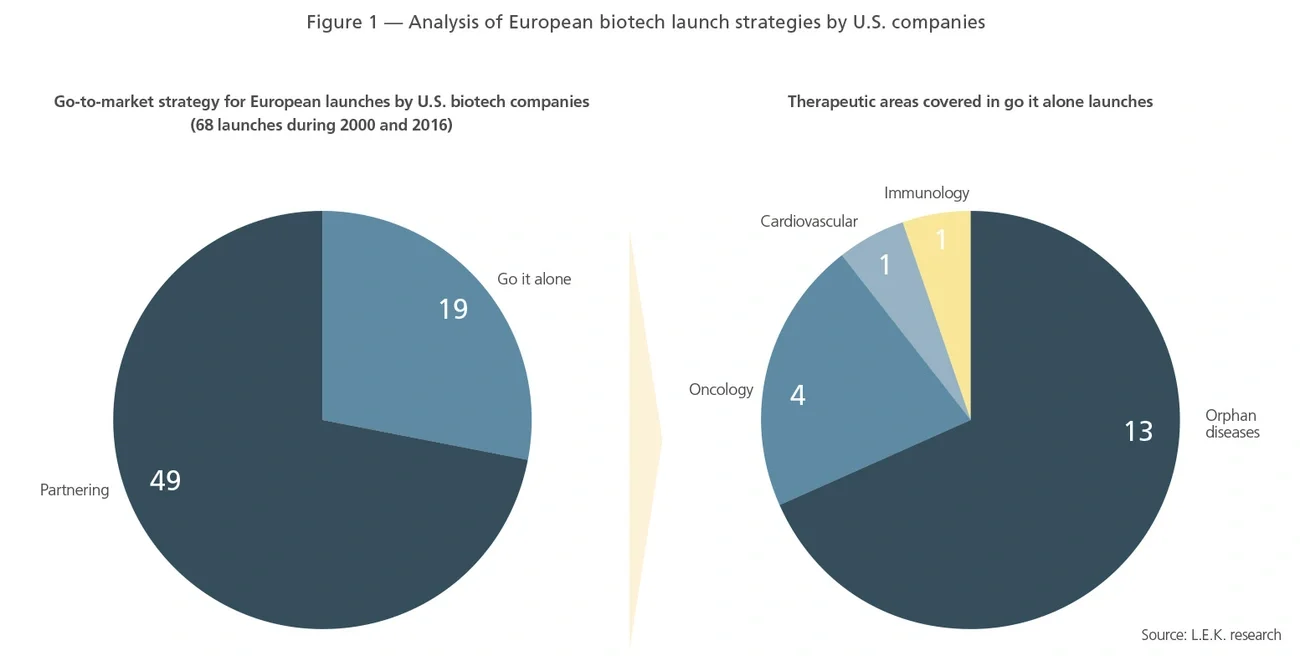
As our panel of experts revealed, the key reason for retaining the European rights of their first product was economic value. A good example was a U.S. rare disease company. When faced with the decision of whether to out-license in Europe or launch internally, the initial plan was to seek a partner. However, after conducting a thorough analysis of going it alone versus partnering, the company realized that the benefits of going it alone outweighed the risks. This was similarly a factor for a U.S. oncology company when it decided to build a presence in Europe for its hematology-oncology franchise. According to our executive interviews, if the commercial investment required is in the 10%-15% range of peak revenues, it is more attractive to go it alone in Europe than giving margins of 30%-40% to local or regional promoters and distributors.
Another reason for retaining European rights is the importance of control over the messaging, pricing, commercialization and branding of a first product. By owning and controlling the full commercialization of a first product, a biotech company positions itself to integrate a global brand plan, harmonize global market access and address the potential reference pricing issues from Europe in other regions of the world. Launching in Europe also helps build international capabilities and a global infrastructure that the company can leverage for a broader portfolio in the future.
