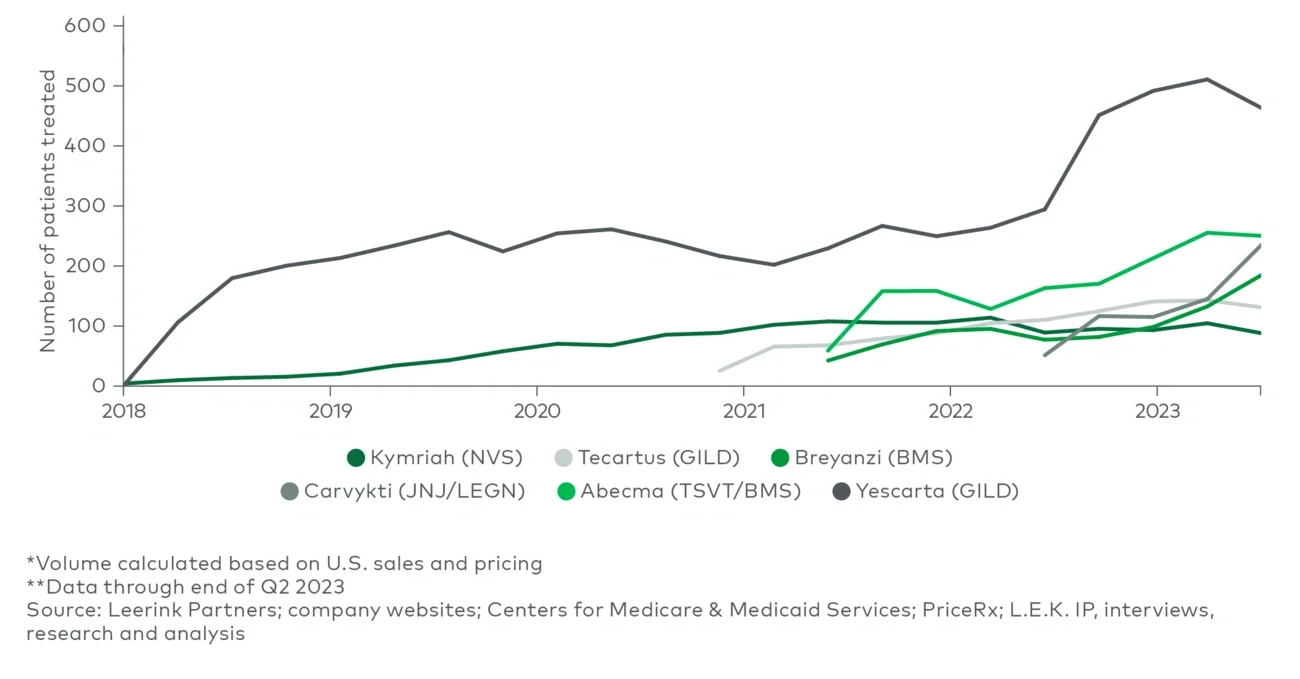
- Minimize off-target effects: Cytokine release syndrome (CRS) is among the most frequent adverse events of cell therapies. The current generation of commercial cell therapies target a single antigen with a somewhat heterogenous drug product (e.g., variations in T-cell potency and persistence), leading to an immune activation cascade and dose-limiting toxicology. Currently, immunosuppressants such as tocilizumab may be administered to treat CRS symptoms while the next generation of cell therapies (e.g., novel cell types, targeting approaches to improve specificity) aim to be designed to minimize the occurrence of CRS. In fact, more recently approved drugs (e.g., Breyanzi) typically exhibit lower CRS prevalence rates than older CAR-T therapies (e.g., Kymriah and Yescarta).
