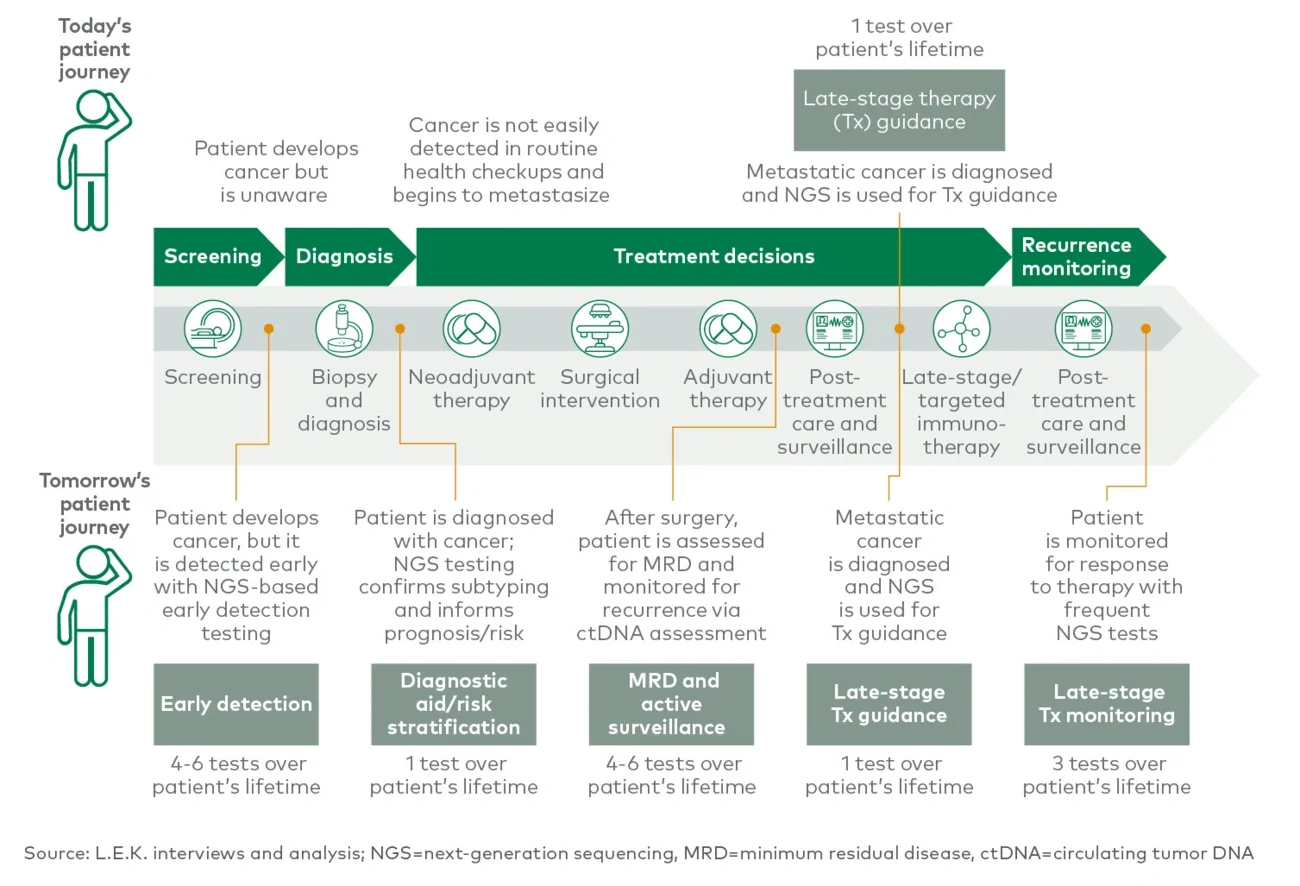
One new application with significant progress in the past few years is minimum residual disease (MRD) monitoring, where ultrasensitive liquid biopsy tests can enable more effective surveillance and earlier detection of recurrence than can existing methods like carcinoembryonic antigen tests or imaging. Adaptive Biotechnologies is a leader in the space for hematological cancers. Natera and Guardant are the most clinically advanced in the solid tumor space, having commercially launched laboratory-developed MRD liquid biopsy tests that measure circulating tumor DNA in 2019 and 2021, respectively. The initial focus for these tests is recurrence monitoring and risk stratification in colorectal cancers, though Natera’s liquid biopsy is now covered by the Centers for Medicare & Medicaid Services for pan-cancer immunotherapy monitoring, and Guardant is also planning to include additional tumor types. Additionally, Invitae offers early access to its MRD liquid biopsy, and NeoGenomics Laboratories is preparing to commercially launch an MRD assay this year. This application is valuable to biopharma companies developing treatments for the adjuvant setting. These technologies are still early in their adoption curve but are expected to see increased uptake, broader reimbursement and expansion into other tumor types.
