to introduce an app to remind women to take their pill on a regular basis. However, a detailed analysis of usage data unveiled that, at the current rate, achieving the envisioned market share growth would take eight years instead of the anticipated 18 months; in addition, the company would have to get 10,000 new prescriptions a month to reach its growth target, which was deemed unachievable with an app alone.
Based on market research and advanced analytics, target audiences with the highest potential for growth were identified, and the company’s sales force was redeployed to higher-potential locations based on refined brick data. Resources were also redirected to focus more on patients, with direct outreach to increase brand awareness and engagement through patient support programmes, as well as content such as blog posts on pregnancy prevention.
The integrated approach leveraging multiple channels helped build greater connections between the organisation’s two target audiences, doctors and patients, and overall sales force effectiveness was increased. In addition, the extensive market analysis created excitement and support amongst senior leadership within the organisation, including shareholders and owners. Overall, the product saw a growth of 23% in volume from new customers as a result of the optimised channel mix.
In the diverse European pharmaceutical market landscape, characterised by country-specific rules and regulations around pharmacy engagement, drug manufacturers are often not able to engage with smaller pharmacies directly to build relationships and offer promotions or discounts.
Leveraging existing technology platforms such as Salesforce and SAP, a multinational pharmaceutical company created a business-to-business (B2B) engagement platform that would allow it to design and deliver locally compliant content to pharmacists across European markets end to end without the need to outsource to third parties. The platform was built on scalable and localised architecture for multiple sites and languages, respecting the legal and language requirements of each country, and offering localised content and personalised communication for targets.
Through a single unified platform for content development, content management, pricing and sales, the multinational pharma company was able to access information and align strategic and business objectives across Europe. Initially released in France, Belgium and Luxembourg, the sales enablement and educational platform for pharmacists reached US $1 million in revenues and registered more than 900 pharmacies in the first three weeks of operation.
Example 3: Biopharma company increasing global ex-US reach and engagement without adding to field force
A biopharma company planned to increase its reach and engagement with key HCP customers for a major dermatology product across its non-US markets in a highly efficient manner, leveraging existing customer intelligence and without increasing field force resources.
Based on its existing intelligence, four key HCP segments were identified and engaged with through an orchestrated campaign covering email, paid media, portals and partner channels. An automated loop was also created to re-engage or progress key messaging based on HCP behaviours.
Through this orchestrated campaign and loop, the company was able to increase its target audience reach by 29% and engagement by 53% over the course of 16 weeks.
Example 4: Pharmaceutical company improving mature product’s positioning in Europe
Across the diverse and fragmented European market, a pharmaceutical company sought to increase the adoption of its mature respiratory product by improving the delivery and impact of its key messages.
Based on market research and analytics, a system of pre- and post-call rep-triggered events was created, allowing reps to choose further engagements and content appropriate to the particular HCP type, the desired key messages and the outcome of the call.
This systematic approach, combined with leveraging of the company’s Veeva software to automatically suggest content for follow-up email campaigns, resulted in increased email open rates among targeted HCPs by 46% and overall engagement by 73% over 10 weeks.
Key success factors
From early examples, we can derive some initial success factors for omnichannel.
- Developing relevant content: Content needs to be curated that fits both the channel and the segment. In order to stand out from the competition, it will be critical to ensure content is of high value — highly relevant, differentiated, well communicated and ideally customised — in order to drive engagement.
- Finding the right mix of channels appropriate for your audience at the time of engagement: This requires a deep understanding of HCP behaviours, meaningful HCP segmentation and identification of needs within each segment, and the ability to adapt as HCP behaviours evolve.
- Orchestrating the channels: Clear roles and interfaces between the channels are needed to ensure a seamless customer experience by mapping customer journeys across channels with appropriate handoffs.
- Establishing a ‘closed feedback loop’ to ensure continuous adaptation of engagement (content and channel mix): Sophisticated data collection and analysis capabilities, agility, and time are needed to optimise omnichannel engagement over time and incorporate potentially evolving HCP behaviours.
The creation of a successful omnichannel model can trigger significant organisational and behavioural changes in pharma companies. A number of factors should be kept in mind during implementation.
- Mandate from the top: Define a clear vision, and ensure that this vision and the resulting need for change are communicated by leadership in order to secure buy-in within the organisation.
- Deliver centrally: Establishing a centre of excellence is beneficial not only in leading the implementation but also in driving capability into business units.
- Review and adapt the operating model: Omnichannel engagement cannot be implemented without a hard look at the underlying operating model, in particular the processes and responsibilities of sales and marketing and medical functions.
