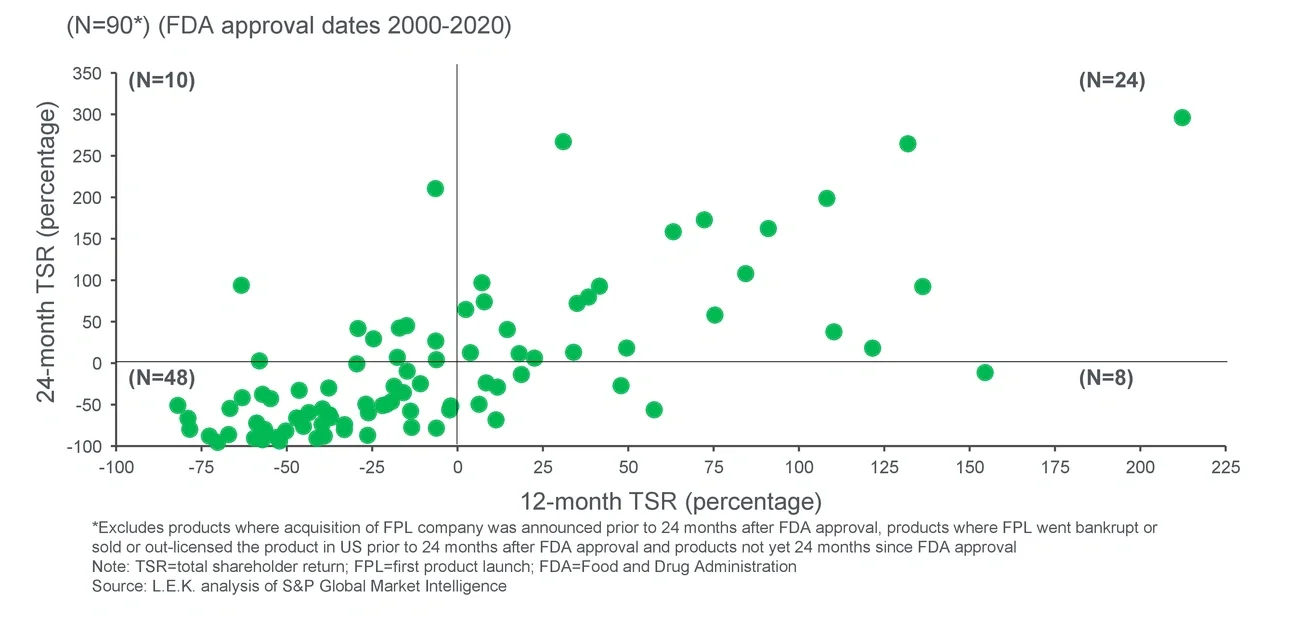
Partnering in the U.S. is a decision that can impact shareholder value creation. Companies that opted for a co-promotion generated more shareholder value than those that went it alone; 45% of first-time launchers that co-promoted in the U.S. generated positive returns at the two-year mark compared with 35% of first-time launchers that entered the U.S. alone. While this may be the result of a selection bias on the part of pharmaceutical partners seeking co-promotion agreements on only the most promising drugs, we found that first-time launchers co-promoting in the U.S. uncovered significant value from their partner’s existing capabilities and established relationship with providers and payers.