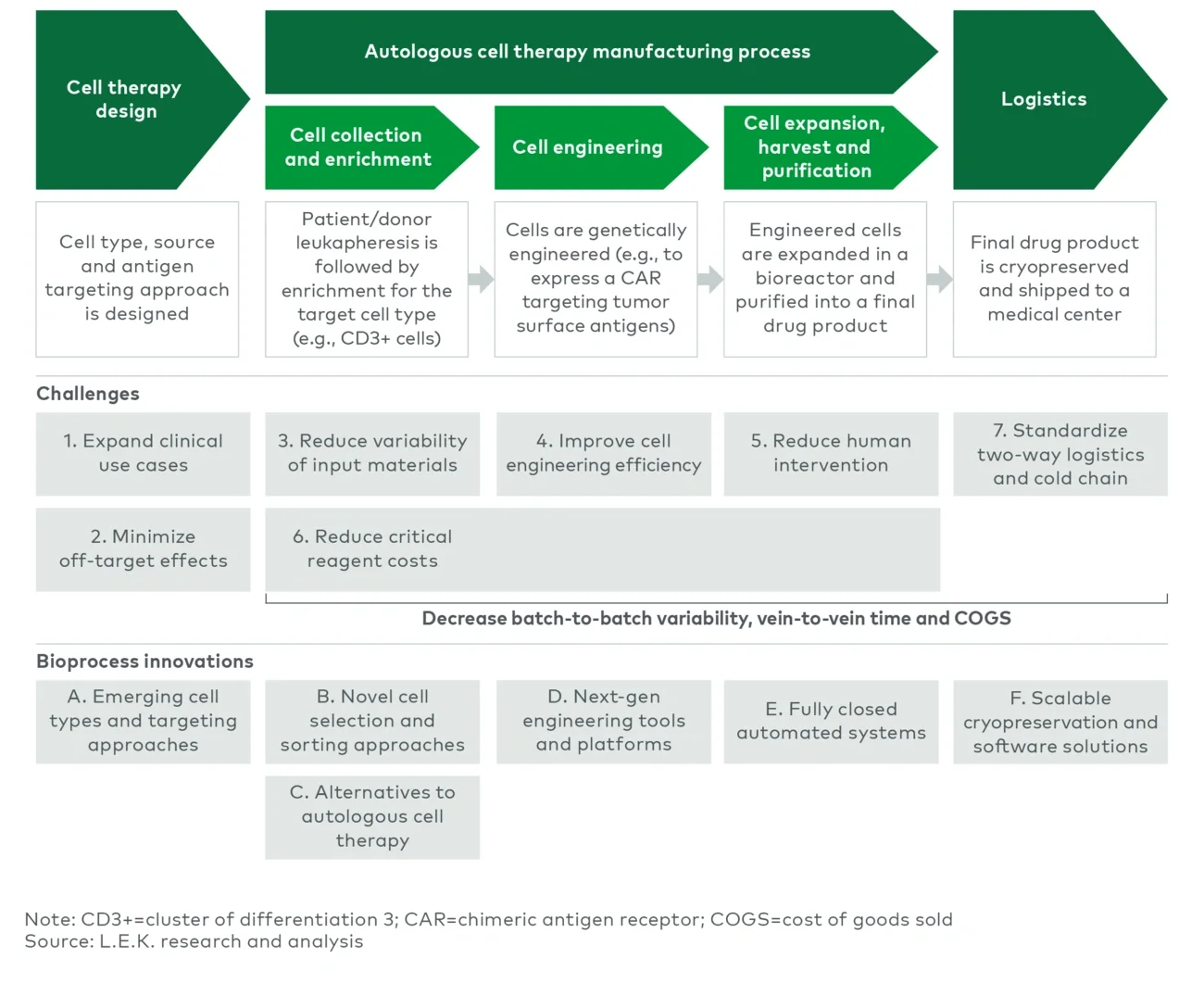
Challenges in the manufacturing process
Reductions in vein-to-vein time, processing costs and batch-to-batch variability are the overarching challenges inherent to personalized cell therapies. Currently, commercial autologous cell therapies have a vein-to-vein time of three to five weeks, driven by transportation of patient cells, lengthy manufacturing processes with batch failure rates that can exceed 10% and coordination of delivery for the final drug product. In some instances, this time delay may require bridging treatments that could lead to increased toxicity and complications or compromised efficacy of the eventual cell therapy.
