Ultimately, clinical trial sponsors are trying to optimize their R&D investments, and so are concerned about risk as well as failure due to high costs and long timelines. Some of the leading drivers of operational risk within trials, such as recruitment and data quality and consistency, could be exacerbated in decentralized trials. To build credibility, distributed functions need to actively mitigate these risks and provide sponsors with the confidence that they will not be an issue. As more decentralized trials are completed, the industry should see additional proof points on data quality and uniformity, the validation of digital health technology for remote assessment, participant retention, data management, safety monitoring, the identification of trial conduct issues, and documentation and record-keeping.
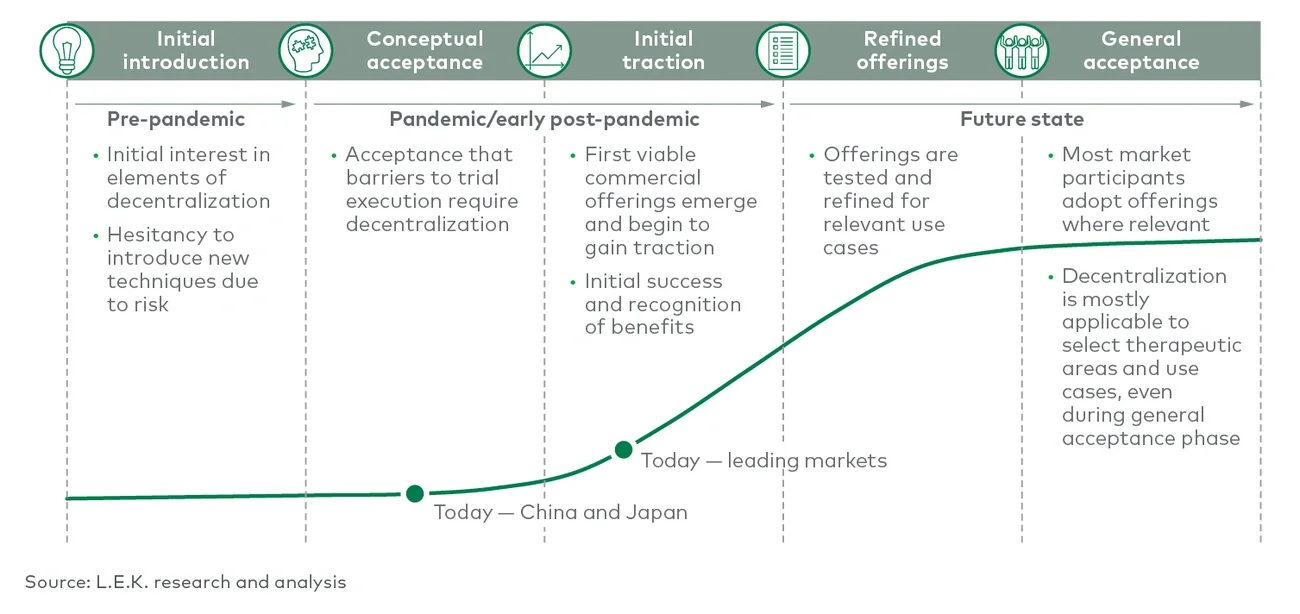
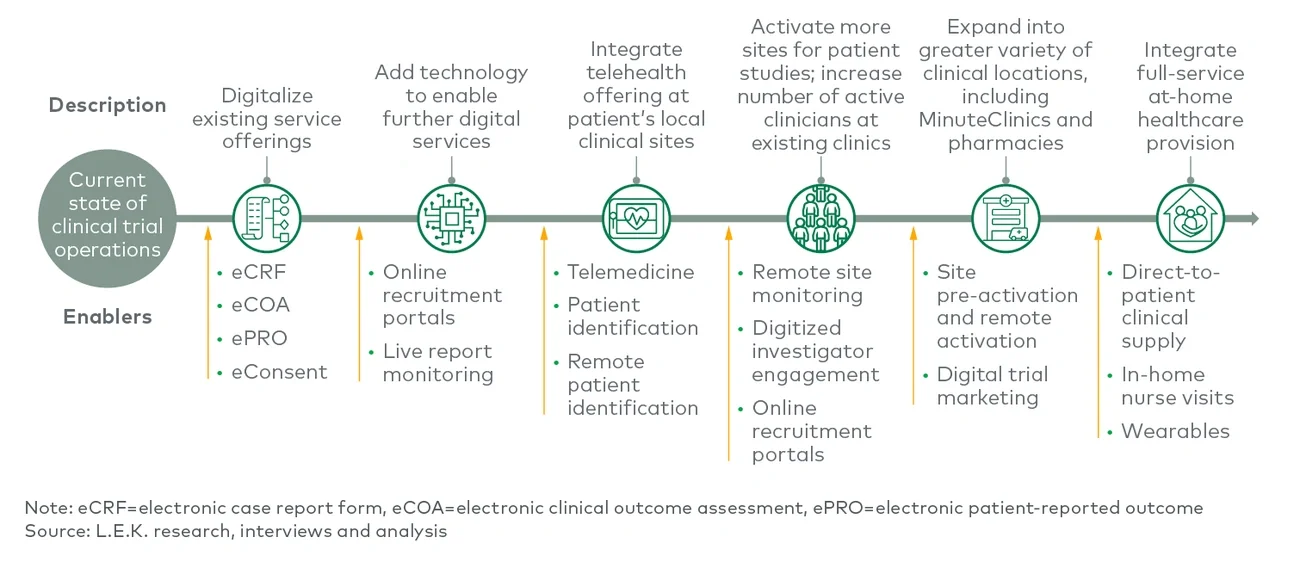
Decentralization is still in its early stages, but there is a growing belief that it will be a main feature in relevant future clinical trials. Stakeholders involved in clinical trials — including sites, sponsors, and service or technology providers — will need to understand how decentralization may impact them and be forward-thinking in preparing for this evolution in the market.