Key success factors for first-time launchers
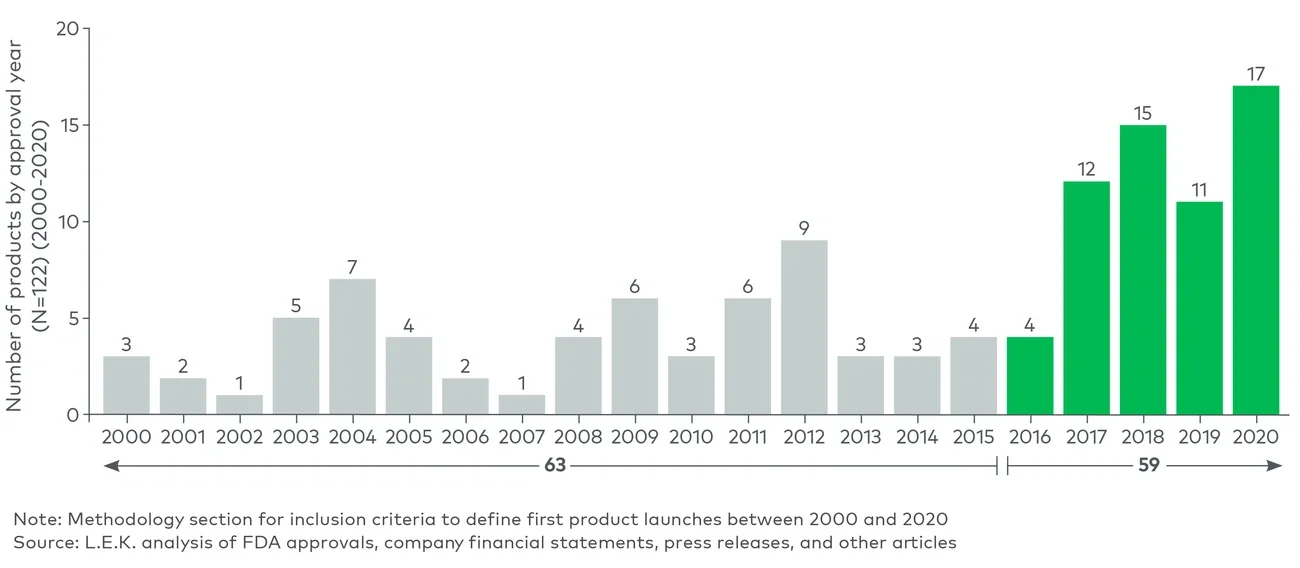
Given the increasing contribution of development-stage companies to biopharma innovation, L.E.K. anticipates the number of companies participating in the commercialization of their first product to continue to increase in the near future. To be successful in this journey, first-time launchers will need to pay close attention to four success factors:
-
Design a clear-sighted operational scale-up
The launch process is complex, often involving unforeseen challenges around everything from supply chain coordination to patient support. Given the focus of first-time launchers on minimizing burn rate and reducing dilution, some of them delay the funding of market-shaping activities as long as possible, favoring just-in-time investments at key de-risking clinical and regulatory milestones. This guarded approach often puts key commercial activities at risk and leads to launch delays that compromise the overall revenue performance given the difficulty in recovering from a slow start and early missteps.
Instead, first-time launchers must invest early, typically two to three years before approval, in building commercial, medical and enabling infrastructure, even while significant technical and regulatory risk around their product remains. Hiring a commercial leader is a critical first step to jump-start activities such as designing the desired end-state for the organization, building teams in critical functions and engaging with key stakeholders to gain early market insights. Although launch teams wear a lot of hats in these organizations, they must be diligent about identifying interdependencies across launch activities and remain coordinated through cross-functional teams to orchestrate the launch journey.
Equally important is the process of setting mechanisms to collect internal and external market information in real time, enabling first-time launchers to pressure-test product positioning, monitor progress and correct the launch strategy when needed. To collect this information in today’s environment, customer-facing teams need to take advantage of remote engagement solutions to interact with key stakeholders and communicate feedback to the organization and a new direction for the launch.
Lastly, boards of first-time launchers can play a critical role in overseeing and approving the launch strategy. First-time launchers benefit from having biotech executives on their boards who have been on this journey before and can bring real-world experience and feedback to the management team.