Manufacturing is a key driver of vein-to-vein time, a fact that underscores the need to both shorten the overall manufacturing timeline and reduce batch failure rates attributed to human error, contamination and batch-to-batch variability. Not only are batch failures costly from the financial and patient health perspectives, but manufacturing production issues could potentially delay the launch of a therapy, as seen with the U.K. launch of Johnson & Johnson and Legend Biotech’s Carvykti. For commercial CAR-T products, it will likely take several years post-launch to improve the manufacturing process and reduce failure rates given that process development is typically not conducted with disease-state material.
- Reduce variability of input materials: Due to the nature of autologous cell therapies, patient health, degree of pretreatment, associated lymphocyte levels and limited quantity (i.e., apheresis yield) can cause significant variability in critical input materials. Additionally, differences across apheresis facilities (e.g., collection process and device, freezing equipment, experience of personnel) leave manufacturers vulnerable to inconsistencies in input material, which may cause unwanted batch-to-batch variability downstream. Organizations such as the Association for the Advancement of Blood and Biotherapies and the Foundation for the Accreditation of Cellular Therapy are starting to create standards across the workflow (e.g., apheresis, final product labeling), and it is imperative to learn from post-commercial optimization of first-generation cell therapies. These efforts will be critical to future success as cell therapies are used to treat larger addressable patient populations, which may require additional apheresis or manufacturing facilities in a broader geographic dispersion.
- Improve cell engineering efficiency: Today, engineered T cells are typically produced by viral-based methods, which use lentivirus (e.g., Kymriah) or gammaretrovirus (e.g., Yescarta) to insert the CAR into the target cells. Viral transduction is used because commonplace, low-cost chemical transfection methods such as those used for cell line transfection in viral vector manufacturing (e.g., polyethylenimine, lipofectamine) typically demonstrate poor transfection efficiency in immune cells. Advances in viral approaches (e.g., Penn Medicine’s 24-hour preclinical CAR-T manufacturing process) have shown promise in accelerating cell engineering, which can have significant downstream benefits on cell therapy efficacy (i.e., reduced need for expansion may lead to higher-quality engineered cells). However, viral vectors remain a significant driver of COGS and a complicating factor for supply chains. For example, lentivirus can account for 10%-25%7 of the total batch costs due to expensive plasmid DNA, high variability and low yield. Furthermore, limitations on lentivirus supply can bottleneck the production of a drug product, as observed with Bristol Myers Squibb’s lentiviral virus supply challenges for the manufacturing of Breyanzi.
Nonviral cell engineering methods have emerged as alternatives to viral transduction. For example, instrumentation-based transfection (e.g., electroporation) has emerged as a viable alternative but still faces several key challenges, including low cell viability due to the stress induced on cells (which is particularly problematic due to the patient-centric nature of each batch and limited number of input cells per leukopak), expansion times (e.g., cell doubling times) and scalability concerns (electroporation requires an additional unit operation before cell expansion). - Reduce human intervention: Current cell therapy manufacturing involves a greater level of human input relative to mature manufacturing workflows, such as those for monoclonal antibodies. Existing cell therapy equipment follows a modular approach that focuses primarily on either a singular unit operation (e.g., transduction/transfection, cell activation, cell expansion, fill/finish) or a small subset of unit operations that require trained workers to manually transfer the batch from step to step, driving up labor costs. There is an ongoing shift toward closed, fit-for-purpose, single-use systems to streamline process development and manufacturing while limiting the potential for contamination issues (e.g., the U.S. Food & Drug Administration (FDA) recently issued a Form 483 to Novartis for Kymriah manufacturing process issues). Today, Cytiva’s Xuri is the primary system used for cell expansion within commercial scale processes, but it may be overtaken by automated systems that integrate multiple steps (e.g., selection, activation, expansion), such as the widely adopted Miltenyi Prodigy or Lonza’s fully closed, automated Cocoon system that is gaining traction in the market.