Lastly, some U.S. biotech companies did not have a choice — they had to establish capabilities in Europe because of the sequencing of their first product launches across the two regions. InterMune is a good example of a company that faced a European launch decision after its first drug, Esbriet, was delayed by the FDA. In that case, the opportunity to own and control the launch in Europe may have held appeal, as it allowed InterMune to transform the company into a commercial organization, generate revenues in Europe to fund U.S. operations, and build market knowledge and relationships with key opinion leaders that the company could leverage to prepare the U.S. launch.
Picking the right drug
Going it alone has a number of benefits, but it might not be right for every drug. Emerging U.S. biopharma companies without a footprint in Europe need to think carefully about whether an in-house launch makes economic sense. Pharma executives point out two triggers for taking the risk of commercializing alone in Europe: a differentiated value proposition that addresses clear unmet needs and provides potential for premium pricing, and focused call points.
- A differentiated value proposition is crucial in Europe’s demanding payer environment, where cost-effectiveness or clear superiority to the standard of care on efficacy are prerequisites for market access and premium pricing. As one industry expert pointed out: “You have to keep in mind the budgetary constraints in Europe. There is essentially one payer — the taxman. So to ensure pricing power, you need a strong value proposition addressing a real clinical need.” Payer concerns around overall budget impact therefore make low-volume orphan drugs particularly viable candidates for European launches.
- Focused call points facilitate a targeted go-to-market approach and lean commercial infrastructure — a key consideration for smaller companies with limited financial resources. As mentioned by an industry expert, understanding the thresholds of call points and the customer model complexity under which you can operate and launch on your own is important. “You have to ask yourself: Is your drug going to be used in a narrow specialty field or by a large group of prescribers? This clearly influences the economic decision. In our case, we were looking at no more than 3,000 prescribers in Europe.”
While not viewed as a prerequisite, prospects of expanding the first product life cycle into additional indications can also justify an in-house launch. This was the case when Intercept built its European commercial infrastructure for Ocaliva, a drug to treat primary biliary cholangitis (PBC). The company made its first launch in an orphan indication, PBC, but also had a second indication in the pipeline: non-alcoholic steatohepatitis, which is much more prevalent and targets more call points. The prospect of a second (larger) launch coming may justify the investment in building out infrastructure in Europe.
Key success factors for launching a first product in Europe
Biopharma executives agree that for the right drug, a go it alone strategy can create more value than out-licensing would. However, beyond the impact of the therapy itself, several other factors determine the overall outcome when launching in Europe:
1. Over-investing in talent
Looking at their European launches, biopharma executives highlight a number of key ingredients for success. First and foremost, hiring and retaining the right talent — high-quality people with a strong track record in launch planning and execution and a good knowledge of European markets: “The first five to six hires are critical. They will determine the culture of the organization … you need people who truly understand the opportunity. It is only when you have been through the European launch process that you understand how complex it is.”
Equally important was the need to create a well-balanced, diversified and dynamic team: “Diversity of background is important. We looked for experience — all senior hires had experience in their particular roles. But we decided that no more than 10% should be from one company, to ensure diversity of opinion within the team.”
Once these initial hires have joined the company, they can drive additional talent acquisition and build a fully integrated team. It is crucial to promote a culture that ensures staff retention. As one industry expert pointed out, “Getting married is easy; staying married is another thing.” So how do you keep the talent? Good career prospects, a company culture that rewards creativity, and a strong mission and sense of purpose are key factors. Talented people want to move up the career ladder. They also want to feel empowered and be able to build on their creativity.
2. Empowering the European team
Freedom to maneuver in Europe should extend beyond hiring. The executives we spoke to stressed the importance of giving the European team the autonomy to make key decisions for their markets — within the confines of the overall launch strategy.
The complexity and diversity of the European pharma landscape makes it particularly important to entrust European decision-making to executives with in-depth knowledge of the market, and to refrain from calling all the shots from global headquarters. On the other hand, L.E.K. experience also suggests that too much autonomy in Europe presents its own risks. As mentioned by an industry expert who oversaw the European launches of multiple first biotech products, “There is a tendency of European organizations to replicate functions already in place at the U.S. headquarters … agreeing on which functions need regional versus global oversight is critical, as duplication comes at a significant cost.”
