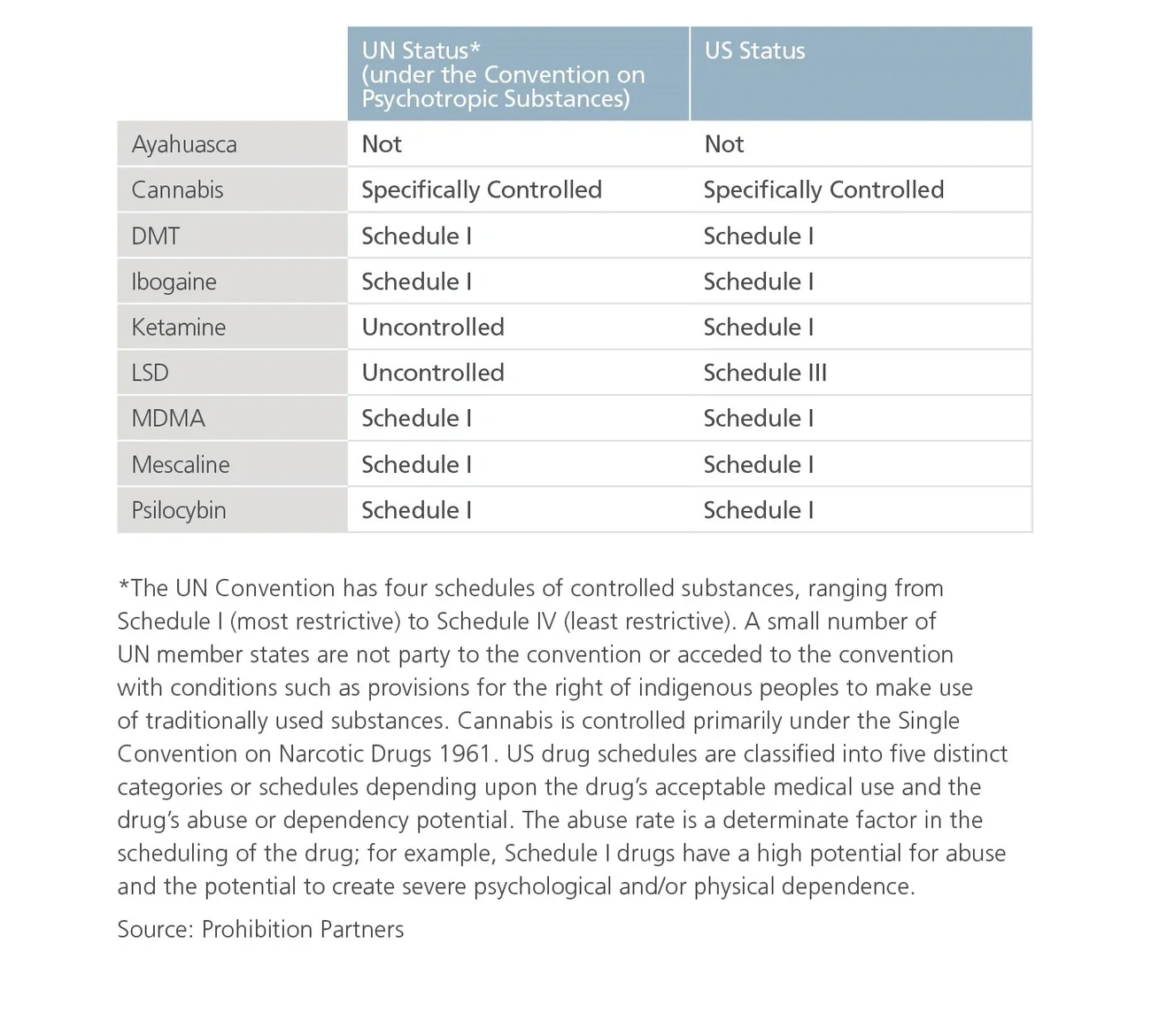
Mental health conditions such as depression, anxiety, PTSD, schizophrenia, and addiction are both widespread and notoriously difficult to treat. Their incidence is also growing, a trend that has been exacerbated by the worldwide Covid-19 pandemic. Unfortunately, this tragedy is compounded by the largely unsatisfactory treatment paradigms that are currently available for patients. Many find medicines ineffective or suffer side effects that are intolerable, and there are high relapse rates. Consequently, mental illness remains implicated in the deaths of thousands of individuals each year and is responsible for massive human suffering and economic productivity loss. The global cost of mental health conditions is projected to reach $6 trillion by 2030. Senior academics, clinicians, policymakers and charities regularly refer to a present-day mental health crisis.
