-
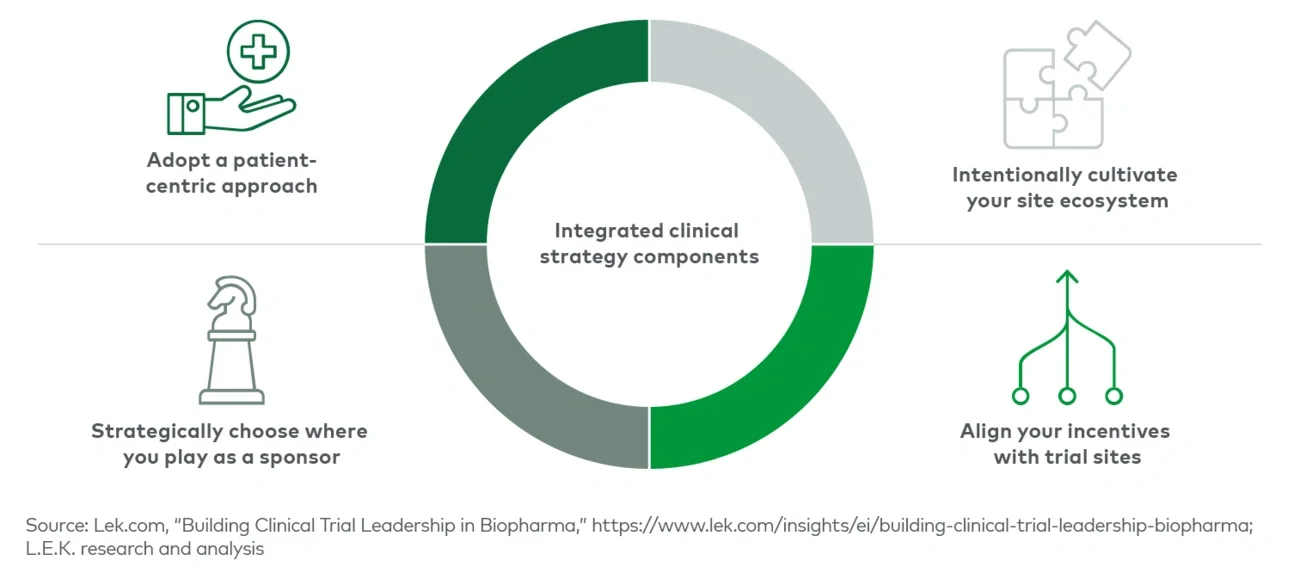
Put patients first — Sponsors need to own and prioritize ensuring potential, current and past patients are supported and engaged as the critical participants they are as trials evolve. Especially important to diversity efforts, sponsors should consider the unique needs and perspectives of underrepresented patient populations.
-
Intentionally cultivate your site ecosystem — Sponsors need to identify the correct sites to target, onboard and cultivate longitudinal relationships at both traditional and emerging site types in order to meet portfolio needs while also developing relationships with new sites and investigators.
-
Align incentives — Sponsors need to work to ensure that sites receive the proper operational, reimbursement and logistical support to serve as the face of the trial to patients and to conduct trials in a sustainable, positive way that meets the needs of their business.
-
Choose wisely — Sponsors need to proactively position themselves as the leader on trials and avoid information disintermediation by sites and/or third parties.
Additionally, achieving diversity action plan goals will require intentionality across these four components to address operational, technical and logistical challenges (e.g., transportation, translation services, administrative support for hospitals with limited resources), as minority patient populations are typically served by underfunded, research-naive hospitals.
As each trial’s and sponsor’s circumstances are unique, focusing on the four key components in an integrated approach can take several forms, for example:
-
Developing long-term relationships with patient populations through sites, patient support networks and physician outreach to drive identification of and engagement with relevant patient populations
-
Integrating patient recruitment solutions with site selection solutions and combining on-site and technical solutions to tailor recruitment to individual patient needs
-
Using tailored communication (e.g., email, mobile app and/or text) and milestone tracking to flag adherence issues before dropout to increase patient retention
-
Conducting decentralized clinical trials to provide patient-centered care in order to promote recruitment and retention
Clinical trial ecosystem players have begun to develop solutions across the four components
Various pharmaceutical services vendors and technology companies have begun to develop solutions to help mitigate the pain points noted above. Regarding the first key component of developing long-term relationships with patient populations by meeting them where they are, players including Javara are developing solutions to enable both experienced and trial-naive clinicians to participate in clinical trials by embedding the necessary clinical research staff and infrastructure into their practices.
Javara’s partnerships with broad community-level healthcare organizations enable it to reach a more diverse patient population, and its innovative communication modalities allow Javara to bring awareness to potential trial enrollees and engender trust. Other companies are forming long-term relationships with diverse patient populations for narrower indications; for example, the OneOncology Research Network uses its operational expertise to facilitate clinical trials in a network of community oncology practices that have diverse patient populations and may lack the necessary trial infrastructure.
