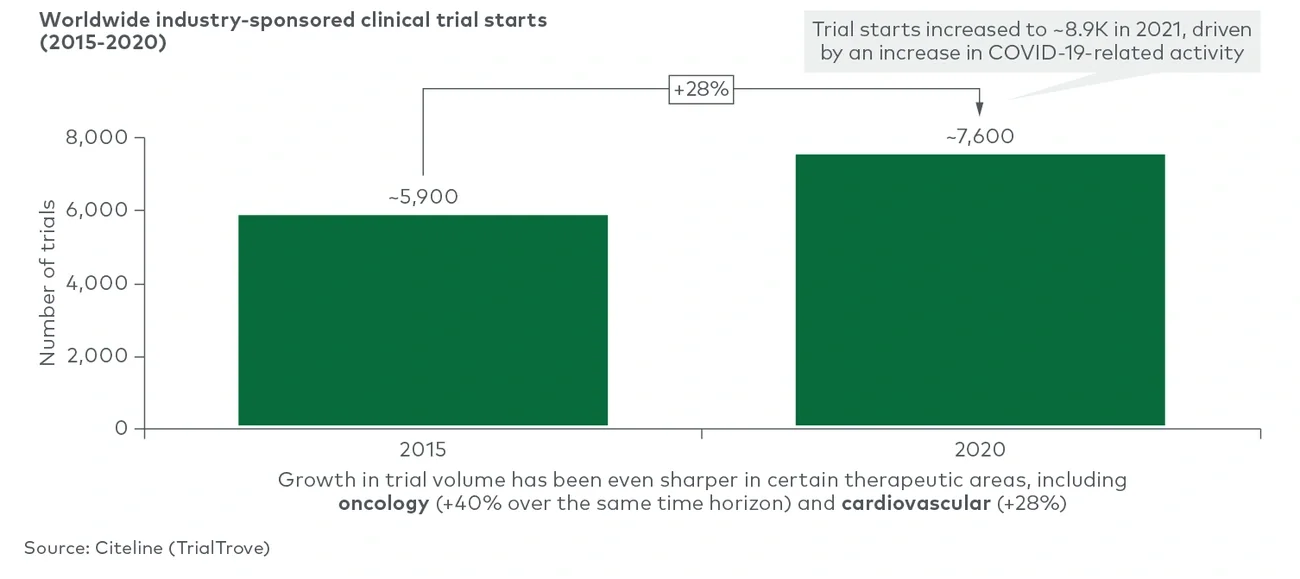
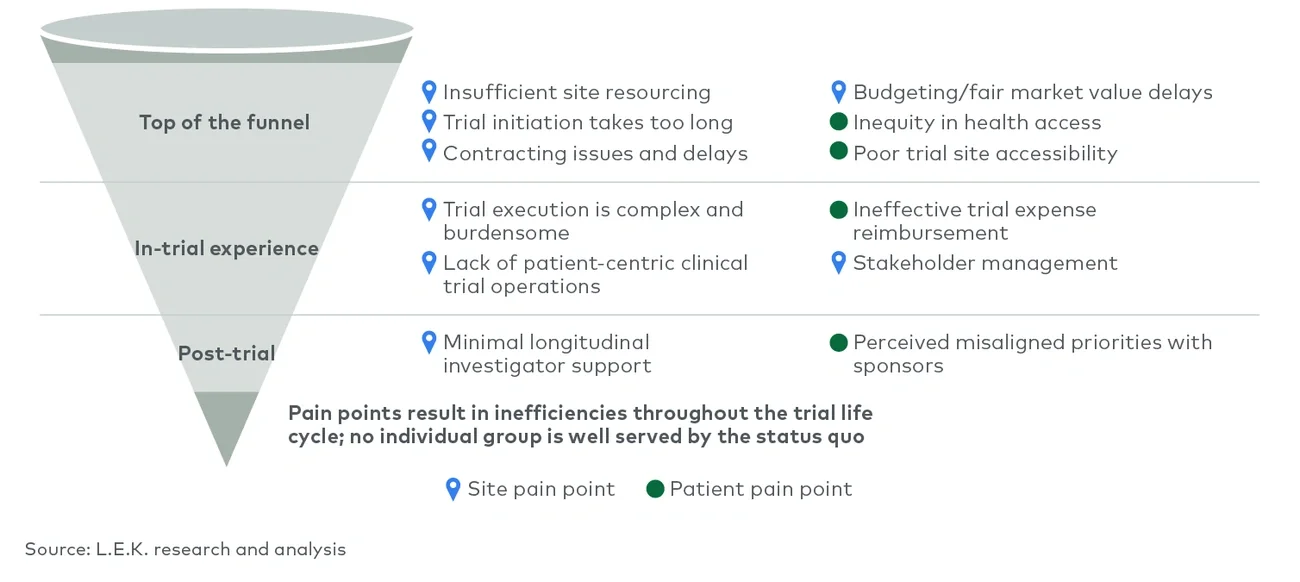
The biopharma industry has undergone tremendous growth over the past decade as technological improvements, new modalities and a supportive fundraising environment drove industrywide expansion. This dynamic has fostered a growing clinical pipeline and trial ecosystem to support it. This growth is particularly notable in highly competitive therapeutic areas such as oncology, where both volume and a diffuse range of trials per asset create a complex trial environment. This increase in volume and activity across the clinical pipeline has created a more competitive, crowded environment than ever before. However, within that increasingly dense development landscape, it is well known that stakeholders — sponsors, trial sites and patients — face a myriad of frustrations.
