Biopharma has introduced a new design paradigm where success is less about highly iterative “discovery” and more about designing and assembling validated components. Rather than focusing on entirely novel targets or uniquely developed molecules for each design-make-test cycle, innovators are recombining proven building blocks to accelerate development, reduce biological risk and improve capital efficiency. The growth of the building-block tool kit, in turn, becomes an additional source of novelty and innovation for the modular medicines approach.
A vivid proof point was the rapid deployment of a personalized gene-editing therapy at Children’s Hospital of Philadelphia in spring 2025 (Baby KJ). While the scientific details of each of these cases will be unique, the operating model enabling these breakthroughs will be the highly modular therapeutics design: Leverage validated components, adjust to patient-specific circumstances, move fast as a disciplined quality and leverage the Food and Drug Administration’s emerging regulatory pathways.
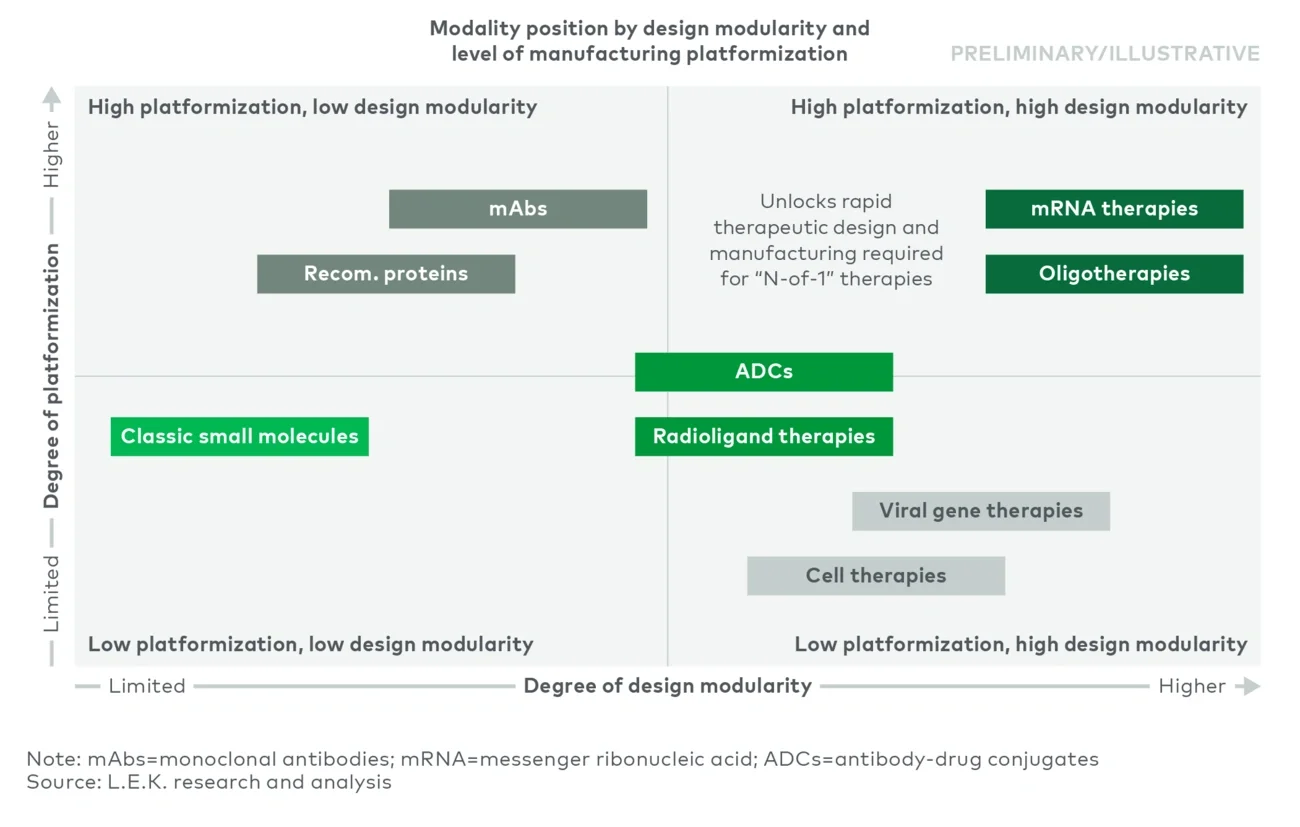
This edition of L.E.K. Consulting’s Executive Insights builds on our prior work on advanced modalities to explain why “design modularity” is emerging, where its impact is most pronounced and what it means for manufacturing partners and toolmakers.