-
Choose the optimal partnering strategy
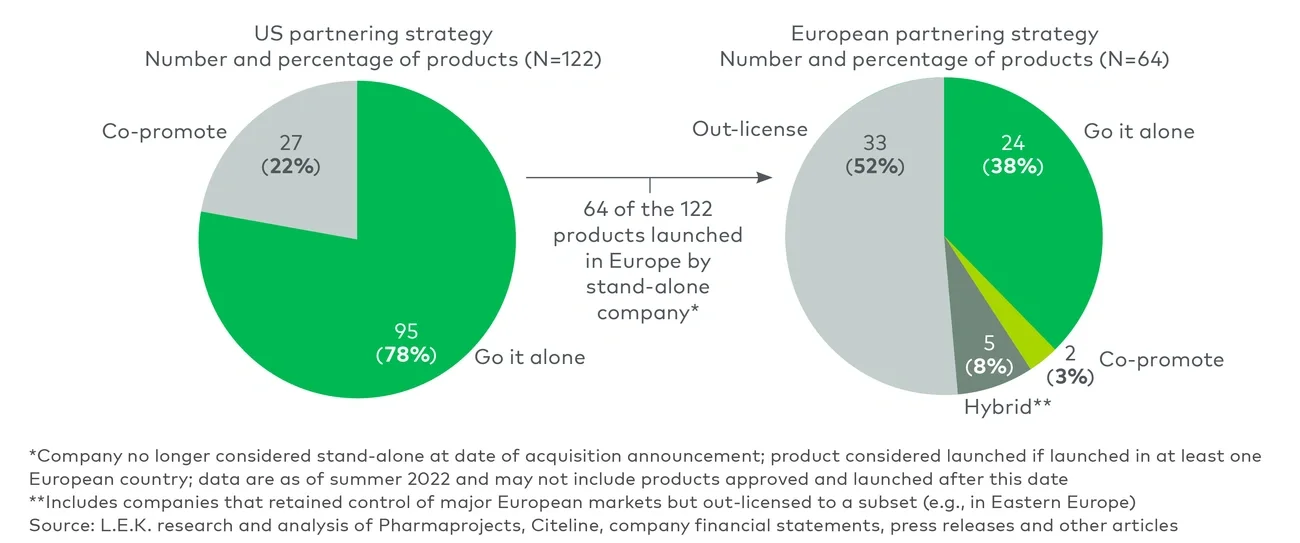
The first step in developing a global partnering strategy is to quantify the magnitude of the product’s commercial opportunity in major markets as well as the operational and capital requirements just for commercializing the product. A minimum revenue threshold must be established to offset the cost of entry and create value across each international market. Launch teams must identify countries meeting these thresholds early to prioritize countries worth pursuing through a direct model.
For orphan diseases and oncology niche indications with little competitive intensity and a modest level of required commercial investment, our close observation of first-time launchers shows that they can target 15 to 25 geographies where value is created on a go-it-alone basis. Direct geographic expansion in these markets also unlocks synergies with the U.S. and provides greater control over global branding and pricing of first-time launches.
However, a direct model is not without risks. Regulators in Europe often call for data from local clinical trials or real-world evidence, payers repeatedly place constraints on the pricing and reimbursement of approved drugs, and launch delays can be substantial. Importantly, executing concurrent launches across the U.S. and Europe presents operational challenges for centralized teams trying to provide as much support as they can abroad without disrupting execution of their U.S. commercialization plans.
Once decisions around international partnering have been made, first-time launchers can turn their attention to the U.S. commercial strategy. We found that many companies created tremendous value by going it alone in the U.S., particularly for specialty markets requiring focused infrastructure that would be difficult to build and share with a partner.
For more complex markets with high barriers to entry, first-time launchers can opt for co-promotion agreements that allow them to leverage the existing capabilities and stakeholder relationships of a potential partner. This model can accelerate the first-product launch trajectory and reduce the investment burden and risk of going it alone. It also allows first-time launchers with deep pipelines to mobilize their resources to develop additional programs or prepare for future launches. That said, co-promotion requires strong capabilities in managing the partner relationship to circumvent governance complexities and lessen cost inefficiencies resulting from duplication of efforts.
