4. Determine capabilities needed
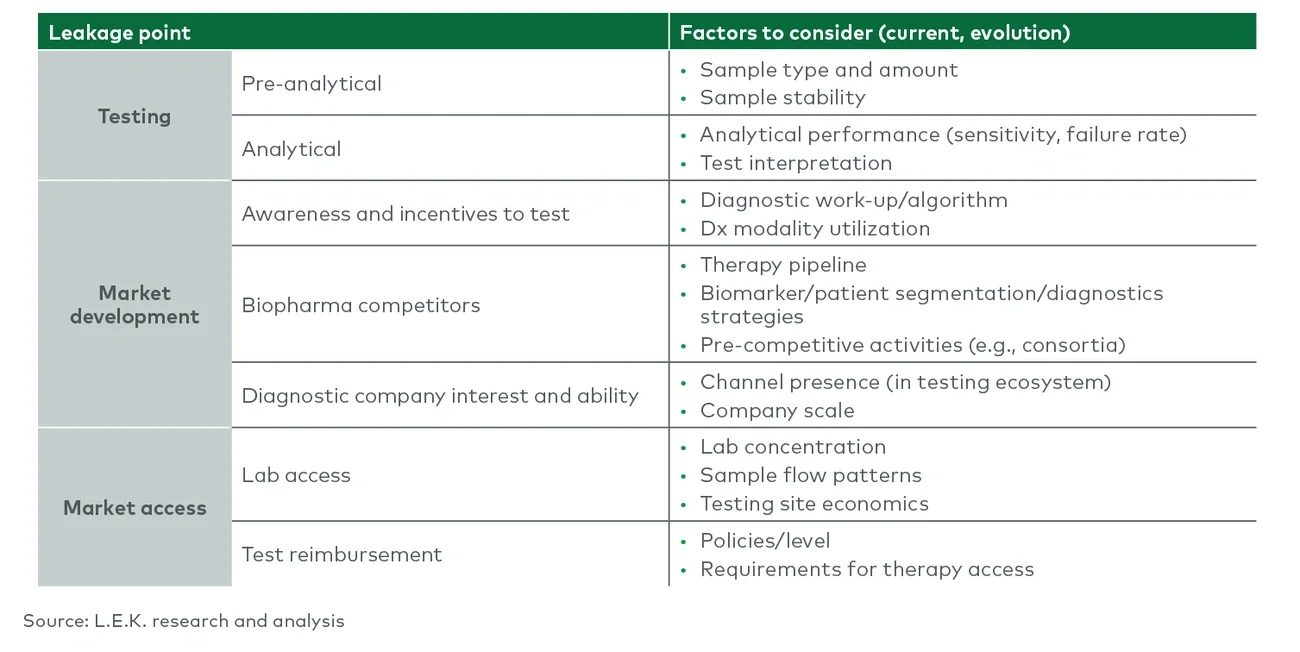
Following an audit of patient leakage across the portfolio, an organization can identify common themes and clusters of potential failure points. Focused discussions with internal and external stakeholders, including lab directors, physicians and patients, can identify capabilities (technological and organizational) and best practices that can address these potential failure points. Key capabilities to consider include biospecimen access for biomarker discovery and validation, longitudinal patient sample collection and biobanking for clinical trials, diagnostics-specific regulatory and reimbursement expertise, diagnostics-specific medical affairs and field personnel, and above-the-test services (e.g., sample collection support, reimbursement support, sponsored testing) for commercial diagnostic tests.
5. Assess capability gaps and determine how to address
A successful PM strategy will include a mapping of current PM capabilities across functions to identify potential capability/capacity gaps. A highly cross-functional team is required for this gap assessment, as the expertise required for PM excellence is often siloed, insufficiently linked between business units or lacking altogether. Next, a strategic options assessment can inform whether the organization should build capabilities in-house, outsource/partner or execute strategic M&A to address the gaps.
6. Implement change: Create a PM transformation management office
Finally, companies should implement change through a fit-for-purpose PM transformation management office. The remit of this office will depend on the organization’s current PM capabilities, breadth of future PM needs and specific new capabilities required.
Best practices for precision medicine enterprise strategy
While the optimal capability set and organizational structure will be company-specific, there are select best practices for PM enterprise strategy that apply across companies. Namely, a successful PM strategy requires seamless integration of PM personnel, capabilities and knowledge into all critical functions across the value chain. Successful integration should also ingrain PM into key processes and decision-making forums, including asset development stage-gates, budgeting and long-range planning. For example, some leading PM companies require candidate target product profiles to include discussion of potential diagnostic opportunities (e.g., potential biomarkers and implications for technology platform partners). Integrating PM efforts across functions naturally reinforces the “start early and reevaluate often” decision-making paradigm that drives PM excellence.
Going forward, companies seeking to deploy PM strategies beyond oncology will struggle to succeed if they benchmark solely against best practices for precision oncology. Instead, companies should aspire to develop fit-for-purpose PM strategies and supporting organizations suited to the capabilities they will need in order to deploy winning PM solutions that help improve the lives of the patients they serve. Now is the time to leverage the power of precision medicine to unlock the next wave of innovative therapeutics in neurology, immunology, cardiology and other high-need therapeutic areas.