Maturation of data aggregation capabilities
Legacy data systems for clinical trials and other sources typically do not communicate well with one another, even though they are collecting similar or overlapping data points. As a result, aggregation techniques for integrating such data are often outdated, suboptimal, or even nonexistent, and biopharma companies are missing opportunities to generate value from their data. The benefits of data aggregation to pharma companies are twofold. First, it provides better insight into in-house data to drive increases in operational efficiency and better decision-making. Second, there is commercial value in the tremendous amounts of clinical, commercial, regulatory and operational data that both pharma companies and providers have amassed. This data could be useful for others in the healthcare value chain if it is aggregated, structured and synthesized. Accordingly, it could be monetized and serve as a new revenue stream. And so, data aggregation technologies in clinical and commercial settings are maturing to enable biopharma companies to realize these benefits.
In the clinical setting, trial sponsors will increasingly demand more tailored, insight-focused data solutions to reduce costs, decrease trial timelines and improve efficiency for use cases ranging from trial design optimization, patient identification and recruitment to site and investigator identification and selection and in-trial decision-making. To that end, service providers are increased aggregating real-world data from multiple sources — including electronic health records and claims — and integrating it with medical records in those trials.
Such databases and tools allow for alternative trial designs that incorporate historic or synthetic control arms as well as more efficient recruitment, including constructing more homogeneous trial arms and identifying hard-to-find patients. They also aid in the identification of sites with high volumes of patients that meet selection criteria. And critically, they facilitate just-in-time in-trial decision-making. In traditional trial operations, data analysis happens after the fact, so when execution fails, trials must be rerun, resulting in high costs. Now, data tools are emerging that analyze and visualize trial data from the cross-trial level to the individual patient level, enabling early decision-making. And a growing ecosystem of companies are gaining traction by providing such clinical data aggregation and/or analysis solutions for one or more of the above use cases, including Saama, eClinical Solutions and Clinerion.
There are also numerous use cases for data aggregation technologies that provide benefits in the commercial setting. These include sales analytics to help with launch planning, sales tracking and targeting, patient care coordination, the demonstration of real-world evidence and value for market access, and health economics outcomes research and product safety. Pharma companies use a range of data sources for these purposes, including claims, patient registries, patient hubs, electronic medical records, third-party transactional records, and inventory and chargeback efforts. Data analytics platforms that integrate these datasets and streamline analysis can help companies execute their launches more efficiently and effectively. These platforms are challenging to build and maintain in-house, so pharma companies are turning to service providers for solutions.
Pharma customers will be more proactive and forward looking with their data strategies and will seek out service providers that are able to support this more sophisticated analysis. Service providers, in order to win target accounts, will need to connect various datasets, apply deep analytical expertise and demonstrate their ability to translate data into insights using a compelling storytelling approach.
Increased need for global coordination of local requirements
The life sciences space is becoming increasingly globalized while local requirements continue to become more complex, creating a need for sophisticated coordination. For example, in 2011, the U.S. represented 26% of the worldwide total clinical trials, while in 2021, it represented 18% of the total5. Sponsors are also running global trials more often in order to increase the efficiency of enabling market access across regions and to recruit patients for increasingly rare indications. This need for coordination is exacerbated by the fact that local regulations vary from country to country and are constantly changing and becoming more complex. Coordinating clinical operations in a compliant manner on a global scale is incredibly difficult to execute.
