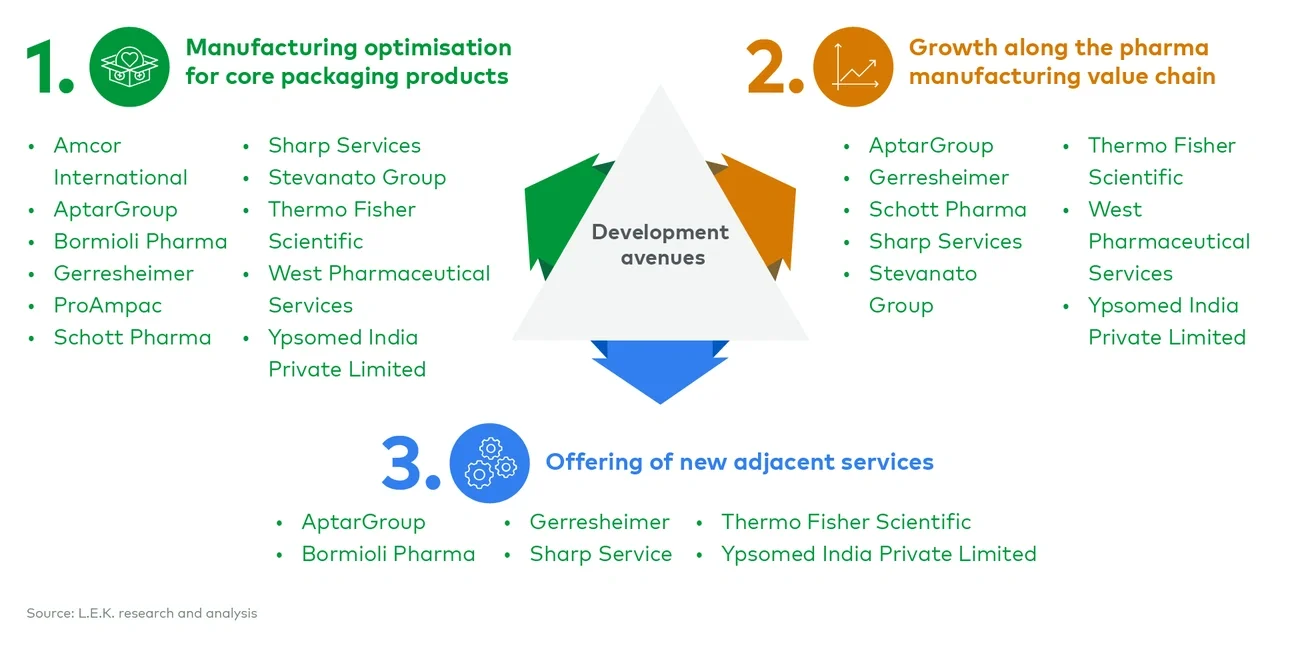
CPOs are under increasing pressure from biopharma customers to reduce costs, enhance the sustainability of their processes, and support Pharmacos in creating additional value for their products. To address these demands and differentiate their service offerings, CPOs have focused on three main growth avenues:
- Optimising manufacturing of core products. CPOs are focusing on efficiency and sustainability in their primary services to reduce costs and meet client demands.
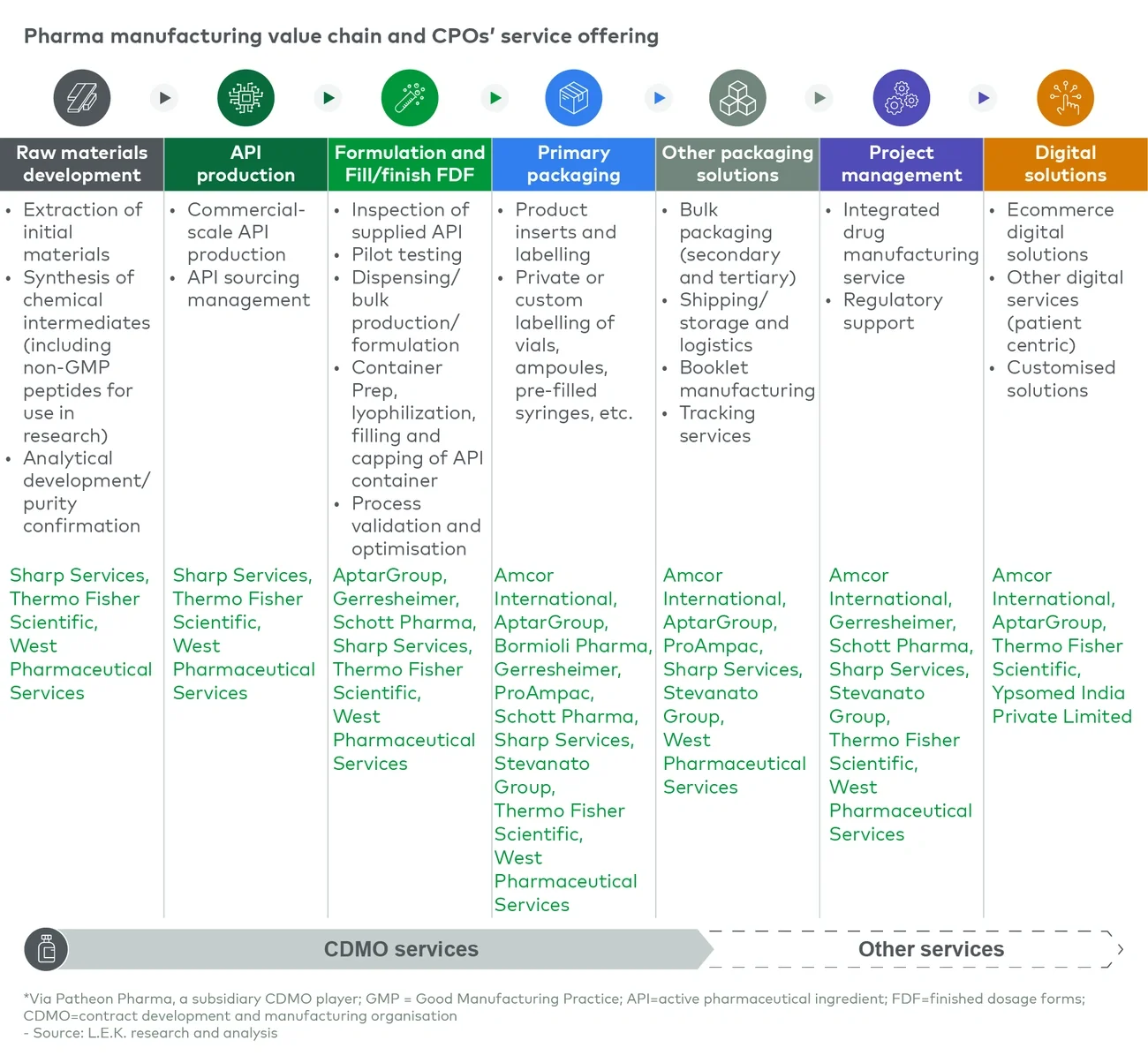
