Healthcare professionals (HCPs), and specialist physicians in particular, are facing an increasing number of challenges related to medical knowledge and information access in their daily practice. Such challenges have grown exponentially over the past few years due to an information revolution in the healthcare ecosystem.
Transforming the Medical Affairs Function
Designed originally with a core purpose of addressing specific information requests from HCPs, the medical affairs function of most biopharma companies has yet to fully adjust to the new reality of its customer needs and take on the role of being a more active player in the creation and dissemination of scientific and medical knowledge. Doing so will deliver true customer-centric value and ultimately benefit patients through improved treatment outcomes. This evolution of medical affairs activities will also result in a redefinition of the function, giving it a more strategic role within biopharma organizations.
In this Executive Insights, we offer our perspective on the challenges faced by today’s HCPs and the changing expectations that biopharma commercial models see themselves confronted with, as well as L.E.K. Consulting’s experience and insights as to how biopharma companies should transform their medical affairs model to harness the full potential of this core function.
Understanding the origins and constraints of the medical affairs function.
Medical affairs functions were originally established with the purpose of delivering scientific information about biopharmaceutical products to HCPs, mainly key opinion leaders (KOLs) and specialist physicians, as well as selected other stakeholders such as researchers, scientists and pharmacists. Given the technical nature of the information, most medical affairs roles generally required (and continue to require) doctoral degrees such as an M.D., Dr. med., Pharm.D., Ph.D. or equivalent. Traditionally, the function was also heavily influenced by — if not directly subordinate to — the marketing function.
Starting in 2002 with the publication of the voluntary Code of Interactions with HCPs by the Pharmaceutical Research and Manufacturers of America (PhRMA), a strict separation occurred between the marketing and medical affairs functions. Consequently, the scope of activities within medical affairs expanded to include several areas beyond the provision of scientific/medical information (see Figure 1).

While a vast majority of biopharma companies have been executing against this scope of activities over the past decade or so, only a few have started to actively evolve the scope of medical affairs in light of significant changes in the practice environment of HCPs.
Focusing efforts on today’s HCP challenges
HCPs want to spend adequate time with patients to provide them with the best care in the most effective manner possible. In doing so, they need to make the right diagnosis and treatment decisions. However, these choices and decisions are becoming increasingly constrained due to two distinct types of challenges:
1. Practice-related challenges. Growing and aging patient populations — combined with an increasing level of patient awareness of and education on their disease treatment from novel channels such as health-related websites, online social communities and mobile applications — are putting an increasing burden on HCPs. In addition, payers and providers continue the drive for efficiency, and are imposing increasing requirements on physicians to justify an ever-greater variety and number of diagnostic and treatment decisions. Depending on location, such requirements emanate from a mix of government, payer, employer or health systems. Prior authorization requirements in the U.S. are the most notable example of this, though analogous requirements do exist in other healthcare systems, such as Cancer Drugs Fund applications in the U.K. This has resulted in an increasing need for competence in practice administration and a high level of compliance with externally imposed mandates.
2. Knowledge-related challenges. HCPs see as a top priority the furthering of their medical and scientific knowledge in order to enhance the effectiveness of their treatment decisions. However, staying up to date with the latest advancements in therapeutic options is becoming challenging for HCPs. This is due to the increasing volume and complexity of medical information, as well as a shortage of sources that combine trustworthiness (including objectivity, credibility and transparency) with easy accessibility and navigability (including ease of use in a timely manner, intelligent tailoring of content, and intuitive, physician-focused user interface).
The impact of these challenges is profound as they impair the physician’s ability to spend sufficient time with individual patients in order to make optimal decisions at critical points of disease diagnosis, treatment and management. Biopharmas can play a significant role in addressing these challenges, particularly those related to HCP knowledge gaps, as they are the best-equipped healthcare player for such tasks. Examples of interventions include:
Addressing patient expectations and managing interactions. Biopharma companies can help HCPs in addressing the high expectations of pre-informed patients or the leakages along patient histories and frequent consultations, through a wide range of solutions such as patient journey platforms, consultation enhancement tools and customized/tailored outcomes databases.
Providing updates on latest research. HCPs lack time to stay fully abreast of relevant therapeutic/clinical developments. A range of digitally enabled tools such as targeted search results, tailored knowledge updates, virtual conferences and bite-sized journals (“one-stop shop”) can be developed and provided by biopharmas to alleviate this challenge.
Aiding comprehension of product data. While relevant product data is easily accessible, HCPs struggle to find objective and credible data. Biopharma companies can help by enhancing the quality, accessibility and timeliness of their information through solutions such as off-label use and adverse events packages, virtual medical science liaisons, and fast-access product information services.
Validating diagnosis and treatment decisions. HCPs view peer and multidisciplinary interaction as a major value driver in improving outcomes. Biopharma companies can address the shortage of time and tools by designing and offering services and solutions such as live chat/P2P collaboration applications, medical concierge services, “second opinion” hotlines and virtual multidisciplinary toolboxes.
In aggregate, the above challenges and potential services and solutions represent a major opportunity for biopharma companies to reshape and transform the strategic relevance and impact of their medical affairs functions.
