China is now the second largest pharmaceutical market in the world.1 China’s oncology market alone represents 37% of all lung cancer, 44% of stomach cancer and 52% of liver cancer patients globally.2 Large multinational pharma companies are carrying out initiatives to succeed in the market, and more biotech companies are entering China through either partnerships or greenfield strategies.3 Diagnosis and treatment patterns in China continue to be diverse due to the variation among different levels of hospitals. A comprehensive view of the oncology market is needed to design a winning strategy. In this introduction, we highlight opportunities for international market entrants and provide an overview of the patient and physician landscape in China using LinkDoc’s disease database. The lung cancer market is featured as an illustrative example.
China has led emerging markets in oncology therapeutics spending and growth, growing by 24% in 2018 to reach U.S. $9 billion in spending.4 Furthermore, China has the largest number of cancer patients globally, with 4 million newly diagnosed cancer cases per year in 2018, more than double the number of new cases per year in the U.S.2 In addition, China’s cancer mortality rate is 40% higher than that of the U.S.5 Lung cancer rate, and is the most common cancer type in China, with an overall five-year survival rate of 36%.6 This results in an urgent medical need and continued demand for better lung cancer treatment options in China.
Innovation and reimbursement trends
To address the high cancer disease burden, China has made efforts to drive innovative oncology drug development, which have seen significant results:
- More than 700 cancer therapeutics from international companies and local Chinese companies underwent nearly 1,500 clinical trials in China between 2009 and 2018.7 Approximately 40% of these cancer therapeutics are targeted small molecules, similar to global market trends.4,7
- Although China had only 10% of the clinical trial volume of the U.S. over the past nine years and its immuno-oncology therapeutics development outside of PD-1/PD-L1 targets is lagging behind the global landscape, recent China policies that enable innovative drug development are expected to further increase the number of clinical trials and innovative immuno-oncology drugs in the future.7,8
- More International Multicenter Clinical Trials are being registered and conducted in China for parallel drug launch in the U.S. and China market.9 Pharma companies now leverage patient resources in China to accelerate enrolment.
As top clinical sites and principal investigators are in high demand, pharma companies need to think about how to strategically design drug development pathways to launch in the China market with first-mover advantage.
Significant increase in volume can take place after reimbursement. For instance, Roche’s revenue has grown at ~28% from 2017 to 2018 with the inclusion of three key products (Herceptin, Avastin and MabThera/Rituxan) on the NRDL in 2017. However, not every reimbursed drug can reach the sales volume necessary to offset the sharp price decline. Therefore, pharma companies with innovative drugs in the China market need to define an appropriate pricing strategy for reimbursement and identify how to optimize the drug’s value proposition after entering the reimbursement list.
Targeting patients
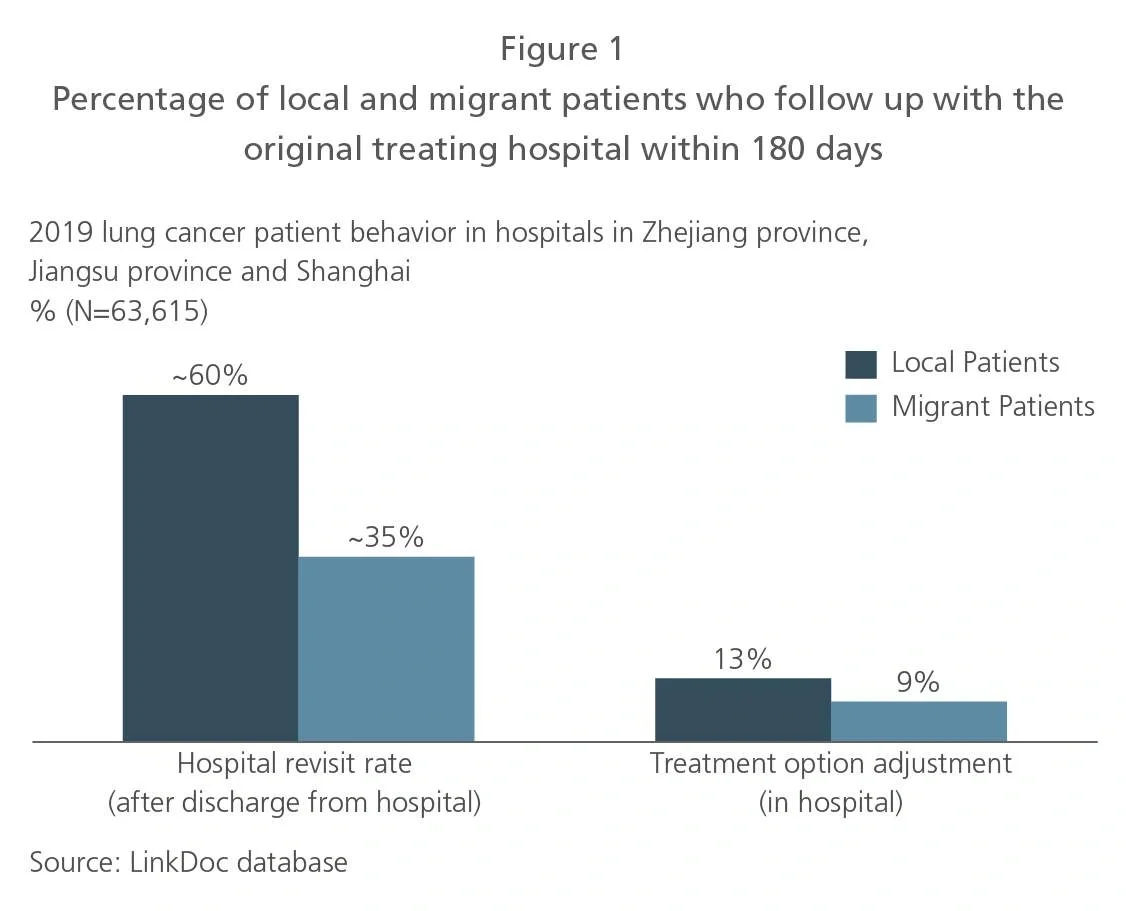
To optimize the value proposition, pharma companies need to be aware of where their target patients are. Given the unequal distribution of healthcare resources in China, lung cancer patients typically go to affluent cities with better hospitals for initial diagnosis and treatment. In Beijing and Shanghai, for example, about 70% and 45% of the respective patient populations are nonresidents.11 Most patients subsequently return to their hometown for ongoing treatment. The percentage of on-time follow-up visits is significantly lower for migrant patients than for local patients (see Figure 1).