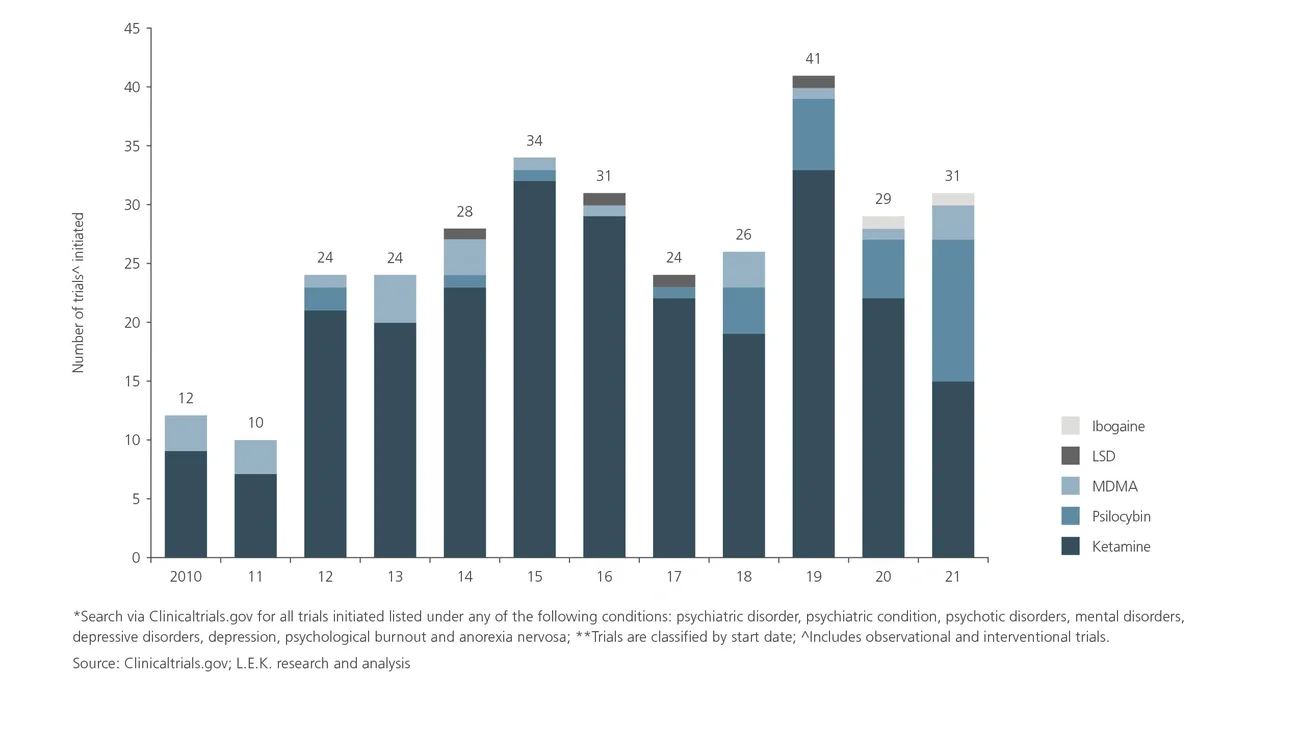
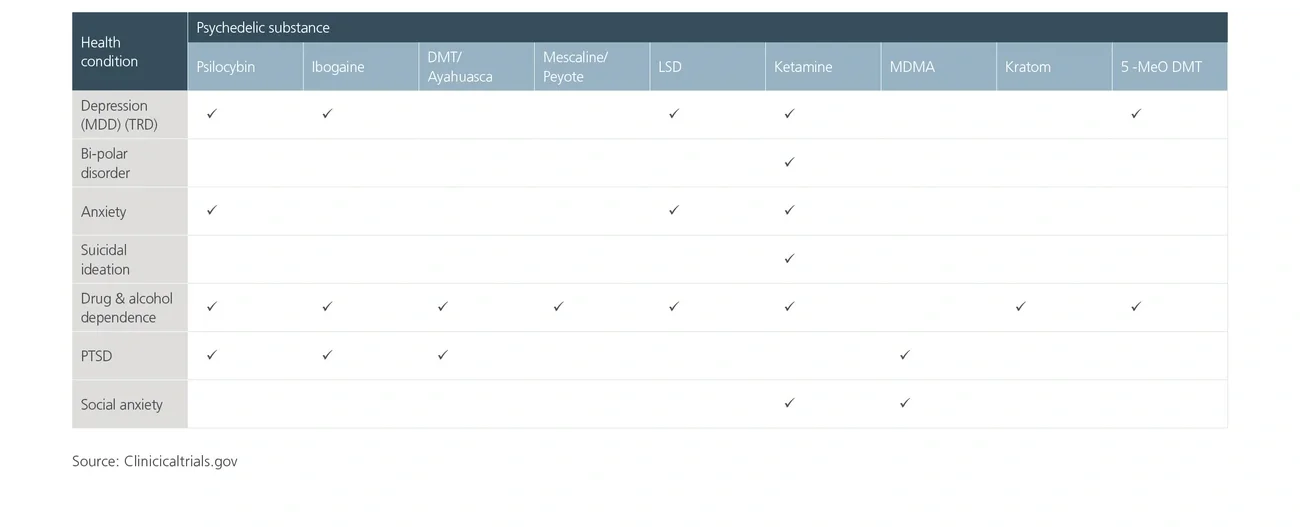
However, remarkable recent successes in psychedelic clinical trials may soon offer hope for patients, particularly MDMA for PTSD and psilocybin for depression and anxiety. As a result of these successes — coupled with US Food and Drug Administration (FDA) breakthrough designation, inward capital investment and evolving sentiment towards these treatments — we are at the dawn of a new era in treatment for mental illness.
These developments present new commercial opportunities in a market projected to be worth more than $40 billion by 2025 for a pharmaceutical sector that has largely retreated from psychiatry research. Whilst there are over 200 biotech companies in North America and Europe focused on the psychedelic space, large-cap biopharmaceutical companies like GSK and AstraZeneca have largely scaled back research efforts in psychiatry. In fact, estimates suggest that psychopharmacological drug research amongst large-cap pharmaceutical companies has fallen by 70% in the last 10-15 years, mainly due to the perceived difficulty in completing psychiatric clinical trials.
Nonetheless, these companies may wish to re-enter the space through acquisition, licensing, partnering or developing competing products. Notwithstanding exciting scientific and clinical advancements, there exist several underappreciated future regulatory and commercial challenges; biopharmaceutical companies should address these proactively now to ensure the best chances of success going forward.