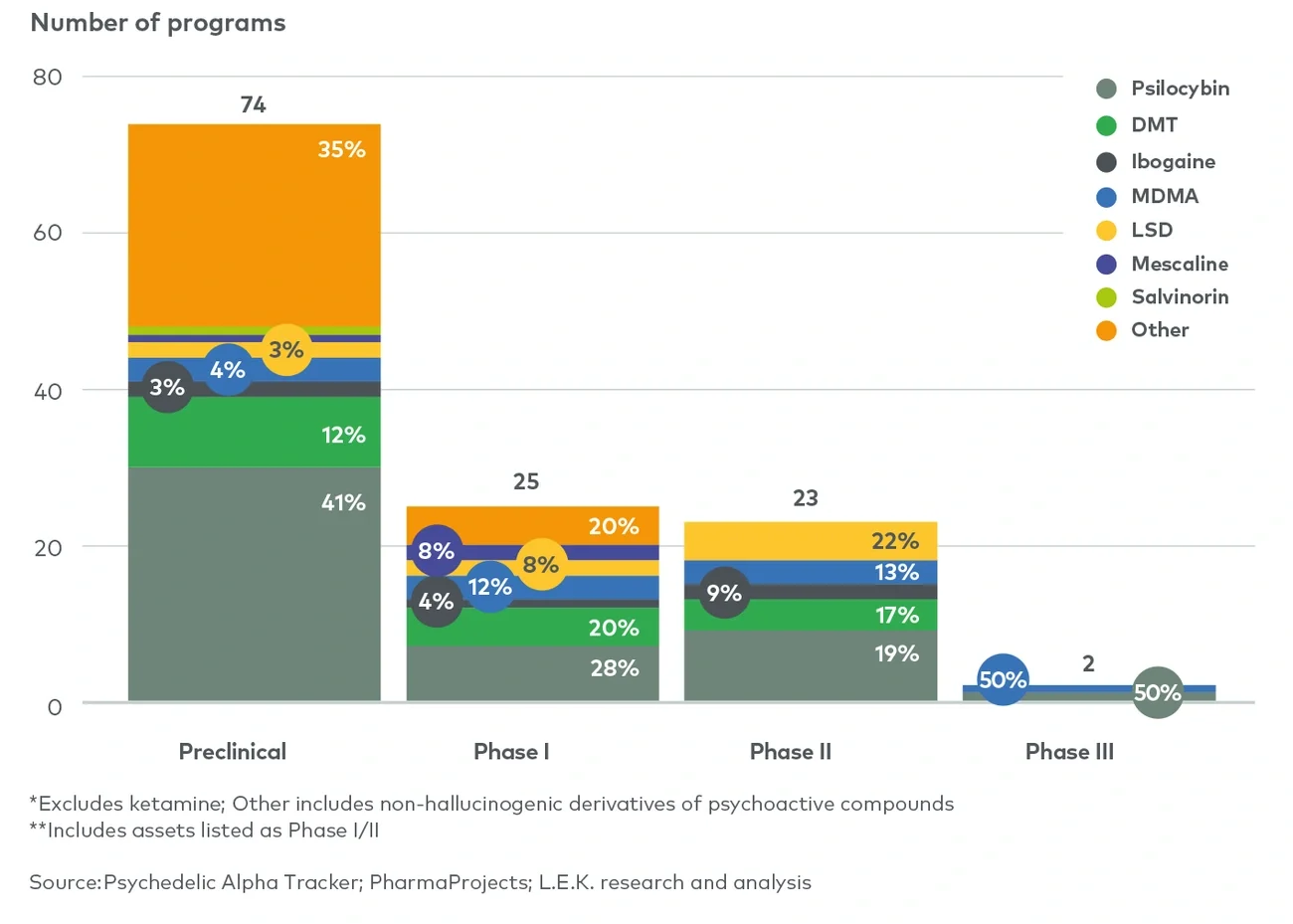
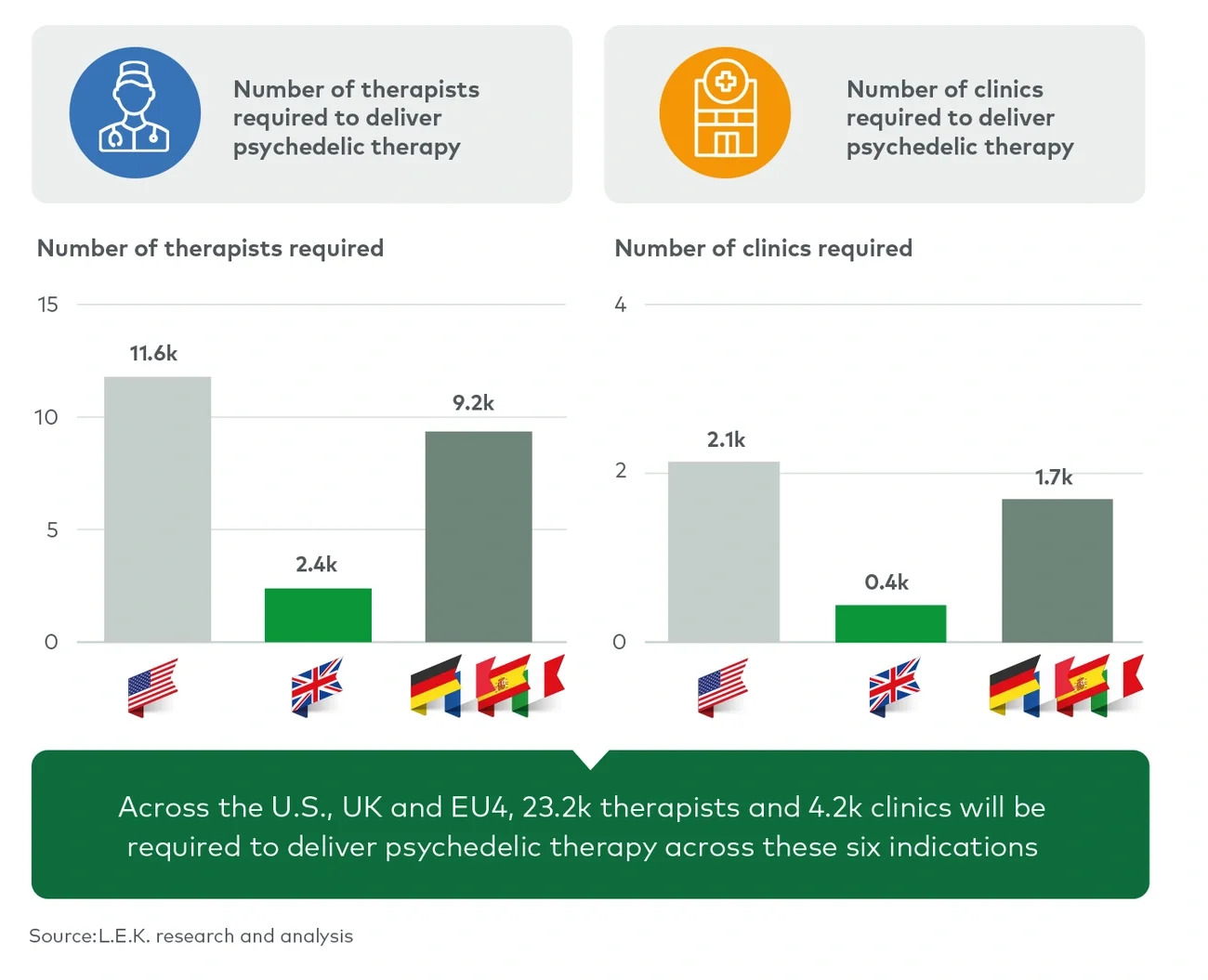
Although this is a substantial figure, an infrastructure of therapists and clinics will not need to be built from the ground up. There are currently more than 110 ketamine therapy clinics in the US, many of which operate as part of rapidly expanding clinic networks aiming to provide a base for the delivery of psychedelic therapy, upon approval. In Europe, psychedelic-assisted therapy could be provided alongside current treatments in existing outpatient psychiatric facilities. In both the US and Europe, clinics offering psychiatric interventions such as transcranial magnetic stimulation or electroconvulsive therapy may be well suited to the provision of psychedelic-assisted therapies due to their familiarity with complex delivery protocols as well as billing and reimbursement processes.
Specialised training is vital
Therapists and other clinicians will require specialised training, which may be delivered by a training provider, such as Fluence or the Institute of Psychedelic Therapy, or through training programmes run by psychedelic drug development companies. There are approximately 200,000 active licensed therapists practising in the US today, and forecasted demand would require about 6% of these practitioners to be trained in psychedelic-assisted therapy. The challenge of reaching this required volume will be amplified by an underlying shortage of therapists and mental health professionals.