- Leverage existing capabilities: Building on existing capabilities in an iterative fashion can drive quick wins.
- Manage change robustly: The changes triggered by omnichannel require many roles across the organisation to shift focus and call for robust change management.
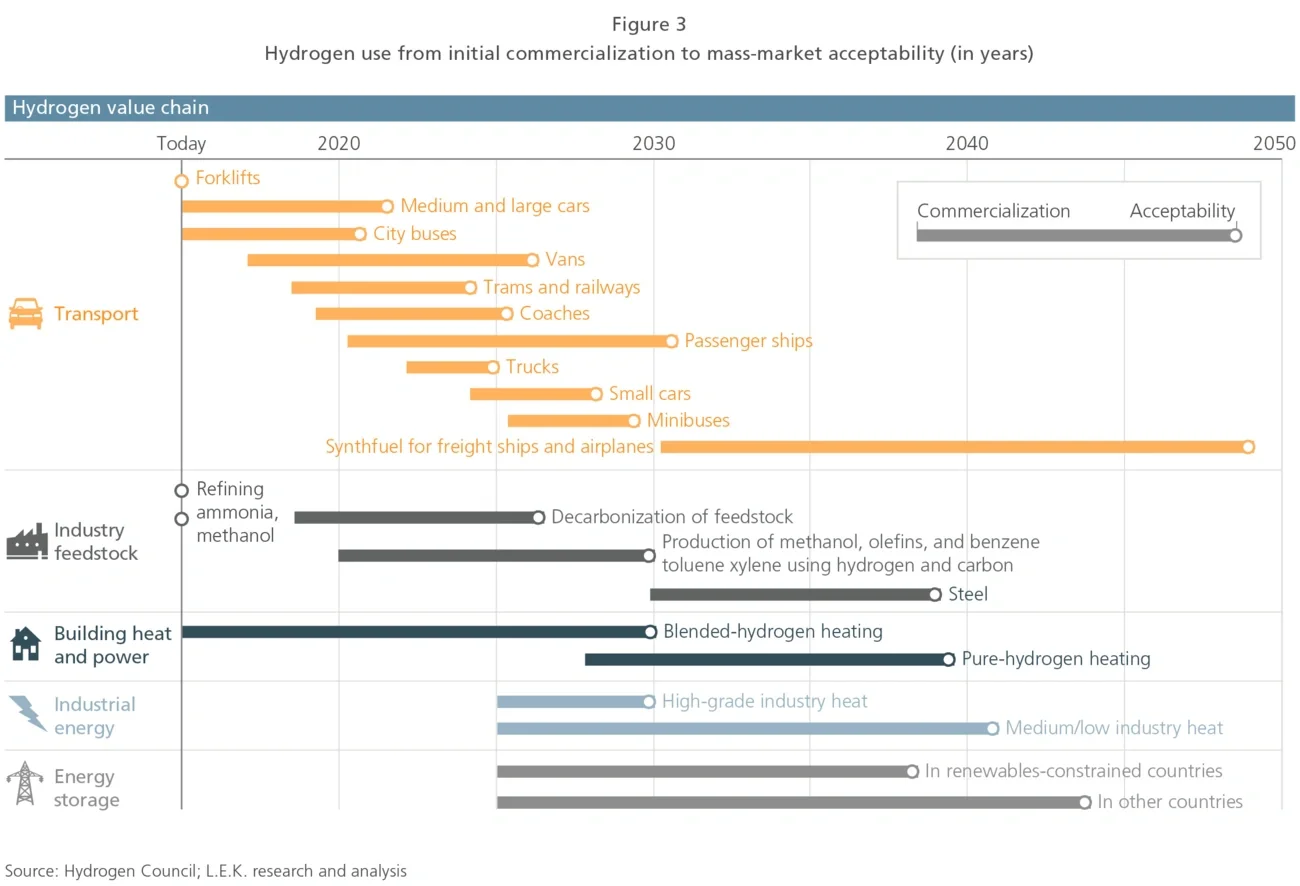
Before developing an omnichannel model, pharma companies need to consider a number of trade-offs around market benefits, existing capabilities and cost. Omnichannel models are inherently complex, and complexity grows exponentially with each channel, customer segment and content customisation, creating the risk of overwhelming marketing teams. It is therefore critical that pharma companies carefully assess the degree of sophistication needed along a number of dimensions to develop a fit-for-purpose and actionable omnichannel approach (see Figure 3) .