While these solutions seek to simplify the process, labor is still a key driver of manufacturing cost, accounting for 25%-50%6,7 of the overall cost per batch depending on the development stage, the modularity of the equipment and the level of automation incorporated into the process. Modular equipment also creates connection points that require manual manipulation, increasing the risk of contamination or human error. Additionally, the current generation of processing equipment is optimized for CAR-T cells and typically lacks flexibility for optimization for other cell types (e.g., natural killer (NK) cells may require larger volumes during cell expansion). - Reduce critical reagent costs: The requirement to homogenize critical material (e.g., CD4 or CD8 selection) for autologous cell therapy creates additional workflow steps (e.g., cell activation) that have cost and time implications.
T-cell activation is critical to cell therapy, with the most prevalent cell-sorting methods utilizing magnetic activated cell sorting (MACS) or fluorescence activated cell sorting (FACS) labels to identify cells. Magnetic beads (e.g., Miltenyi’s MACS, Thermo Fisher’s Dynabeads) are the preferred option for late-stage and commercial cell sorting and selection today but are a significant driver of costs at >$5,0007 per selection (5%-10%7 of the overall per-batch costs). This can be greatly magnified if multiple markers are necessary, which would require sequential steps and associated beads. Additionally, these beads may require additional (typically manual) workflow steps to reduce residuals down to an accepted clinical quality attribute level, which incur additional time and costs. FACS is an alternative to magnetic beads that offers a greater degree of freedom to separate cells based on surface markers, but limited throughput, few good manufacturing practice (GMP) options and a significant full-time equivalent commitment from experienced personnel restrict the adoption of FACS for bulk isolation at scale.
Beyond beads, immune cells are highly reliant on specialized consumables (e.g., cytokines) and GMP media, which are significant drivers of COGS, particularly for early-stage development where some level of customization may be required. This customization drives significant cost, as achieving economies of scale on the supplier side is challenging due to low volumetric demand of consumables required at this stage of development. - Standardize two-way logistics and cold chain: Autologous cell therapy manufacturing deviates from the traditional centralized manufacturing model in that patient cells must be collected from a medical center (e.g., apheresis facility), transported to a centralized manufacturing facility, processed into a drug product and then shipped to the patient’s bedside. Pilot-scale processes were rapidly adopted to meet commercial demand, resulting in cryopreservation, chain of identity (COI) and chain of custody (COC) solutions that are not optimized for large-scale production.
Currently, most cryopreservation processes involve liquid nitrogen (LN2), which struggles to maintain temperature uniformity when freezing and has challenges with scalability and sustainability (e.g., bulk storage and shipment of LN2). These challenges will likely be further magnified as manufacturing demand increases. LN2 freezing also poses a contamination risk due to off-gassing, which prevents LN2 controlled-rate freezers from being used in GMP suites. Transportation outside of the suite may cause prolonged exposure to cell-freezing media, such as dimethyl sulfoxide, prior to freezing. This could impact product quality as cell losses from biochemical toxicity and osmotic stress are correlated with pre- and post-freeze time of exposure.
The process for COI and COC has not yet been standardized, creating gaps in the value chain as stakeholders continue to transition to electronic systems and electronic batch records. This has caused the audit trail to be captured with a mix of paper documentation and multiple software solutions, resulting in challenges with software incompatibility between different stakeholders (e.g., developers, contract development and manufacturing organizations (CDMOs), medical centers) and regulatory differences across regions (e.g., stricter data residence rules in France).
Bioprocessing innovations
L.E.K. has identified six ongoing innovations with the most potential to address the aforementioned challenges and improve the cell therapy bioprocessing workflow:
- Emerging cell types and targeting approaches
- Novel cell selection and sorting approaches
- Alternatives to autologous cell therapy
- Next-generation engineering tools and platforms
- Fully closed automated systems
- Scalable cryopreservation and software solutions
- Emerging cell types and targeting approaches: To advance beyond liquid tumors, key innovations — including the use of novel cell types — targeting approaches and mechanisms to improve cell therapy persistence are being investigated. An exhaustive discussion of these ongoing scientific efforts is beyond the scope of this report; however, we highlight below key focus areas within autologous cell therapy.