Modular medicines defined
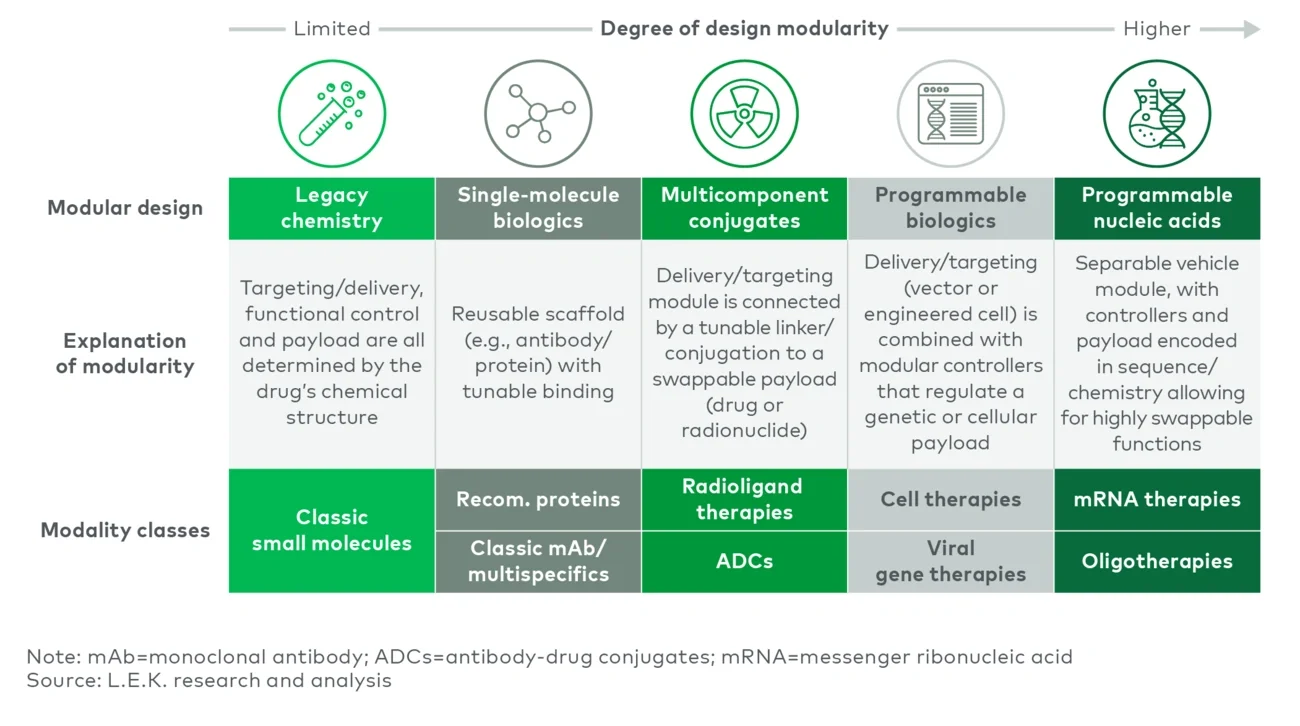
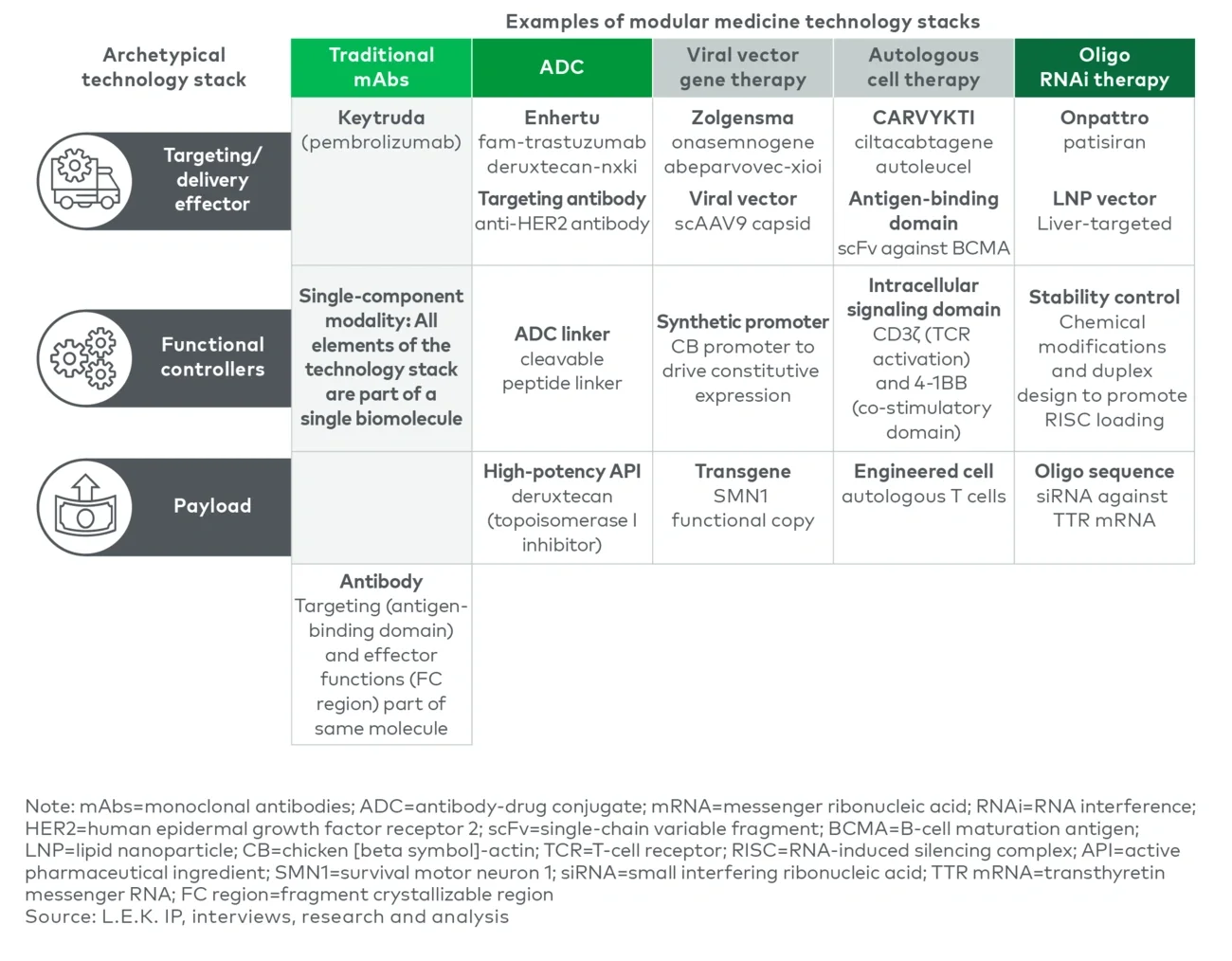
Unlike traditional drugs — constructed as individually optimized unique molecules resulting from intensive iteration and screening — modular medicines separate core functions into components that work together as an integrated system. Those components can be reused and recombined to design (rather than to discover de novo) new therapeutics, and the components themselves can improve over time, expanding the scope of design space.
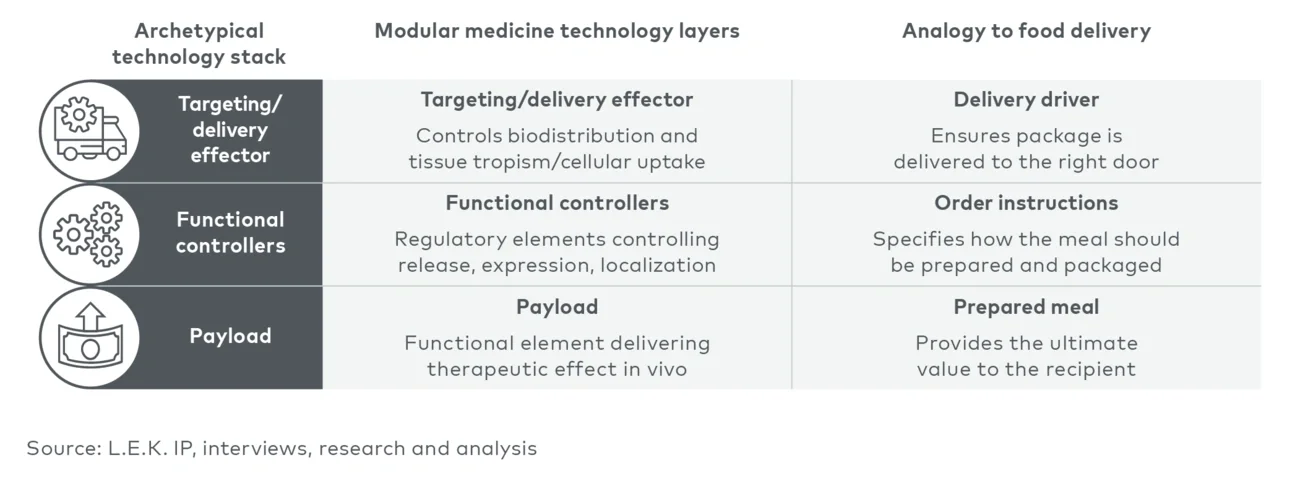
A practical segmentation is three technology layers:
- Payloads: the therapeutic “work” (e.g., genetic payloads, high-potency active pharmaceutical ingredients) that drives the biological effect
- Functional controllers: regulatory/tuning elements (e.g., untranslated regions, promoters, linkers, “kill switches” that turn off expression and mediate safety) that shape potency, pharmacokinetics, localization or expression
- Targeting/delivery effectors: the mechanism (e.g., adeno-associated virus (AAV) capsids, lipid nanoparticles (LNPs), monoclonal antibodies) that governs biodistribution and uptake
Food delivery is a useful analogy: The payload is the meal itself, the controller is the specific order instructions (“leave on the doorstep”) and the targeting/delivery effector is the driver (see Figure 1).