In the regulatory and commercial setting, biopharma companies require global coordination of numerous functions, including pharmacovigilance, medical communications, regulatory affairs, and finance and distribution, particularly for advanced therapies. The complexity of compliance requirements is increasing, as are the consequences of failing to meet them. It is critical that companies seamlessly adhere to local regulations on a global scale, and they are increasingly turning to service providers to ensure that adherence.
In pharmacovigilance, companies need to aggregate safety signals globally through multiple channels — from email to phone to fax to social media — translate the reporting from local languages, standardize the reporting content and aggregate all of it on a single platform. Responding to product inquiries in a compliant manner involves similar challenges. As a result, local expertise and integrated services are among the most important selection criteria for pharmacovigilance and medical information providers.
This need for global-local agility and fluency is driving innovation in pharma services. It will also inform the next wave of winners in the space. Service providers are already rolling up international firms into consolidated entities to provide the global reach demanded by pharmaceutical companies. Beyond that, they will need to provide truly integrated oversight and visibility while demonstrating that they have boots-on-the-ground presence, knowledge and relationships across markets. Service providers should also build technology solutions to make data codified, centralized, compliant and accessible. These capabilities will increasingly become “table stakes” in the industry.
Growing tension between integration and specialization
All this change — in trial deployment, data aggregation and global coordination — has important implications for how service providers position themselves in the market. Service providers will feel the increased tension of scaling/aggregating services and developing/demonstrating therapeutic and scientific expertise in particular areas. This tension will further stratify the competitive landscape into scaled multiservice providers and niche specialists, a natural outcome of providers solving for distinct customer needs.
Multiservice providers can drive productivity via mitigated development and process risk, time savings, reduced complexity, cost savings, better results, stronger evidence, and improved compliance. To that end, contract research organizations (CROs) and other clinical and nonclinical service providers have made acquisitions to fill gaps in their portfolios by service line (e.g., safety testing, clinical enrollment and recruitment, clinical data management, project management, regulatory support, clinical study design, and biometrics/biostatistics) and/or therapeutic modality. Increasing their breadth of offerings can allow them to provide a more complete service suite to customers and, in the process, be more competitive.
WCG Clinical, a clinical services company, is an example of a multiservice provider that has created scale effectively. It has made over 30 acquisitions in its history, 15 of them in the past five years, to improve its clinical services offerings and provide its clients with a more comprehensive suite of services to address clinical trial pain points. Another example is Charles River Laboratories, which has invested more than $4 billion in over 25 acquisitions since 2012, $1.7 billion of which were made since the start of 2020. Most of these acquisitions have allowed the company to expand its nonclinical portfolio of research and discovery services, safety assessments, and clinical and commercial manufacturing. They have also allowed Charles River to bolster its expertise in certain modalities such as cell and gene therapy.
The contract development and manufacturing organization/contract manufacturing organization industry is not as concentrated as the CRO industry, but similar consolidation is expected as companies seek to expand geographically, bolster their capabilities in high-margin end markets and invest in platforms to provide a wider array of services. As more pharma services companies offer broader portfolios, there is an opportunity for niche players to offer better, more-specialized services than the aggregators. Companies focused on gene and cell therapies and personalized medicine, for example, need highly specialized offerings that reflect specific expertise. Niche specialists, some of which focus on specific therapeutic areas, are also more responsive to the needs of smaller biopharma companies.
In the central nervous system (CNS) space, Evolution Research Group, a clinical research site company, has focused capabilities within CNS studies, including psychiatry, neurology, pain and other areas. Clinical Ink is an electronic clinical outcomes assessment provider that has built a strong track record in lupus, which includes developing a data capture platform to streamline lupus trials.
Going forward, integration and niche specialization will continue to be cyclical. And as the next generation of best-in-class niche specialists emerge, they will become compelling acquisition targets for multiservice providers. The providers should be thoughtful in how they expand their services, as customers will ultimately continue to choose those that are best of breed. They will need to understand where they have a “right to win” based on their starting capabilities and assets. They will also need to ensure areas of expansion have their customers in mind, avoid acquiring suboptimal service providers and provide superior coordination to deliver on the value of a “one-stop shop” value proposition.
The next wave of consolidation will likely involve structuring and scaling around unique expertise. This could include, for example, building a best-in-class, end-to-end platform for a specific therapeutic area (TA) rather than expanding agnostic of TA, modality or specialization. Indeed, to deliver winning value propositions to their clients, integrated service providers will need to be extremely thoughtful about their service offerings.