Finding the right balance between centralized decision-making from a U.S. headquarters and regional autonomy is therefore an imperative for building a winning European organization.
3. Planning and preparing early
The fragmentation of the European commercial and market access landscape makes launch readiness a lengthy process. Complicating the process is the scarcity of biopharma hubs where U.S. biotech companies can easily hire large-scale teams. Early planning is therefore critical for any product launches. A lead time of two to three years to plan a European launch is often recommended. As mentioned by an industry expert: “Some companies wait until the last minute to think about Europe and expect miracles that do not happen. You need at least two years before launch to be effective.”
And there is a clear hierarchy in the hiring process: Start with the region head, then prioritize the regulatory, market access and medical affairs leads. As some experts mentioned, “You build out your medical liaison team early — that is key. Do that 12-18 months before the product launch. It is important to build KOL [key opinion leader] relationships early on.” In a second wave, build commercial teams at a country level, with a strong country manager as the first hire. A staggered hiring process helps maintain focus and flexibility and avoids overspending on upfront costs, thus de-risking the launch.
4. Sequencing the country launches
Differences in market access among European countries call for a sequenced launch. While there is no set launch sequence, all the executives we spoke to agreed that the countries that provide early access and reimbursement should be tackled first.
In many cases, Germany is the first E.U. launch, as reimbursement is automatic for European Medicines Agency-approved drugs, and manufacturers are granted a year of free pricing while P&R negotiations are ongoing. Launches in Austria, Switzerland and the Nordic region frequently coincide with the launch in Germany due to relatively higher pricing levels compared with other regions, e.g., Southern Europe. For certain indications, early access programs can facilitate launches in France, Sweden and Italy shortly after approval.
The U.K. and Spain often follow in a later wave, as reimbursement is dependent on the outcome of cost-effectiveness assessments (U.K.) and lengthy negotiations with national and regional decision-makers (Spain).
5. Balancing regional vs. local European resourcing
At an operational level, building out the European organization requires careful timing and efficient use of resources. Successful European launches often benefit from a lean regional structure where some regional functional activities can be centralized. To account for national differences in payer and provider landscapes, experts recommend placing customer-facing roles at a local level in the countries: “In our organization, everything customer-focused was at a country level; everything strategic and the supporting functions were regional.”
Key implications for biopharma executives
The task of commercializing a first drug in Europe alone is a challenging one. Licensing out the European rights to an established commercial partner avoids many of the operational, regulatory and commercial pitfalls of going it alone. However, for the right product, Europe is an attractive market with clear value, despite its complexity and budgetary constraints.
For biopharma executives weighing whether to out-license or build a direct presence in Europe, here are some key strategic considerations:
- Building a commercial team and a “bricks and mortar” operation across multiple countries requires significant capital. Understand the financial impact of going it alone in Europe and the potential shareholder dilution.
- First product launches in Europe often unnecessarily take place with a timing close to U.S. market entry. Do not underestimate the operational distraction of a dual launch into two regions.
- The pool of resources and talent is dispersed in Europe, and there are few biopharma clusters. Select the location of your European headquarters early, and over-invest in a high-quality team with commercial experience and an entrepreneurial, “startup” mindset.
- Commercialization and market access in Europe are fragmented and complex. Initiate launch planning at least two years prior to approval.
- Many functional activities necessary to ensure a successful launch in Europe need to take place at a local level. Give the European team flexibility and the right level of autonomy to operate in its targeted geographies.
If all the above criteria are satisfactorily addressed, building a European presence and launching directly can provide a solid platform for international growth and expansion, creating meaningful shareholder value while also making a difference to patients.
Recommended reading: Biopharma Operational Scale Up for First Product Launch: Planning for Successful Execution in Challenging Times
1 Views in this article reflect the combined experience of L.E.K. Consulting and Korn Ferry, as well as guidance from interviews conducted with seven commercial leaders from biotech companies that have built commercial operations in Europe to support their first product launch.