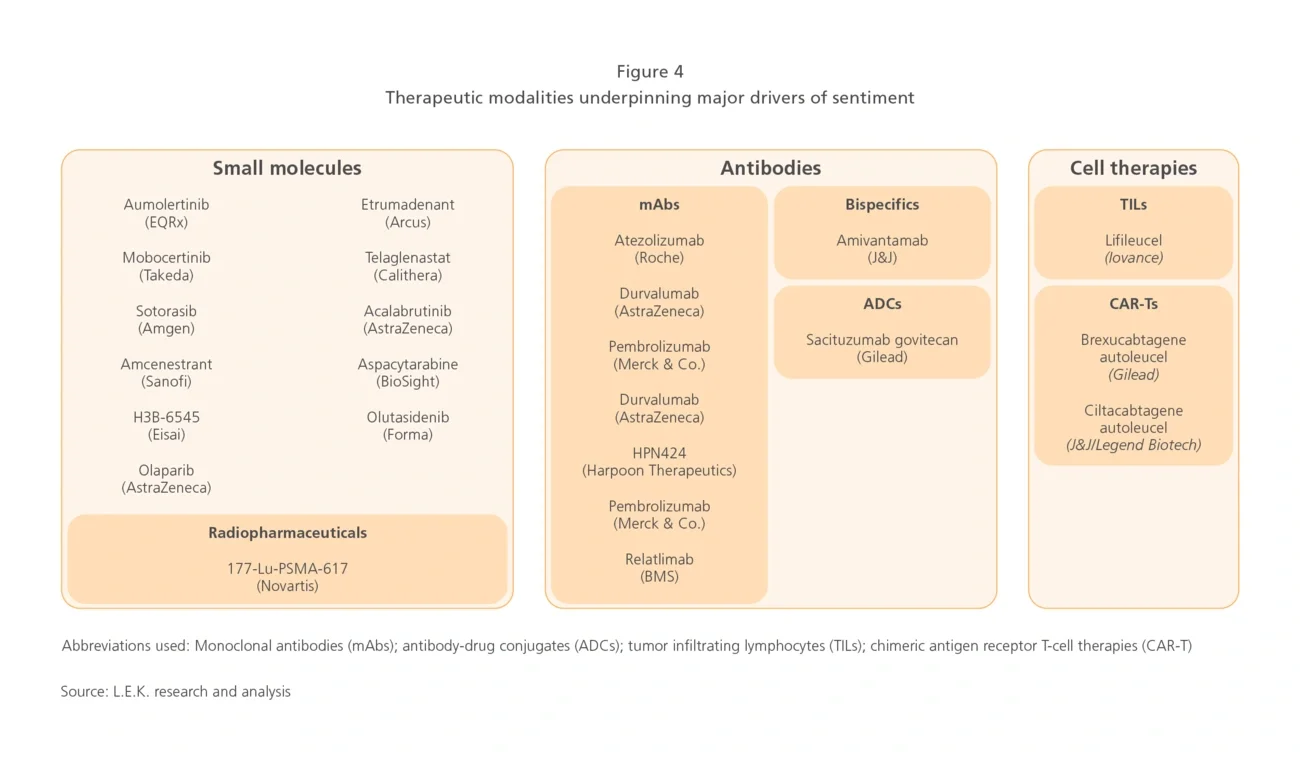
Breakthroughs in adjuvant settings
Data readouts for Olaparib (Lynparza; AstraZeneca) in HER2-negative breast cancer and pembrolizumab (Keytruda; Merck & Co.) in clear-cell renal cell cancer were notable as both demonstrated benefits in adjuvant settings. Both drugs were used following surgery and were shown to reduce the risk of disease recurrence or death. These and other drugs are aiming to move earlier in the treatment paradigm and follow the success of osimertinib (Tagrisso; AstraZeneca) which was approved for adjuvant use in EGFR-mutated NSCLC last year. Being used to prevent recurrence requires meeting a significantly higher standard for safety than when being used to treat active disease in later lines. This is because side effects are less acceptable in populations in which only a subset experience disease recurrence.
Rise of novel checkpoint inhibitors
Within the lucrative checkpoint inhibitor category there were several readouts for the established PD-1 and PD-L1 antagonist mechanisms. There is representation for LAG-3 antagonists, which is a highly anticipated mechanism being pursued by over 25 preclinical and clinical programs currently. Adenosine receptor antagonists are another novel checkpoint inhibitor mechanism which is being pursued by a similar number of programs.
Beyond these novel mechanisms, the next wave of innovation is expected to be in bispecific checkpoint inhibitors. They target various combinations of PD-1, PD-L1, CTLA-4, LAG3 and other targets, and there are more than 30 in Phase II development and earlier.