Currently, engineered T cells are the most well understood, but additional immune cell types (e.g., NK, tumor-infiltrating lymphocyte (TIL), dendritic, macrophage) are being investigated for the potential to increase clinical efficacy and/or safety. For example, NK cells show promise in lowering toxicity and infiltrating solid tumors but still need to overcome challenges related to durability, while TIL cells are specific to a patient’s tumor, allowing for high specificity of a targeted antigen, and can be expanded in high volumes allowing for potential redosing of patients. Critically, cell therapies with novel cell types will convey different bioprocess requirements (e.g., culture density, reagents, culture days, activation process) than today’s T cell-derived therapies.
In addition to novel cell types, new approaches like dual targeting may help address the heterogeneity of solid tumors, while leveraging alternative receptors (e.g., T-cell receptor) facilitates a broader selection of targetable antigens (i.e., through improvements to tumor cell detection and killing due to increased sensitivity and coupling with physiologic signaling). Additionally, other mechanisms are being explored to better infiltrate TME (e.g., armored cells) and evade unwanted immune responses (e.g., cloaked cells) to target difficult tumors and/or increase persistence and potency.
These innovations can unlock the full therapeutic potential of cell therapy but may also introduce new bioprocessing challenges or variations (e.g., NK and TIL cells have higher cell expansion volumes, rendering current equipment nonoptimal) of those associated with