-
Plan for sufficient capital formation
Companies preparing for a first product launch need to carefully manage the financing of multiple critical, costly priorities. That includes developing a compelling clinical plan for their first product, building a robust pipeline to lay the foundation for future growth and scaling up commercial operations to support the first launch.
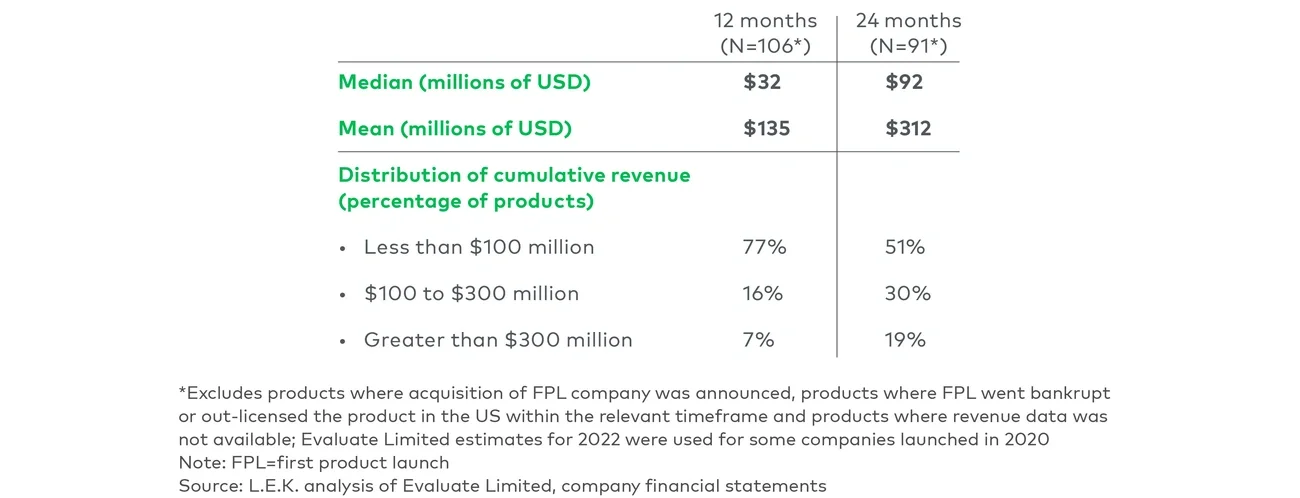
Our analysis shows that first-time launchers often tend to underfund these priorities and underestimate their capital requirements. It is therefore critical for these companies to build clinical and commercial scenarios that account for regulatory delay, suboptimal labelling or unexpected competitive threat to their first product launch. A careful evaluation of cumulative cash requirements in a worst-case situation can help shape the financing strategy by informing choices between nondilutive upfront payments from partnership, dilutive secondary equity offerings or alternative financings such as synthetic royalty monetization. And in today’s challenging environment, first-time launchers need to be more vigilant than ever in assessing these funding options and ensure they build a runway to long-term financial sustainability.