- Integrating services along the pharma manufacturing value chain. Covering a broader set of services to consolidate buying points and interfaces along the value chain for the client.
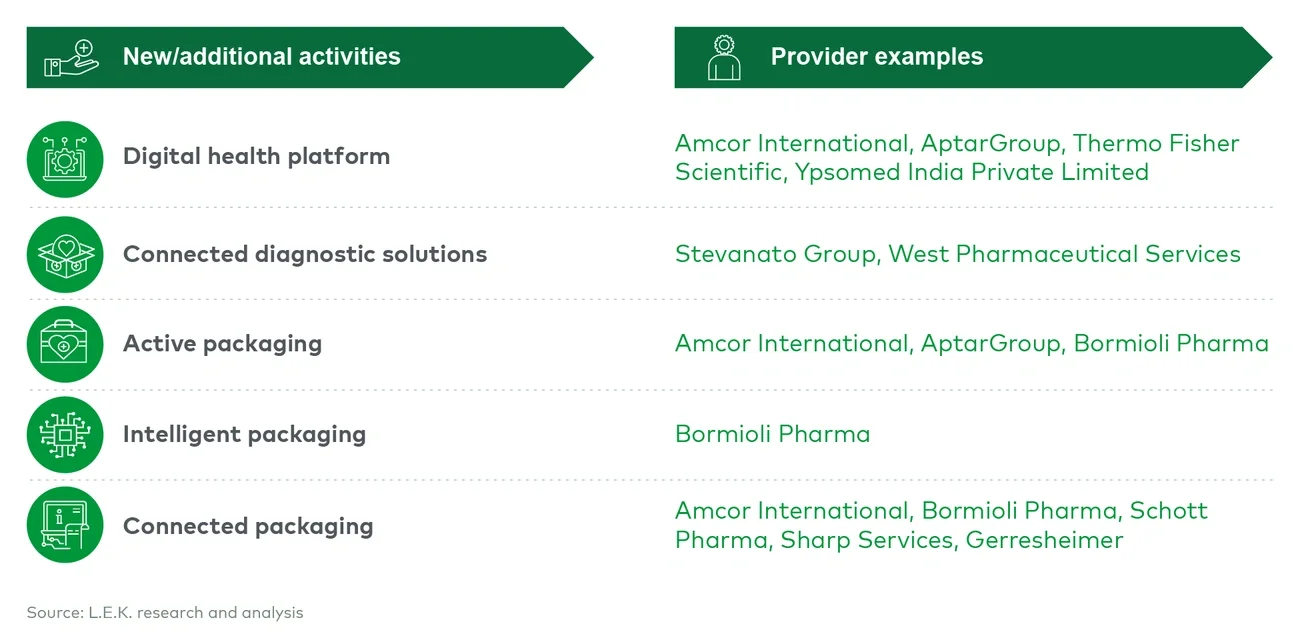
- Expanding offerings into new adjacent services outside manufacturing. CPOs are looking beyond traditional boundaries to find new opportunities for growth and differentiation.
In this Executive Insights, L.E.K. Consulting outlines the rationale supporting these growth avenues and reviews some of the key areas that major players have explored in recent years (See Figure 1). This research was recently presented as part of L.E.K.’s panel discussion at PharmaPack 2024.