first-wave CAR-T therapies. - Novel cell selection and sorting approaches: Cell sorting can be accomplished using approaches that either use a label (e.g., MACS, FACS) or are label-free. New label-based technologies (e.g., Cellular Highway’s vortex-actuated cell sorting, Akadeum’s buoyancy-activated cell sorting) were developed to overcome the challenges related to cost, throughput and cell loss associated with conventional MACS and FACS methods, but validation at scale has yet to be confirmed.
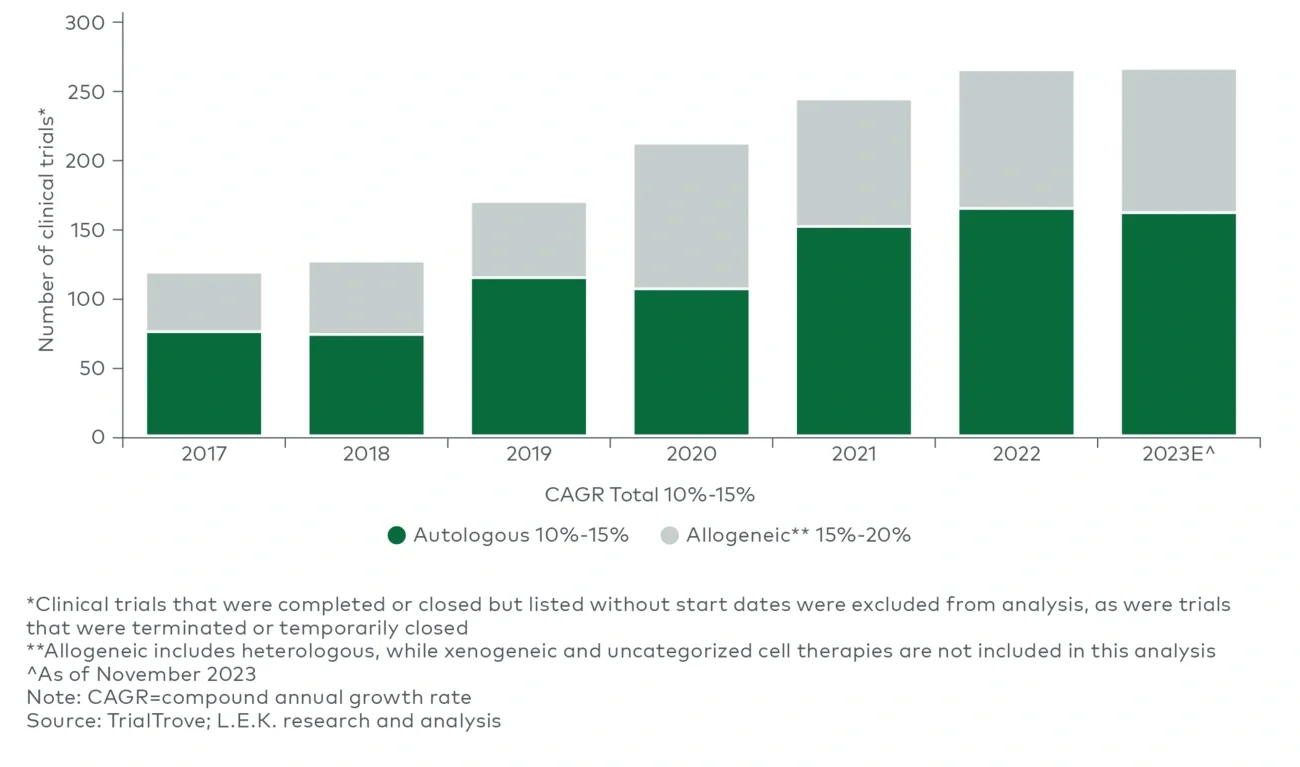
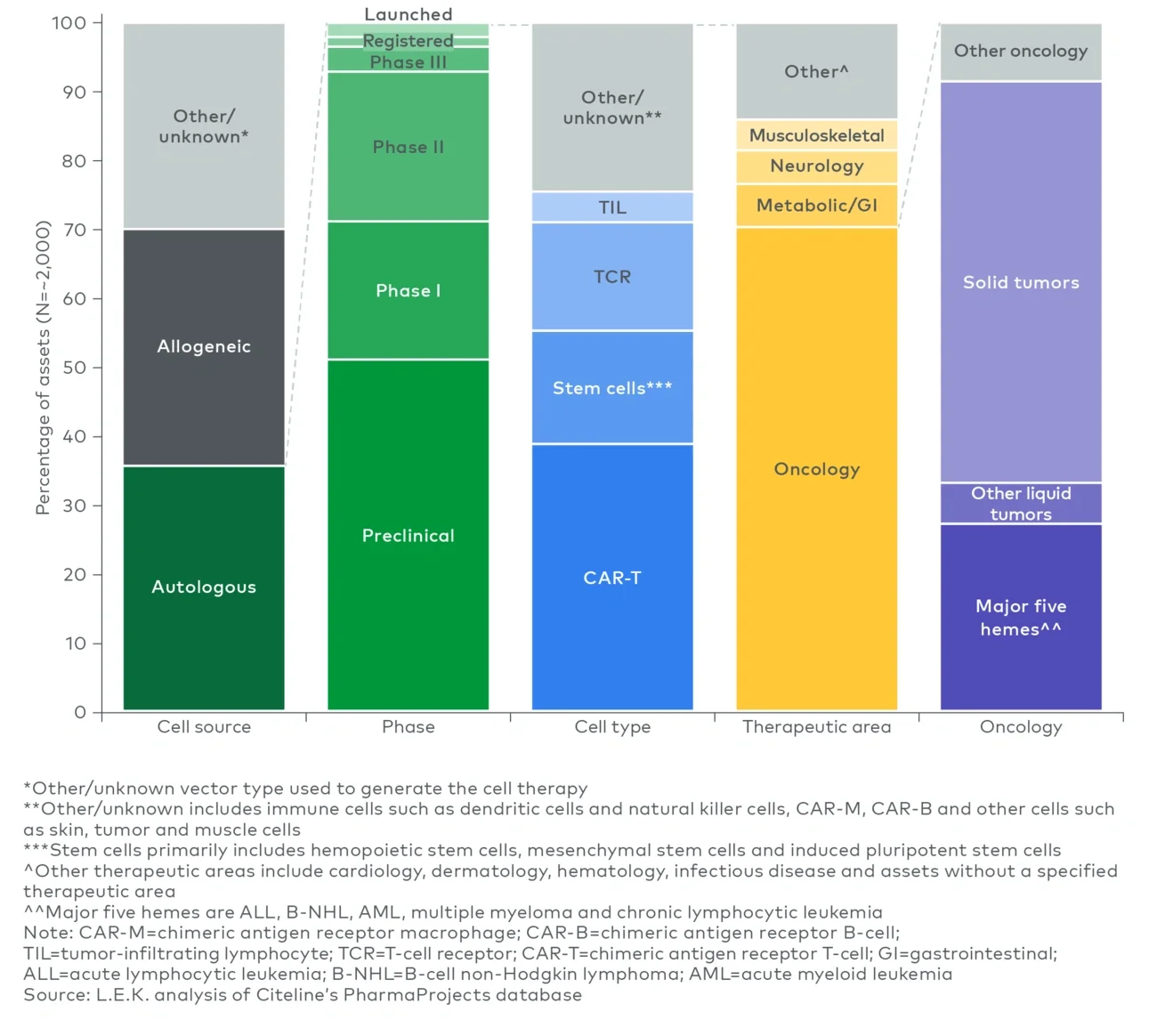
Novel label-free methods have emerged as intriguing alternatives for both bulk isolation and finer selection to avoid unwanted cell activation, remove the risk of reagent contamination of the final drug product (e.g., beads), and potentially expand the number and diversity of selection markers that can be used without linear growth in COGS (e.g., use of two selection markers doubling batch spend attributed to selection beads). Among the emerging approaches is cell separation based on physical properties (e.g., size, density, compressibility) using either ultrasonic waves (acoustofluidics) exemplified by Aenitis, Draper and FloDesign Sonics or microfluidics by companies such as Curate Bioscience and Cytonome. While primarily focused on R&D, ThinkCyte is using in silico methods to apply a nonphysical “label” in combination with flow cytometry to sort cells based on phenotypes (e.g., activated versus rested) which, if viable, could pave the way for other in silico approaches for bulk isolation at scale. - Alternatives to autologous cell therapy: While there is a significant focus within the industry on autologous cell therapies (35%-40%3 of the cell therapy clinical pipeline), other emerging approaches (e.g., allogeneic, in vivo) aim to circumvent many of the bioprocessing challenges for autologous ex vivo engineered cell therapy. Should these approaches succeed, these alternative therapies will likely coexist with autologous cell therapy, given the impressive efficacy and durability observed with autologous therapies, rather than replace the autologous modality entirely. While the manufacturing process will differ greatly (e.g., allogeneic cell therapy following the scale-up approach of conventional biologics), these approaches carry an entirely different set of challenges to address, such as cell donor management, transplant cell persistence and immune rejection for allogeneic therapies, or cell-specific transfection and risk of off-target editing for in vivo approaches. Furthermore, processes supporting allogeneic cell therapies currently lack the tools and fit-for-purpose manufacturing processes developed for autologous T-cell therapies today; they will require distinct manufacturing workflows, infrastructure and process solutions.
- Next-generation engineering tools and platforms: Moving beyond viral-based and legacy electroporation transfection methods, the next generation of transfection platforms aims to reduce manufacturing cost (e.g., eliminating the need for expensive lentivirus), increase compatibility with a broader suite of cell engineering approaches (e.g., CRISPR, transposases), improve commercial scalability and enhance post-editing viability.
