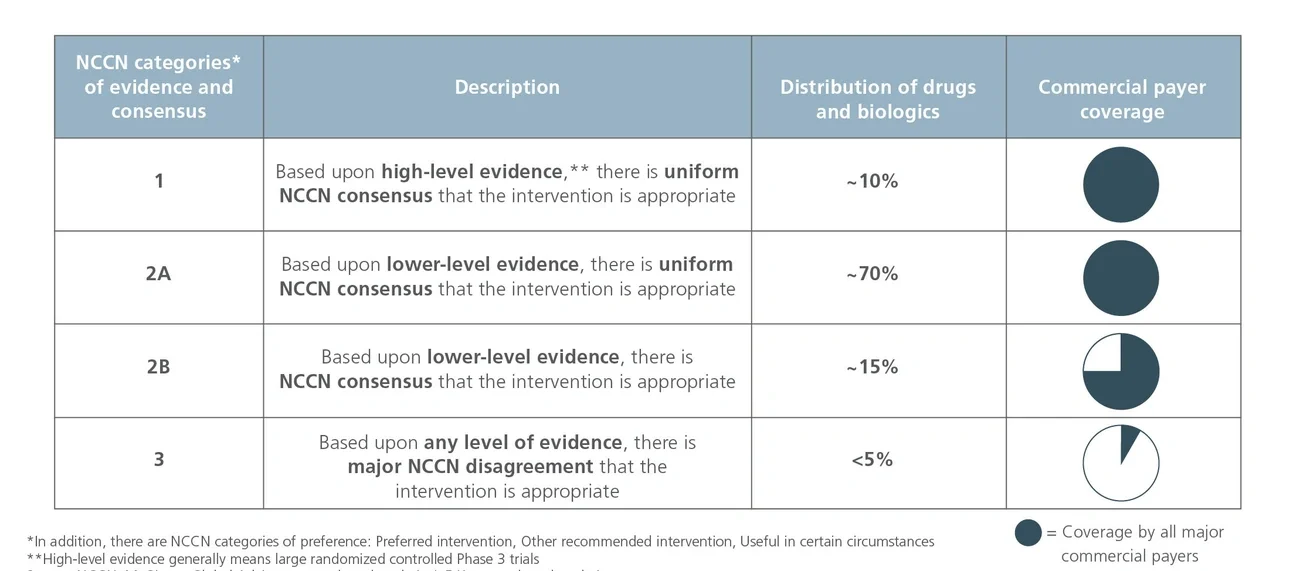
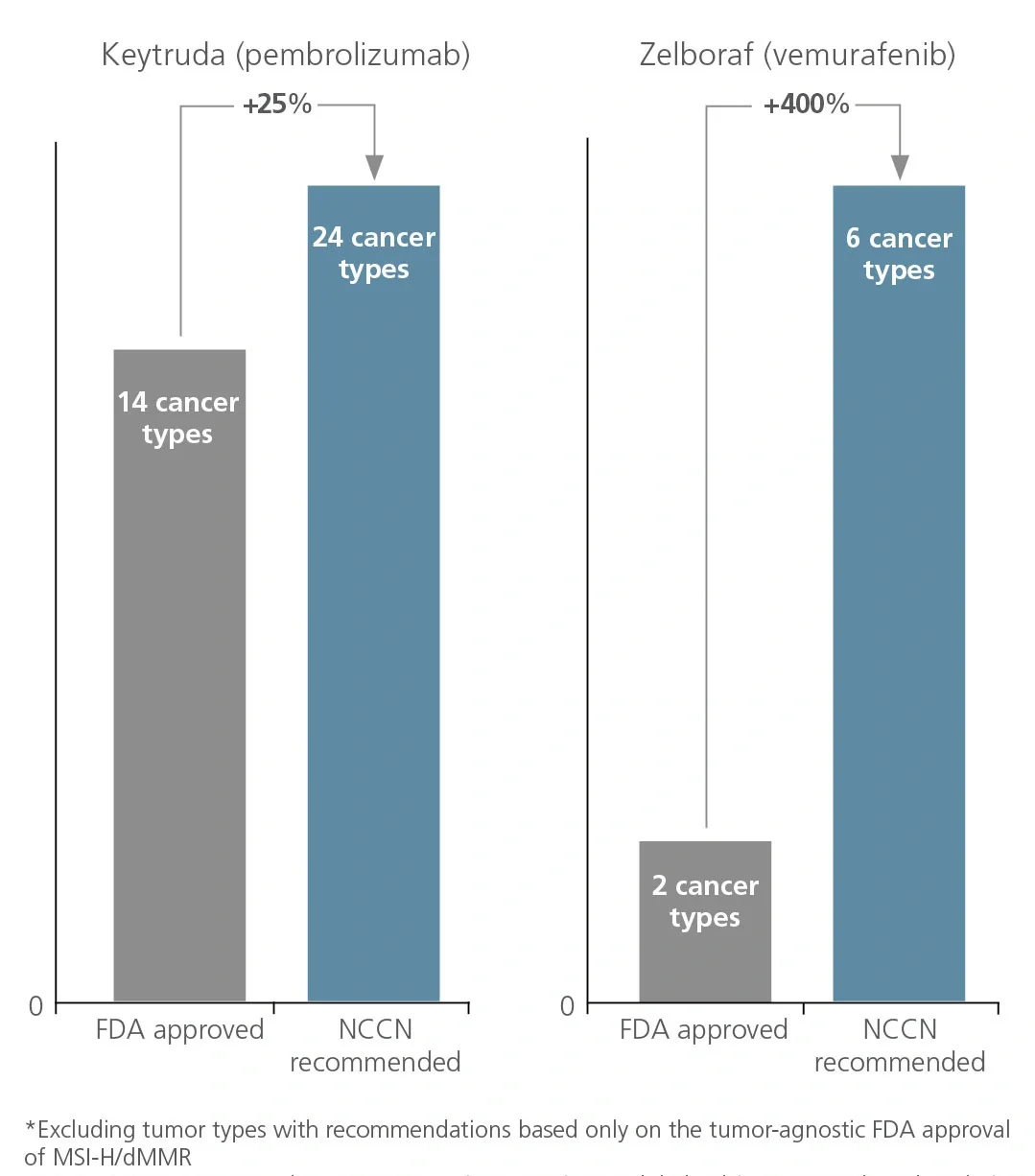
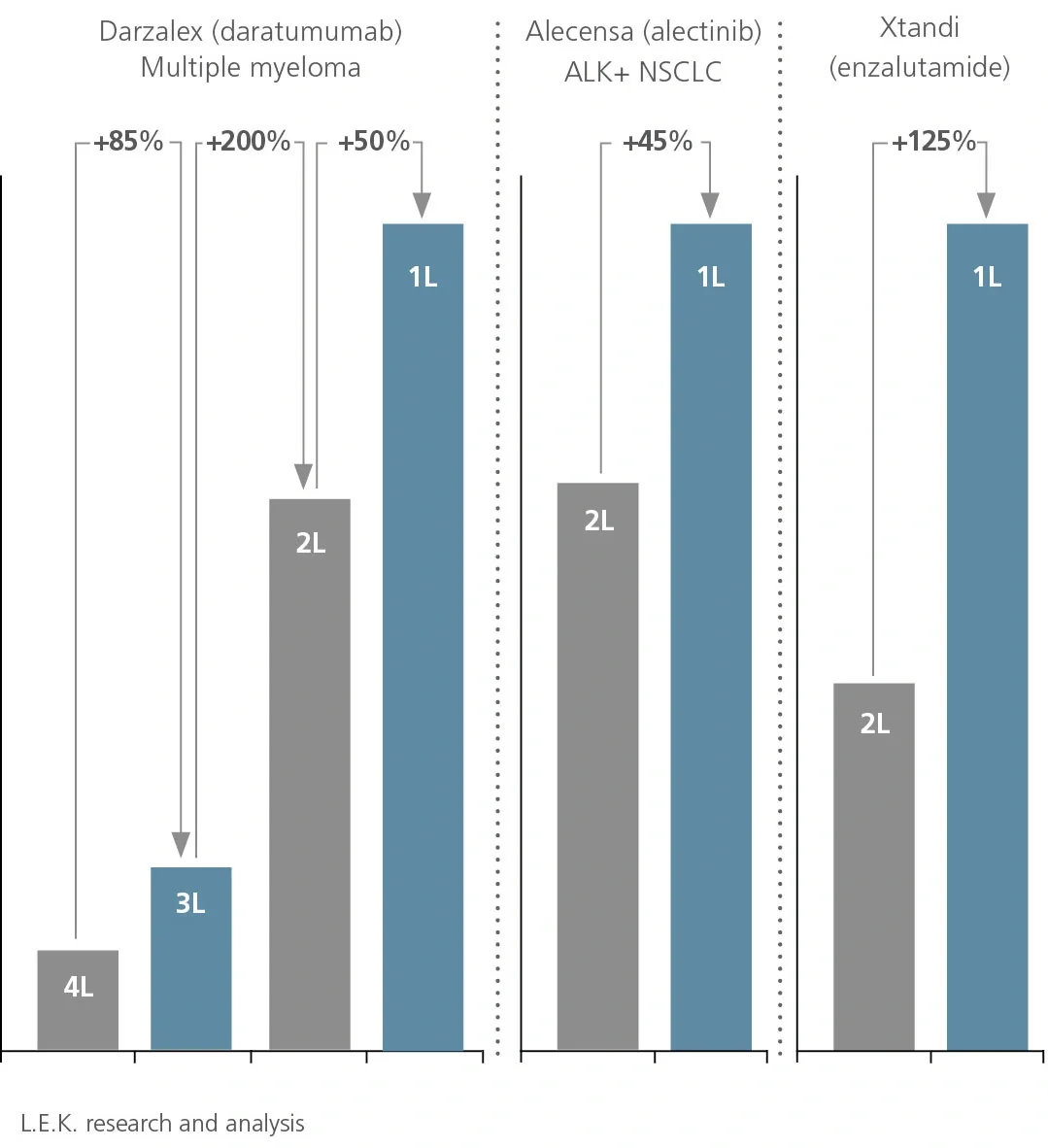
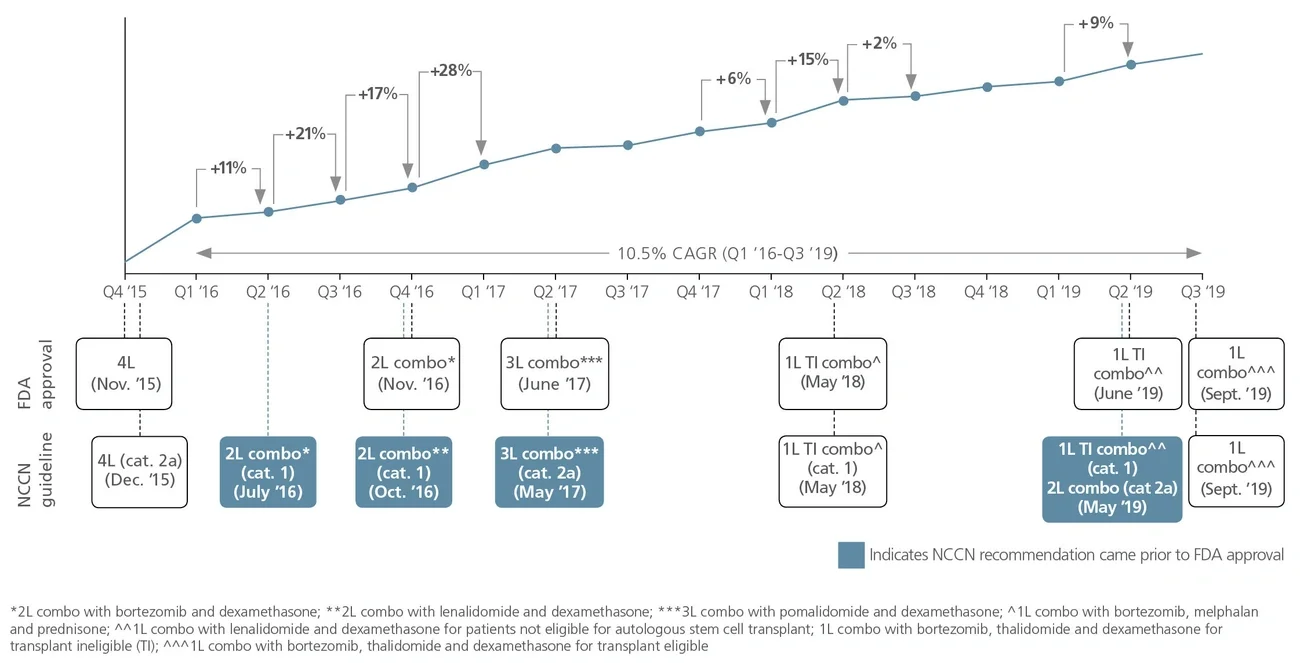
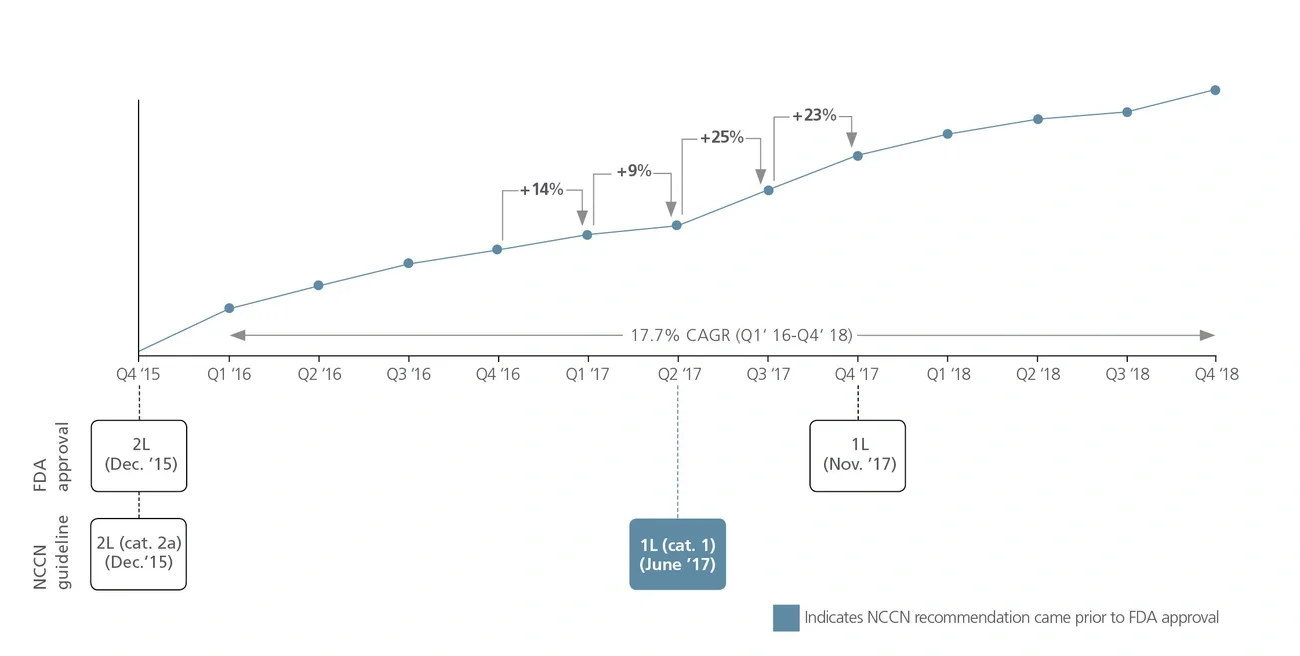
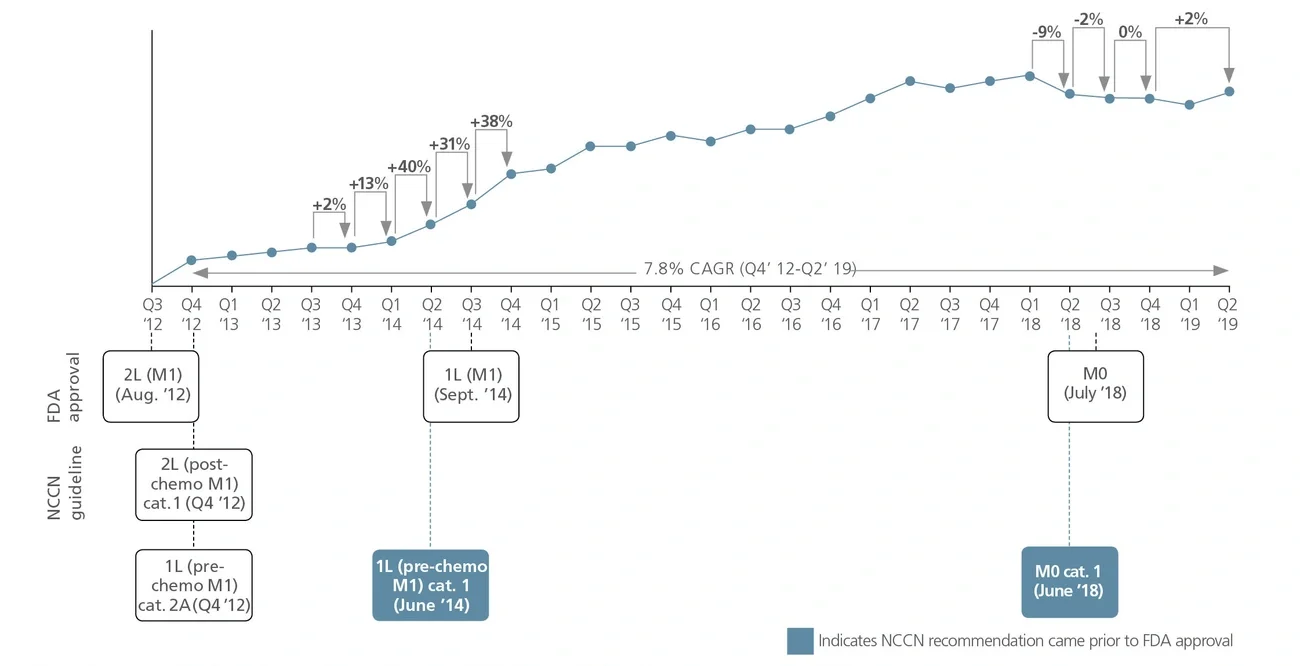
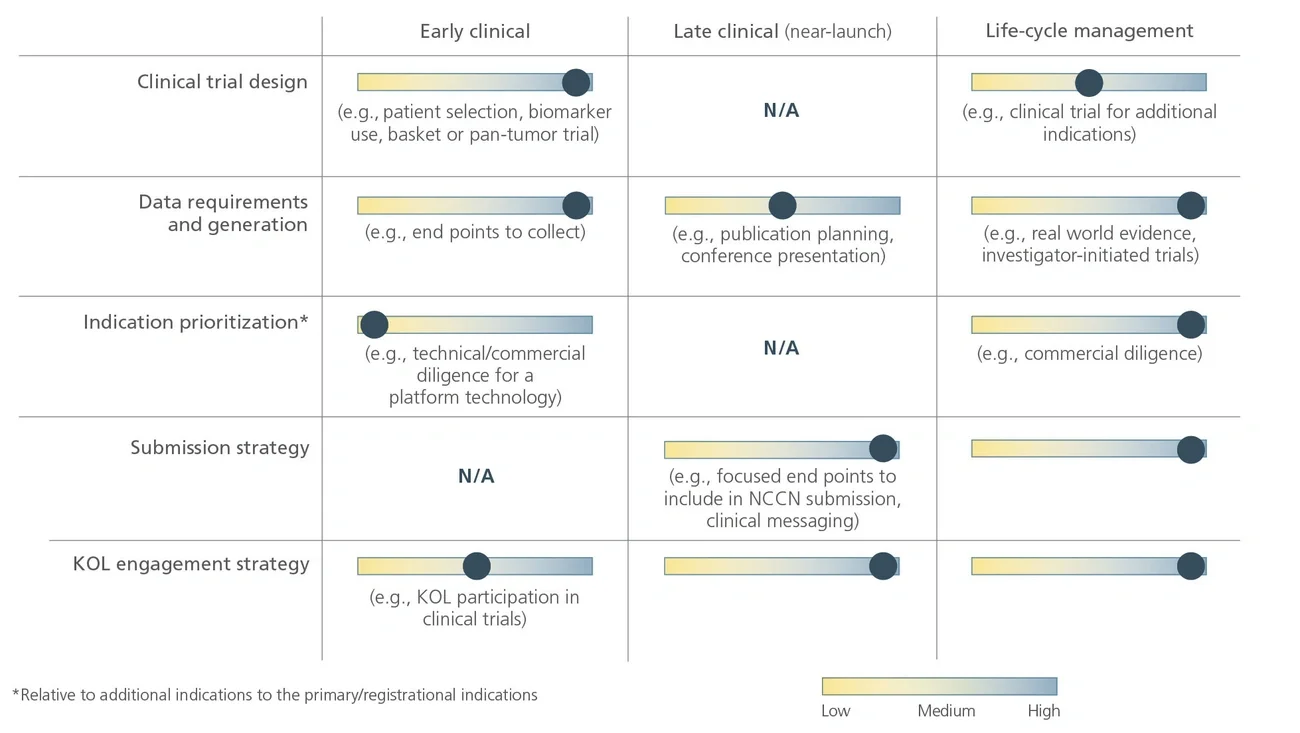
National Comprehensive Cancer Network (NCCN) recommendations are considered to be the gold standard for public and private insurers to use in making coverage decisions for both labeled and off-label use of new and existing oncology medicines. NCCN recommendations drive drug adoption in