-
Manage investor expectations carefully
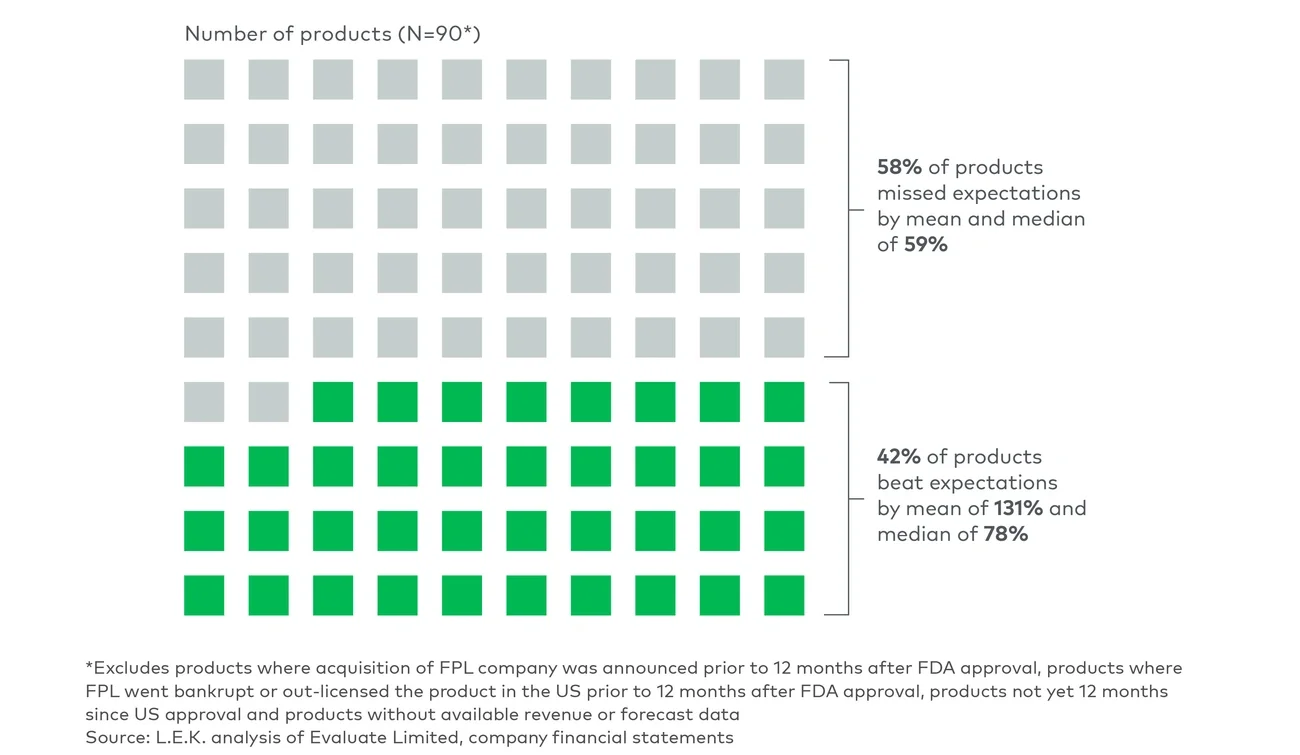
Armed with a deep knowledge of the product value proposition, first-time launchers must communicate their product differentiation in the market and ensure achievable revenue expectations on the part of investors. Overstating the magnitude of their product’s commercial opportunity or allowing misguided Street projections to prevail can lead to short-term share price gains but quickly expose the company to valuation correction once their product starts missing consensus estimates.
Guiding investors on achievable revenue expectations is therefore a more prudent approach but a difficult one to manage. Biopharma leaders first need to be cautious about over-communicating to investors before launch. Assumptions to share externally include detailed clinical trial data, facts about disease epidemiology and comparative analysis of competitor product profiles. Beyond that, companies should be cautious about disclosing too many views on the anticipated revenue performance of their product. Instead, they should react in a subtle manner when needed to counter investors’ presumptions. A recent example of such judicious intervention involves a first-time launcher that was exposed to investor overconfidence in the adoption of its first product and shared a range of ramp curves from analog products to set more realistic growth expectations.
Leaders who can execute on the right commercialization plan, make the optimal partnering decisions, secure adequate capital, and meet or exceed investor expectations will prosper through first-product launch. Short of an exceptional product profile that can offset setbacks in planning, execution or communication, these four strategic levers are essential to the success of biopharma companies launching a first product.
For more information, please contact lifesciences@lek.com.
Endnotes
1Includes two companies that filed for bankruptcy in the 24 months post-first product launch
2Excludes two companies for which share price data was unavailable from S&P Global Market Intelligence