Flow electroporation platforms, including those from MaxCyte, Kytopen and Draper, represent an evolution of traditional electroporation and aim to reduce processing time and minimize scalability and cell viability concerns. The recent approval of Vertex’s sickle cell gene therapy exa-cel represents a key regulatory milestone for MaxCyte’s ExPERT platform, as well as establishes precedent for FDA acceptance of nonviral cell engineering alternatives within commercial processes. Other players, such as CellFE, Avectas and SQZ Biotech, utilize a microfluidic-based mechanical approach (e.g., rapid cell compression/expansion). This approach reduces the consumables required (e.g., buffers) and may increase transfection efficiency as well as improve cell recovery/viability. Consequently, it could unlock the therapeutic potential of more fragile and harder-to-engineer cells (e.g., NK cells) as well as reduce expansion time and ultimately vein-to-vein time. - Fully closed automated systems: The first generation of cell therapy manufacturing relied on disjointed equipment that performed singular unit operations, requiring an operator to transfer materials from unit to unit. The industry is increasingly integrating flow path connectivity and automation for key unit operations through modular product ecosystems (e.g., Thermo Fisher’s Cell Therapy Systems product line), but true stand-alone, fully closed automated systems are still somewhat rare today.
The next wave of cell therapy bioprocessing equipment aspires to enable centralized parallel processing (e.g., batches for multiple patients in the same clean room) and pave the way for decentralized manufacturing by addressing key concerns regarding the contamination (and cross-contamination) risks of open systems, heavy reliance on trained personnel and batch-to-batch variability.
In addition to Lonza’s Cocoon system, companies such as Adva and Ori Biotech are focusing on turnkey GMP-in-a-box solutions that integrate multiple workflow steps in a fully closed, fully automated system to enable both centralized parallel processing and point-of-care (POC) manufacturing. Xcellbio aims to improve potency through metabolic conditioning during transduction and expansion of cell products with its AVATAR Foundry platform that automates liquid handling to streamline unit operations. Alternatively, Cellares’ Cell Shuttle approach can operate 16 concurrent batches in a preassembled, fully automated, ISO 8-compliant pod. MultiplyLabs and Cellular Origins are augmenting the existing manufacturing and quality control testing approach by utilizing robotic arms to execute unit operations from market-leading equipment (e.g., Cytiva’s Xuri) to avoid significant process development and revalidation.
Technological advancement in process equipment could progress decentralized manufacturing or even unlock POC manufacturing, where batches are produced in nontraditional GMP environments such as medical centers. Such manufacturing strategies have been a point of emphasis for FDA’s Center for Drug Evaluation and Research but will come with additional challenges, including site-to-site variability and analytical comparability between sites, which may further increase the need for more advanced in-line analytical testing solutions in the future. - Scalable cryopreservation and software solutions: Limited LN2-free options currently exist in the market — most notably, Cytiva’s electricity-based controlled-rate freezer (VIA Freeze) and shipper (VIA Capsule). The economics of LN2-free options are still a hurdle due to the considerable cost of the electricity to operate LN2-free systems and backup power systems (i.e., during outages) for storage. Other players such as Cryoport Systems, BioLife Solutions and CSafe are looking beyond freezing/thawing and focusing on end-to-end cryopreservation solutions to simplify the process for manufacturers. However, more broadly, fully closed automated systems may be the long-term solution for scalable cryopreservation by unlocking accessible POC manufacturing of autologous cell therapies and obviating the need for cryopreservation altogether.
While equipment is a key challenge for cryopreservation, software to manage the audit trail is equally as important for commercial-scale workflows. Software is an integral part of the ecosystem to connect various stakeholders (e.g., apheresis centers, developers, CDMOs) and minimize the delays to patient delivery caused by incomplete or inaccurate documentation. Integrated software solutions such as OCELLOS by TrakCel exist to eliminate the need for overlapping systems to manage COI and COC data across the entire value chain. For example, TrakCel offers a platform to track data from initial cell collection through drug product transportation that encompasses both clinical- and commercial-scale processes.
Conclusion
For more information, please contact healthcare@lekinsights.com.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2024 L.E.K. Consulting LLC
Endnotes
1EvaluatePharma database
2L.E.K. analysis of Citeline’s Advanced Therapy Database
3L.E.K. analysis of PharmaProjects
4L.E.K. analysis of TrialTrove
5Piper Sandler, “Cell Therapy Compendium 5.0 – Tackling Solid Tumors.”
6Cytiva, “COGS process economics for autologous cell therapy.”
7L.E.K. research, analysis and interviews