earlier lines of therapy and in other indications (i.e., beyond primary/registrational indication), including off-label use. It is critical for biopharma companies to have a robust NCCN strategy that encompasses multiple cross-functional stakeholders across all stages of a drug’s life cycle to maximize the patient and commercial impact of lifesaving cancer treatments.
Oncology is an extremely attractive market for biopharma companies. Total worldwide sales are expected to grow from ~$125 billion (~15% of total worldwide market share) in 2018 to ~$235 billion (~20% of worldwide market share) in 2024, which is a compound annual growth rate (CAGR) of ~11%.1 Additionally, ~35%-40% of blockbuster drugs launched between 2011 and 2016 were oncology drugs,2 and most came with a high price. However, despite recent advances in drug development, there is still a significant unmet need across almost all cancer types, driving a significant amount of R&D investment and a large pipeline with thousands of drugs under development by hundreds of companies. Consequently, these complex dynamics impose unique planning challenges for biopharma companies such as:
- Driving or catching up with the rapid pace of innovation (e.g., emergence of biomarker-based pan-tumor therapies) and evolving treatment paradigms such as targeted therapies3 and staged treatment
- Navigating the uncertain, expensive and long FDA approval process
- Gaining favorable payer coverage in an increasingly competitive and value-focused environment
- Maximizing the addressable patient population size
- Facing fierce competition across all stages of a drug’s life cycle
- Planning and managing indication expansion to extend a treatment’s life cycle and finally
- Securing payer coverage for use beyond the FDA-approved label (i.e., “off-label” or “expanded indication,” which is a significant component of many successful oncology treatments)
Some of these challenges also complicate decision-making for physicians (e.g., choosing among treatments and making sure they are covered by insurance), patients (e.g., deciding between competing options), employers (e.g., determining how to provide appropriate access to novel treatments), and government, as well as private payers (e.g., deciding on the appropriate level of access to provide).
Importance of CMS-recognized drug compendia
To help navigate the complexities mentioned above, several organizations have developed drug compendia that contain information compiled by experts based on clinical evidence about the drug’s characteristics (e.g., pharmacologic profile, dosage, clinical performance) and that may include recommended uses. Currently, there are five drug compendia recognized by the Centers for Medicare and Medicaid Services (CMS) for use in the determination of medically accepted indications and to inform coverage decisions (see Figure 1).