Early detection is an even more nascent application of liquid biopsy that has long been considered the holy grail of cancer screening. Two FDA-approved liquid biopsy early detection tests are Exact Sciences Laboratories’ stool-based Cologuard and Epigenomics’ blood-based Epi proColon, both for colorectal cancer (CRC). In 2021, GRAIL launched Galleri, the first multicancer early detection (MCED) test commercially available in the United States as a laboratory-developed test. The test sequences cell-free DNA in the blood to detect more than 50 cancer types. The company is planning to pursue full FDA approval for the test in 2023. Other companies developing early detection tests include Exact Sciences, which is developing a CRC blood screening test and a multiomic MCED test analyzing DNA and proteins; Guardant, which is currently focusing on CRC but planning to expand to other tumor types; Freenome; Bluestar Genomics; and Singlera Genomics. Early detection tests are an incredible achievement, with the potential to have significant impact on patients, especially since the majority of cancer deaths occur in cancers without recommended screening tests. Sufficient sensitivity for early-stage cancers and broad reimbursement are two key requirements for realizing the full potential of these tests.
Research in liquid biopsy applications beyond oncology is also increasing. Alzheimer’s disease, for example, needs an inexpensive and scalable diagnostic to replace positron emission tomography (PET) scanning and allow for earlier identification of patients, even in pre-symptomatic stages. In 2020, C2N Diagnostics launched a blood test for levels of amyloid beta that could enable this. Nonalcoholic steatohepatitis (NASH) is another disease with a need for a less invasive, inexpensive test to replace liver biopsy and other less-specific blood test methods to screen for, triage, diagnose and/or monitor disease. Eighty million Americans are estimated to have nonalcoholic fatty liver disease, many of them undiagnosed, and of those, 10%-30% may have NASH. Last year, Glympse Bio and DiscernDx both reported early proof-of-concept results for blood-based liquid biopsy tests for NASH. These tests, if fully validated, could drastically increase the pool of diagnosed patients appropriate for disease-modifying treatments in development.
For liquid biopsy to move into early screening and detection in oncology and beyond at a wide scale, it will need to meet key requirements including test performance, pull through to confirmed diagnoses, and reimbursement. Minimizing false negatives and false positives will be important for driving widespread adoption. In oncology, physicians need to be able to reliably find the tumor, following a positive liquid biopsy screen, for the tests to be consistently useful. Then, a key question will be whether the healthcare system will be able to afford the earlier and more frequent testing. For example, the total addressable market for pan-cancer screening in the U.S. could be ~$20 billion, assuming all ~120 million Americans aged 50 or older receive a test at ~$500 per test every three years. Cologuard is currently reimbursed by Medicare at ~$500, but Galleri’s list price is nearly twice as high at $949. It is not yet clear what the screening frequency will be, what price the market would accept and whether NGS-based assays would be profitable at that price given the inherent costs. It will be important to monitor the market’s progress on the above requirements.
Accelerated consumerization of diagnostics
Consumers are increasingly interested in taking their health into their own hands, and diagnostic tests are no exception. The COVID-19 pandemic has made diagnostics a more common part of everyday life and has popularized at-home testing as an alternative to less convenient in-lab testing. Now, consumers are “shopping” for diagnostic tests that are accurate, convenient, fast and affordable. At-home diagnostics may benefit the healthcare system as well, by reducing the number of in-person visits for routine tests and lowering the cost of care.
At-home sample collection is a common direct-to-consumer model outside of COVID-19 self-tests. In this model, consumers order tests, send samples to Clinical Laboratory Improvement Amendments-certified labs and then receive results, sometimes reviewed by physicians. Companies like LetsGetChecked, Everlywell, Verisana Laboratories and myLAB Box follow this model, offering menus of elective tests for (to various degrees) vitamin levels, thyroid levels, cholesterol levels, sexually transmitted diseases, fertility and more. Many of these companies were founded several years before the pandemic. However, investment in the space has been significant in the past two years — Everlywell raised a $175 million Series D round in 2020, and LetsGetChecked raised a $150 million Series D round in 2021.
Leading diagnostic companies and laboratories like Abbott Labs, Quidel, Quest Diagnostics and Becton Dickinson have also made significant investments in new channels for diagnostic tests. Abbott Labs and Quidel are looking to expand in direct-to-consumer diagnostics, Quest Diagnostics is offering consumer-initiated lab testing in partnership with Walmart, and Becton Dickinson acquired Scanwell Health, which has a smartphone app to analyze and interpret test results. The market will likely experience the most growth in respiratory diseases, sexually transmitted diseases and colon cancer screening.
