Background
Japan has historically been a favorable geography for medical device launches. But increasingly frequent and significant price cuts and the detrimental effects of international reference pricing have put this traditional practice in jeopardy. Representatives of the medtech industry consulted with Japan’s Ministry of Health, Labour and Welfare (MHLW) regarding its vision for how the pricing and reimbursement system should evolve in Japan.
The client asked L.E.K. Consulting to help develop its position on what the future of pricing and reimbursement for medical devices should look like in Japan.
L.E.K.’s approach
L.E.K. conducted individual workshops with the client’s medtech business units and aggregated the findings to produce a high-level summary of the company’s vision for the healthcare industry in Japan; the challenges to reaching this vision; the role that the medtech industry can play in Japan to help achieve the vision; and the required government policy changes.
Through internal and external analysis, we assessed each proposed government policy change in terms of its business impact on the client as well as its feasibility and urgency, in order to identify which government policy changes were higher priority for the client to consider and address.
Value delivered to the client
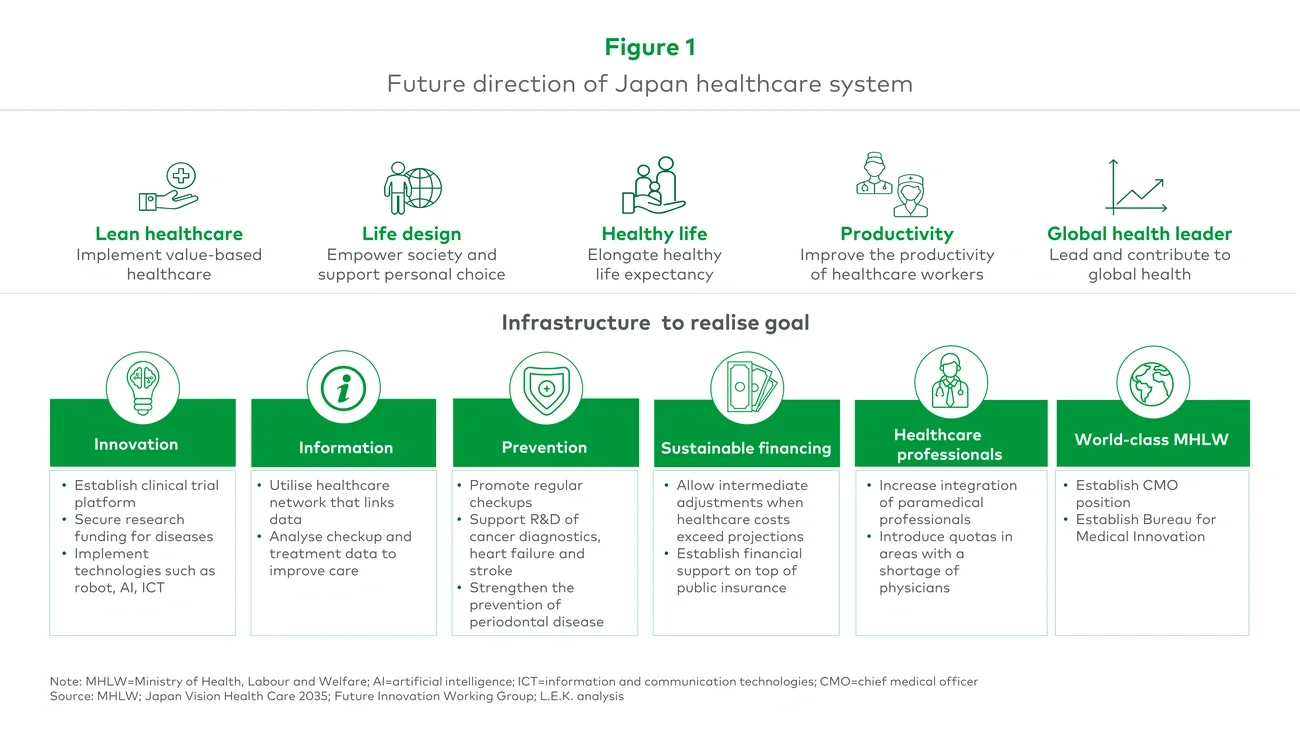
L.E.K. developed a policy paper that the client can provide to external stakeholders (e.g., industry associations or MHLW) to explain why proposed policy changes are necessary and important (see Figure 1).