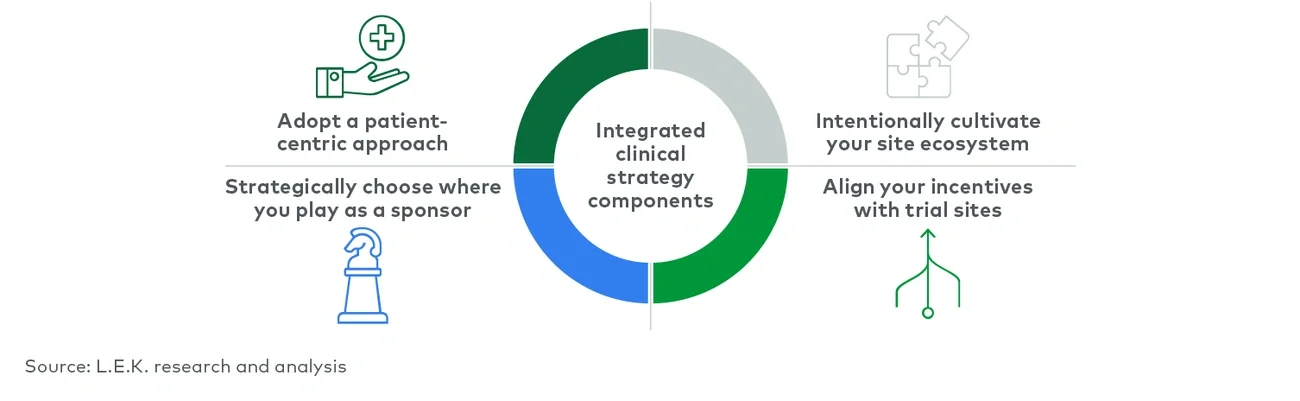
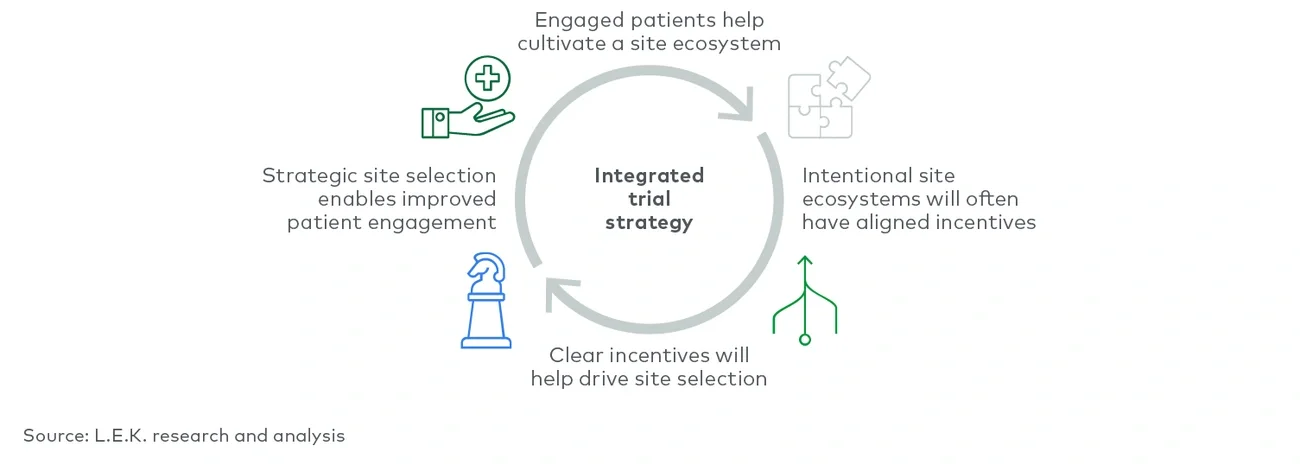
The status quo for trial execution is focused on getting patients into investigational therapy at a sufficient rate to meet endpoints; this method fails to account for an integrated, holistic approach to clinical trial execution that meets the needs of all stakeholders and fails to account for opportunities for leadership in meeting the diverse needs of patients, sites and principal investigators. Thoughtfully developing an integrated clinical trial approach enables sponsors to differentiate and develop solutions that work to strengthen and reinforce stakeholder relationships rather than fray them.