Several challenges with the at-home testing market will need to be addressed, as these tests shift some responsibility to the consumer for selecting the right test, conducting the test properly, paying for the test and, in some cases, interpreting the result correctly. It can be difficult for consumers to assess or compare the reliability of different tests. Many of these tests are not covered by insurance, which will be a barrier to widespread adoption. Interpretation and counseling support will also be critical. As the market for more-complex at-home tests like cancer screening and organ function tests grows, patients will be discovering consequential health information at home and need support interpreting the results and determining next steps. These tests are often screening tests and not diagnostic tests, and patients need to understand the possibility of a false positive. Consumers, insurers, regulators, physicians and test providers will all need to confront these challenges as the market matures.
Conclusion
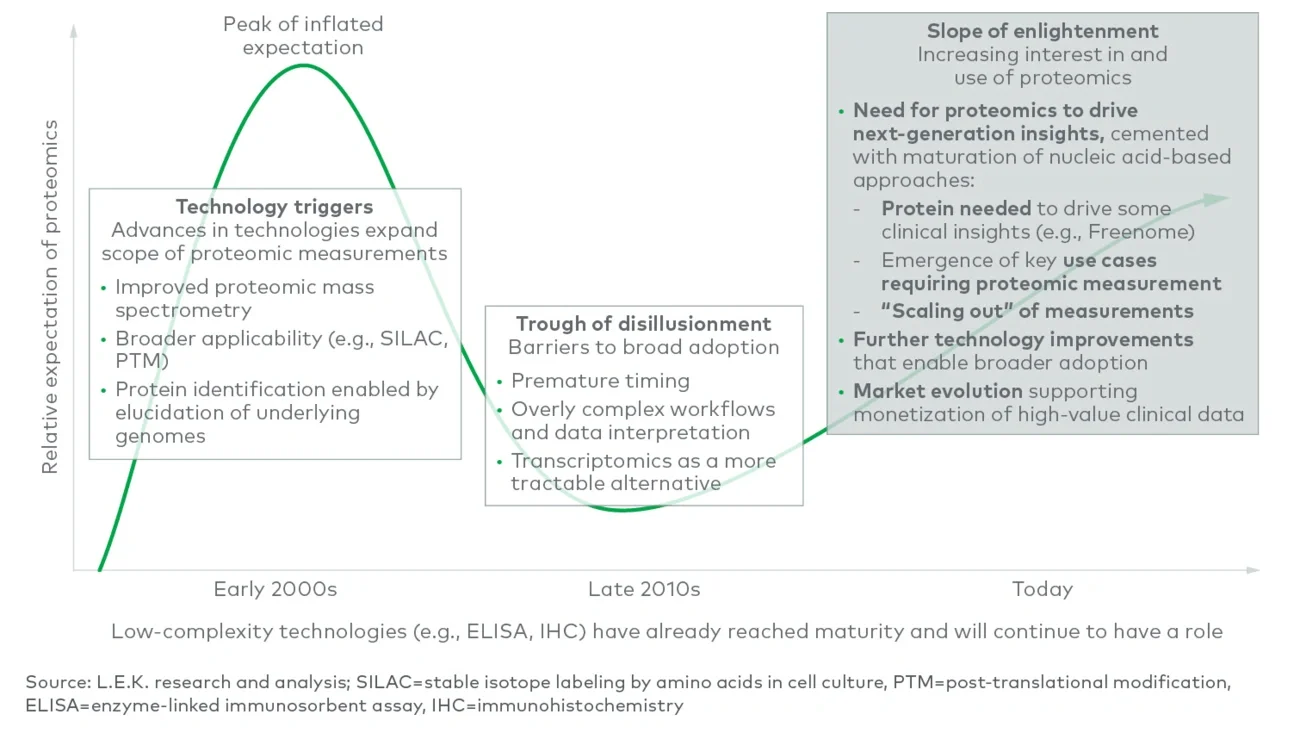
The diagnostics and research tools fields are expanding in new directions. New research tool technologies like proteomics are increasing scientists’ understanding of human biology and disease. New diagnostic tools like liquid biopsy are making possible earlier detection and more targeted treatment of some of the most serious diseases. And new business models are bringing diagnostic tests to patients at home. These developments have the potential to transform the industry and have ramifications across the value chain, touching patients, providers, insurers, scientists, laboratorians and the biopharma industry. Diagnostics and research tools companies should consider their participation in these emerging fields as they build winning strategies.
Endnotes
1Advancements are also occurring in the bioprocessing industry, but these will be discussed in a subsequent “Looking Ahead” Executive Insights