India faces rising healthcare expenditure and a limited government healthcare budget, resulting in a predominantly out-of-pocket (OOP) payer landscape. In a predominantly low-to-middle-income country like India, unaffordable OOP costs constrain access. To reduce OOP payments, several state-level and nationwide government health plans have been implemented.
Unfortunately, the reimbursement evaluation criteria of most government health plans put too much weight on cost and too little on long-term outcomes, stemming from limited resources with evolving technical skills to perform health technology assessments (HTAs) and a system that underrepresents the private sector in the decision-making and evaluation process. This partial evaluation further results in low reimbursement prices and deters widespread rollout of government health plans into the private sector.
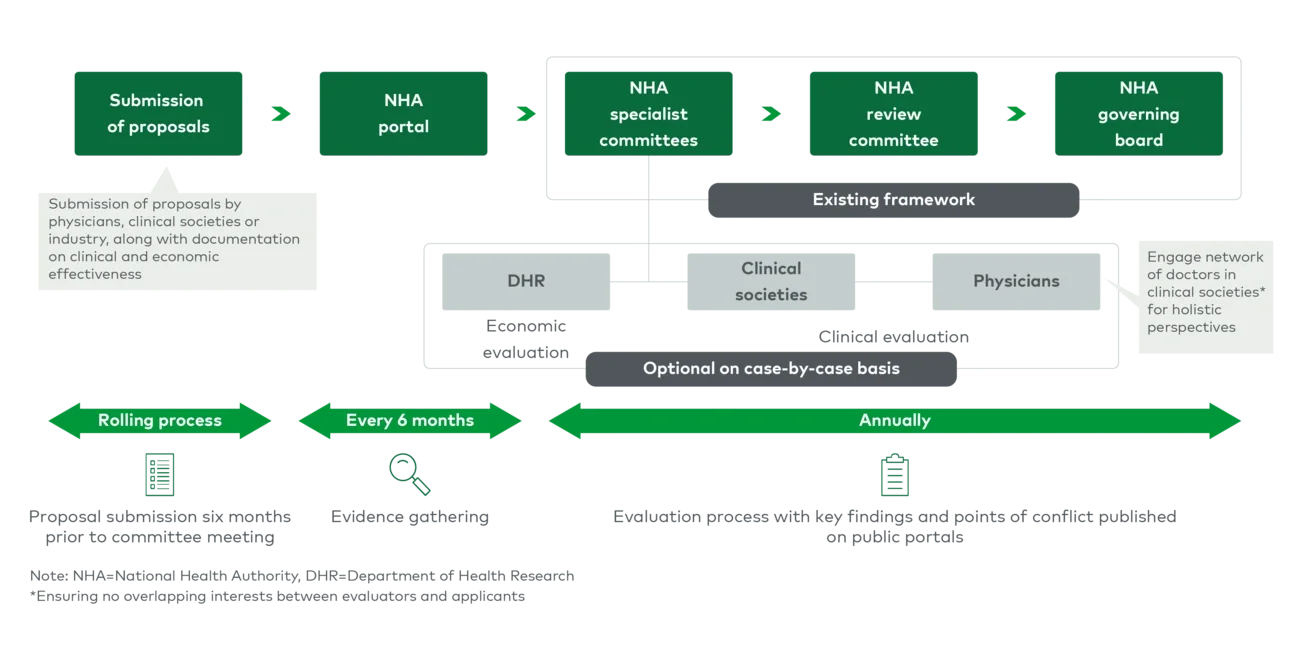
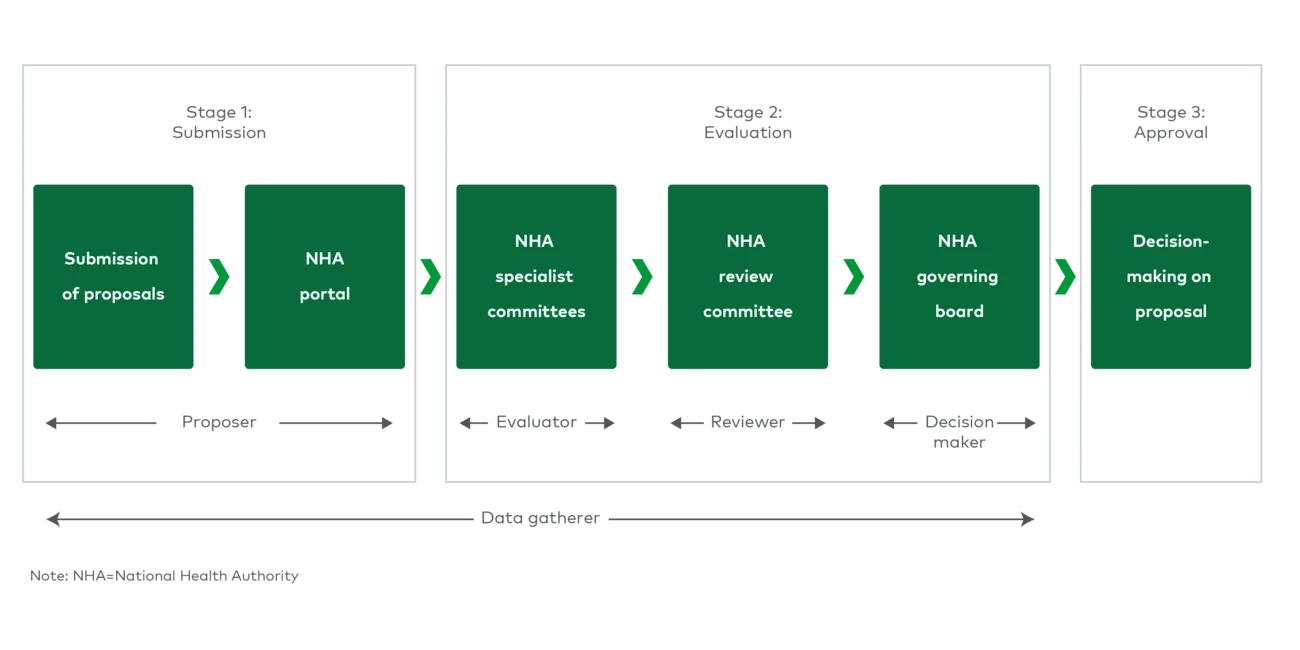
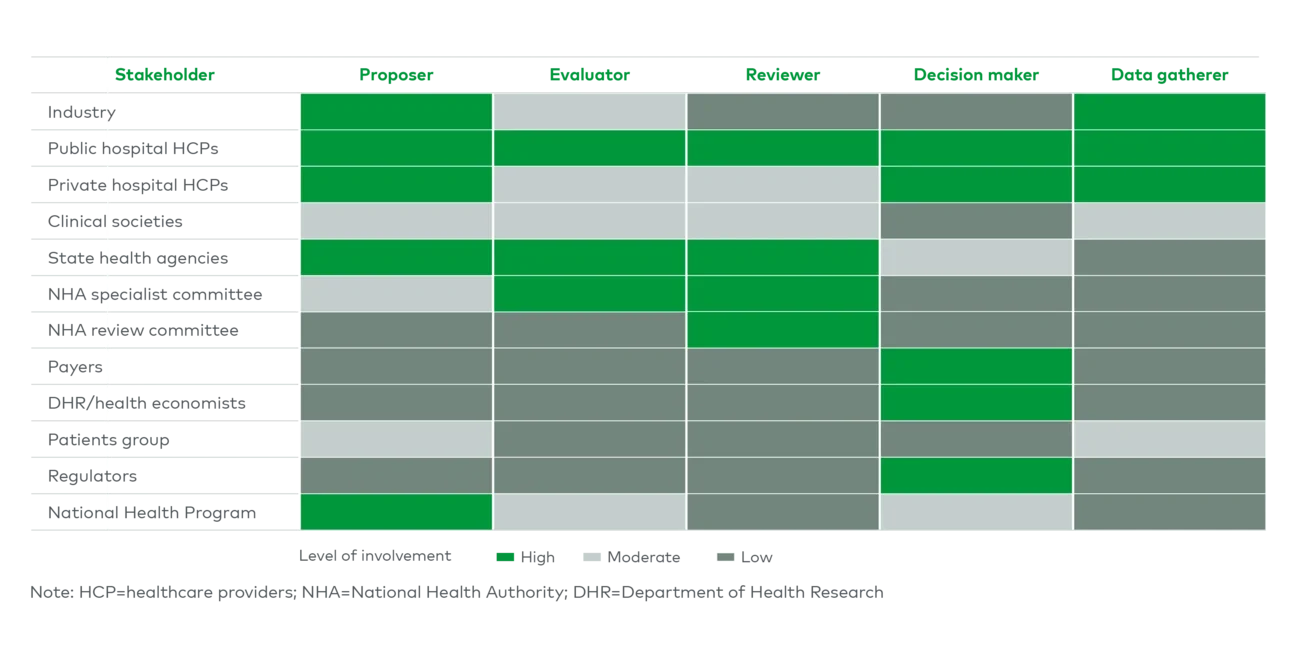
This calls for a more structured reimbursement system to select medical technologies that demonstrate clinical evidence (e.g., improved long-term outcomes) and in turn contribute to overall cost savings, reducing burden on public systems and payers. As such, changes to the current medical device reimbursement evaluation process have been proposed to address limitations.