Beyond reaching these broader capacity requirements, the most significant overall challenge — in terms of both physical and human infrastructure — is likely to be the ability to meet initial levels of demand as the therapies first launch.
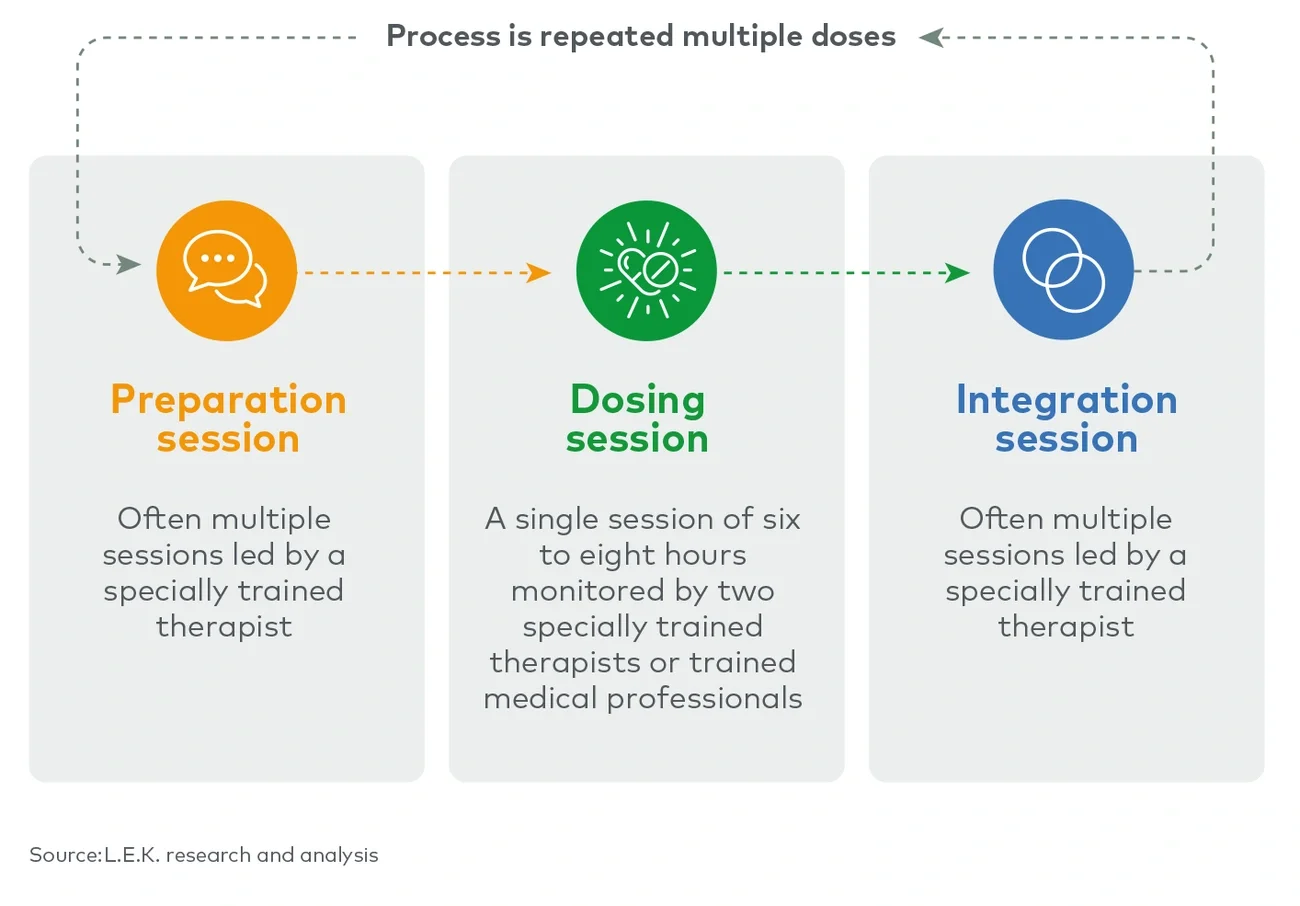
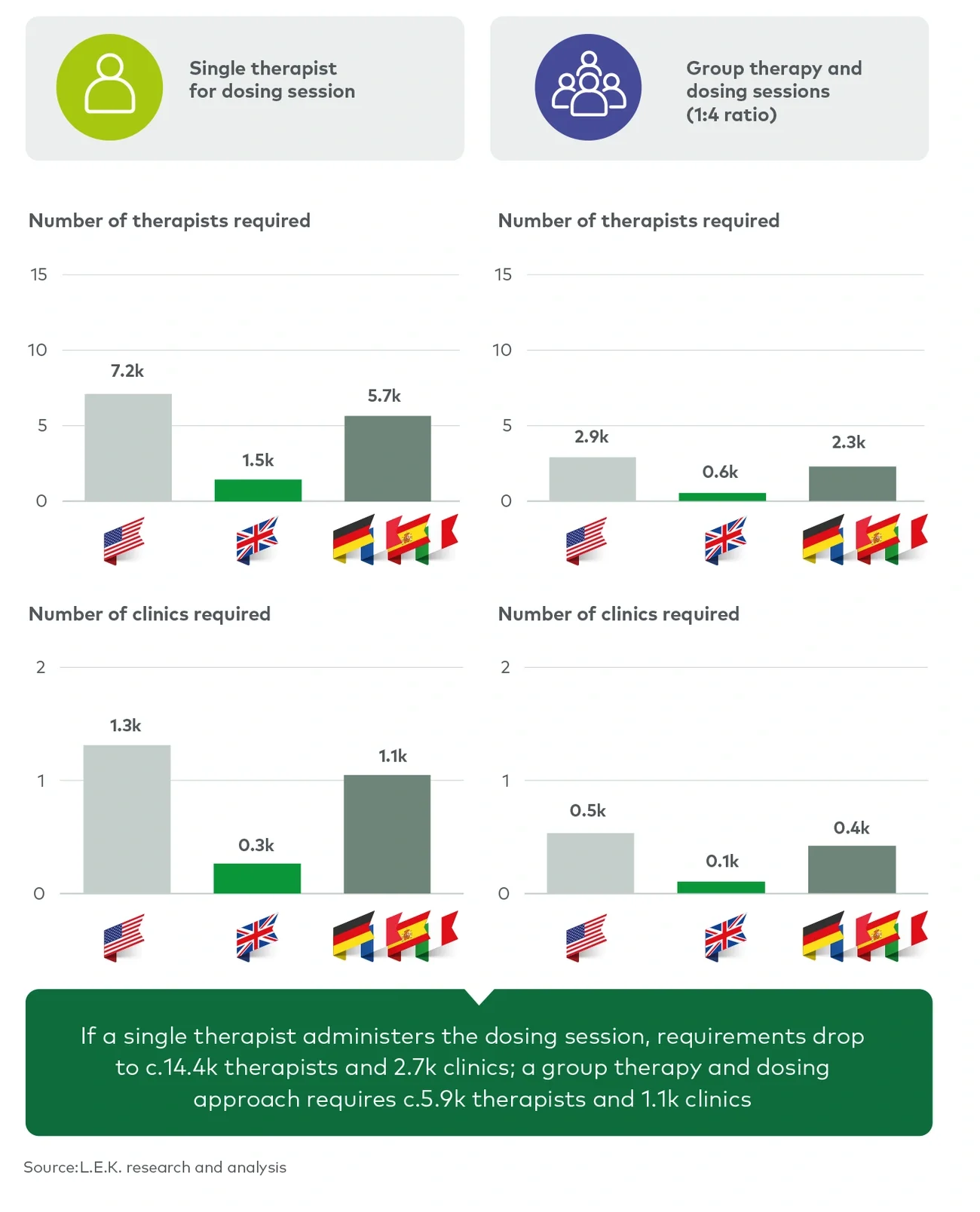
There are several key areas where delivery optimisation could reduce infrastructure and capacity challenges. Firstly, during the development of psychedelic-assisted therapies, the number of therapists specified in protocols for preparation and integration sessions has been reduced from two to one. Over the next two to three years, we expect protocols for dosing sessions to similarly reduce requirements to a single supervising therapist or two less-specialised assistants. The speed of this change will depend on how comfortable regulatory authorities are with the psychedelic therapies, and protocol reviews are likely to take place once the first psychedelic products have been launched. In this single-therapist scenario, only 14,000 therapists and 3,000 clinics would be needed to treat 400,000 patients per year (see Figure 4).