Rethinking the medical affairs function
This functional transformation requires a meaningful shift in the mindset of biopharma companies. Classic approaches to knowledge-related HCP interactions remain overly focused on selling (i.e., delivering product information/promotion for proprietary drugs) but now need to evolve toward a more objective, HCP-centric engagement focused on addressing current challenges. To that end, medical affairs functions must organize themselves around three sets of activities and solutions:
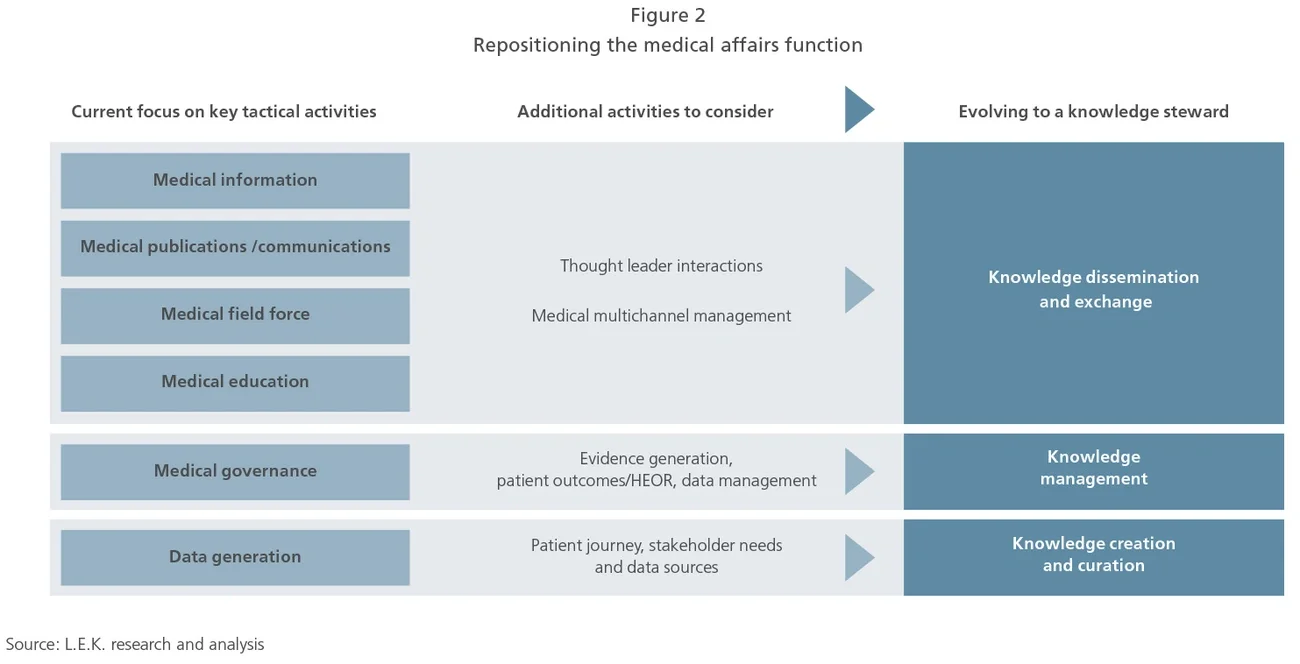
1. Knowledge creation and collection. Medical affairs needs to broaden its focus in terms of content knowledge. It needs to complement a historically deep focus on product technicalities by providing greater contextual insights. Key offerings in this regard include gathering insights within relevant indications and disease areas (e.g., underlying pathologies, role of genetics), deepening their understanding of key stakeholder needs across different clinician segments, evaluating patient treatment outcomes, and developing the ability to identify key data sources available to HCPs as well as gaps to be addressed.
2. Knowledge management. Medical affairs needs to better lead the internal management of a biopharma company’s medical knowledge. In this regard, a purely administrative and review role restricted to external communications is insufficient; the function needs to be at the forefront of generating evidence regarding the value of therapies through the use of big data. This implies the need for strong cross-functional engagement with relevant functions, such as health economics and outcomes research (HEOR) and real-world evidence, and building out of newer capabilities (e.g., healthcare informatics, predictive analytics).
3. Knowledge dissemination. Interactions with external audiences are often narrowly focused on KOLs and do not leverage the full range of channels and formats available. Medical affairs needs to expand its focus beyond KOLs to drive interactions with a wider range of HCPs. This requires careful consideration and planning around the use of communications, publications, education and digital services. Furthermore, medical affairs should also focus on replacing a one-way information push with an approach that fosters a meaningful two-way dialogue around medical knowledge (e.g., through deeper collaborations, investing in exchange forums) with their customers.
This evolution of medical affairs activities will result in a redefinition of the function with a more strategic role, and reposition it as a “knowledge steward” that becomes the backbone of cross-functional activities among internal functions and a trusted partner for external stakeholders (see Figure 2).
Key steps to enable a transformation of medical affairs
We recommend that companies look at medical affairs with a critical lens and act quickly in transforming this function; doing so will enable them to sustain competitive advantage and build lasting relationships with HCPs, one of their key customers.
This transformation is a lengthy process that will need to address the inherent complexity of changing the role and scope of a core function across an entire organization. Critical hurdles to overcome most often include divergent views among senior leadership on the optimal scope and breadth of the redefined function, the delineation of responsibilities versus those of other key functions (particularly market access, commercial and R&D), and the adaptability of the function to changing needs across therapeutic franchises, product life cycles and geographies.
Fortunately, there are a number of steps biopharma executives can take to be successful in transforming their medical affairs function. For most, they will need to consider:
- Developing a clear strategic vision for the medical affairs function that reflects the current healthcare market dynamics, addresses HCP practice realities and fits within the scale of the organization
- Understanding the current individual roles of medical affairs and other functions, such as R&D and commercial, and how they will interact with each other
- Mapping how the redefined medical affairs function will operate across different channels, therapeutic areas, product life cycles and geographic markets
- Assessing the resource and capability requirements for successful implementation
- Designing a structured system to measure the impact of the new medical affairs function and its activities
By addressing these steps, biopharma executives will enable true customer-centric value and ultimately benefit patients through improved treatment outcomes.