The second key component, cultivating the site ecosystem through integrating patient recruitment with site selection and tailored recruitment solutions, is being addressed by players such as SubjectWell and 83bar, which deploy educational campaigns and the creation of high-intent, qualified patient recruitment platforms to accelerate trial recruitment and minimize dropout rates. Lack of awareness of the benefits of clinical trials and how to enroll prevents most patients from enrolling in trials, and the lack of effective patient screening can result in high dropout and screen fail rates. Solutions are evolving to address these pain points and improve trial cost and time efficiency.
Aligning sponsor and site incentives, the third key component, has the goal of driving outcomes such as higher patient retention. Clincierge, a pharmaceutical services vendor, helps biopharma sponsors coordinate patient travel logistics for clinical trial participation, which is a service that can improve patient retention as well as trial population diversity due to the alleviation of transportation barriers. Adoption of new strategic trial solutions is not limited to the U.S healthcare market. Our November 2022 report discussed China’s adoption of smarter clinical trial tools, in addition to other strategic trends driving high-quality clinical development growth throughout the country.
Players such as Greenphire have developed other solutions to minimize financial barriers to clinical trial participation. Greenphire developed the ClinCard solution that automates study participant compensation with debit cards, virtual cards and direct deposit. The solution also includes a tax toolkit for sponsors and integrates with ConneX, a platform for travel services for clinical trial participants. Travel and financial barriers are a major reason for patient dropout, but emerging solutions are beginning to address these concerns to maximize sponsor and site incentives.
The last key component, strategically choosing where to sponsor clinical trials, extends beyond a sponsor determining at which sites it can establish itself as a leader and is rapidly beginning to encompass elements of decentralized clinical trials. Science 37 provides technology-enabled solutions to allow certain elements of clinical trials to occur remotely. The platform facilitates virtual recruitment and enrollment, electronic informed consent, electronic sources, electronic clinical outcome assessments, and telemedicine, among other solutions, to reduce geographic constraints in clinical trials. Similar to previous solutions, technologies and services allowing distributed functions enable greater diversity within clinical trials due to the removal of transportation barriers.
Beyond pharmaceutical services vendors, companies in adjacent fields (e.g., biopharma, technology) are entering this space to develop solutions that address these same pain points through different pathways. For example, Roche has developed a clinical trial match application that can be utilized by oncologists to set up a new trial, which could help alleviate the pain point of the trial initiation process being a time-consuming one. Apple has developed ResearchKit and CareKit, which enable researchers to develop apps that facilitate the collection and aggregation of data on various medical conditions, making clinical trial operations more patient-centric and alleviating inequity with respect to clinical trial site accessibility.
While industry players have developed interesting solutions, sponsors that consider all four key components in an integrated clinical trial approach will have the greatest success in facilitating cost- and time-efficient trials with diverse patient populations.
Conclusion
While a number of point solutions to address patient identification, recruitment/enrollment and retention challenges exist in the market today, maximizing them will require a tailored strategy that is proactive and intentional toward and integrated with each of the four key components. Putting patients first, intentionally cultivating the site ecosystem, aligning incentives and choosing wisely where to sponsor trials will enable sponsors to achieve diverse and targeted populations in their clinical trials and to facilitate timely and cost-effective trials that will benefit all patient populations.
As the number of clinical trials being conducted continues to grow, therapies become more complex, the clinical trial landscape becomes more competitive and new legislation that promotes diversity within clinical trials is introduced, all four of the above components must be intertwined to create a longitudinal clinical trial network.
Putting patients first through cultivating long-term relationships, using tailored communication and implementing patient-centric monitoring will improve patient enrollment and retention, thereby reducing trial length and cost.
Intentionally cultivating the site ecosystem entails implementing a proactive approach to engage with existing and emerging trial sites to reduce the time necessary to initiate a new clinical trial. Additionally, experienced trial sites can be valuable resources when developing protocols for new trials.
Aligning incentives is critical to streamlining the clinical trial process at chosen sites and ensuring proper infrastructure is in place. And last, selecting wisely where to sponsor clinical trials ensures that sponsors are positioned as the medical and scientific leaders of trials and that data is handled in an efficient manner.
Integration of all four key components in an intentional and proactive manner drives solutions that achieve diverse trial populations in a cost- and time-efficient way, providing benefits to both diverse patient populations and trial sponsors.
The authors would like to thank Grace Mizuno (L.E.K. Senior Life Sciences Specialist) and Chris Rood (L.E.K. Life Sciences Specialist) for their contributions to this Executive Insights.
For more information, please contact lifesciences@lek.com.