The speed of COVID-19 vaccine development is unprecedented in modern medicine, as is the volume of vaccine demand. To meet this demand, stakeholders have had to rethink their production approaches, making large investments, repurposing existing facilities and developing complex supplier networks. In this Executive Insights, we discuss the major challenges associated with meeting the global demand for COVID-19 vaccines, solutions that stakeholders have adopted and the longer-term implications for the pharma industry.
COVID-19 Vaccine Manufacturing: Challenges, Solutions and Implications for the Pharma Industry
Key takeaways
To overcome production bottlenecks, COVID-19 vaccine stakeholders have increased investments in manufacturing capabilities, optimised their processes, secured additional capacity through partnerships and are increasingly focusing on local supply chains.
Manufacturers have also leveraged technology transfer of techniques, equipment, and, more importantly, skilled personnel, to enable the mass production of innovative vaccines at a remarkable speed.
Since the COVID-19 pandemic accelerated the development and commercialisation of new vaccine platforms, we believe that the pharma industry is likely to apply these newfound capabilities to develop other vaccines and therapeutics − well beyond COVID-19.
There are opportunities for continued, broader collaborations between pharma companies going forward, and we expect partnerships between pharma and contract development and manufacturing organizations (CDMOs) to become increasingly strategic, with longer and deeper agreements.
The progress rate of the R&D response to COVID-19 has been unprecedented, with multiple vaccines approved within a year after identification of the virus. This created the need to scale up production much more rapidly than is common in pharmaceutical development, a challenge further exacerbated by the global nature of vaccine demand. Vaccine manufacturers and suppliers of vaccine components are aiming for cumulative supply targets of up to 14 billion COVID-19 vaccine doses by the end of 2021. This target is three- to fourfold higher than the estimated annual global demand for all vaccines pre-COVID (3.5 billion to 5.5 billion doses).
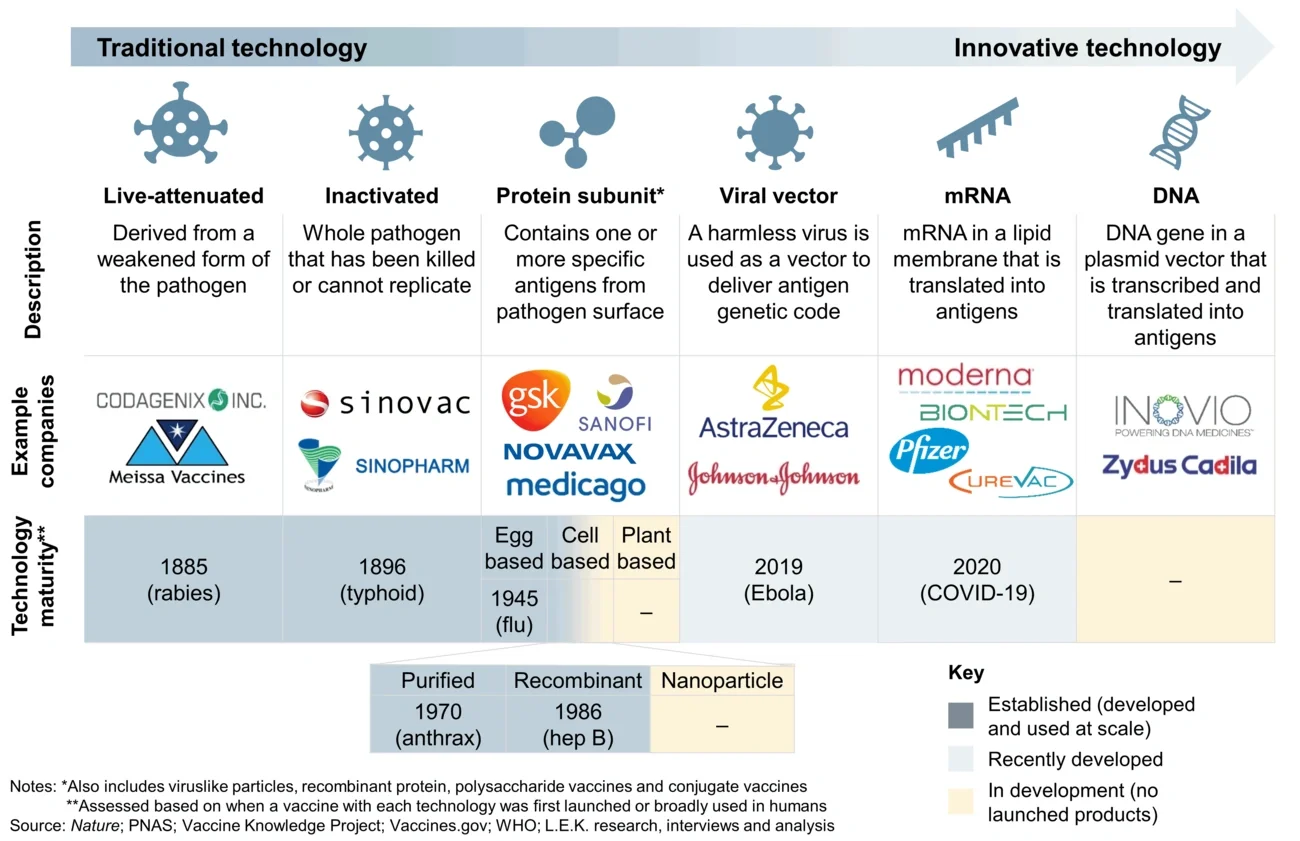
Vaccines have been developed using a range of different technology platforms (see Figure 1), including several novel technologies that had limited clinical validation prior to COVID-19 (e.g. mRNA, viral vector). The industry’s lack of prior experience in scaling up manufacturing of these technologies created additional challenges.
Figure 1
A range of traditional and innovative vaccine technologies are being utilised in the battle against COVID-19
This remarkable vaccine demand has unsurprisingly led to manufacturing bottlenecks, which have slowed the world’s exit from the pandemic. Some of these bottlenecks apply to all vaccines, whilst others are specific to individual technology platforms (see Figure 2). We have categorised bottlenecks as being related to material and equipment shortages, manufacturing process complexity, or capacity constraints.
Figure 2
COVID-19 vaccines are subject to a range of pre-existing and pandemic-related manufacturing bottlenecks
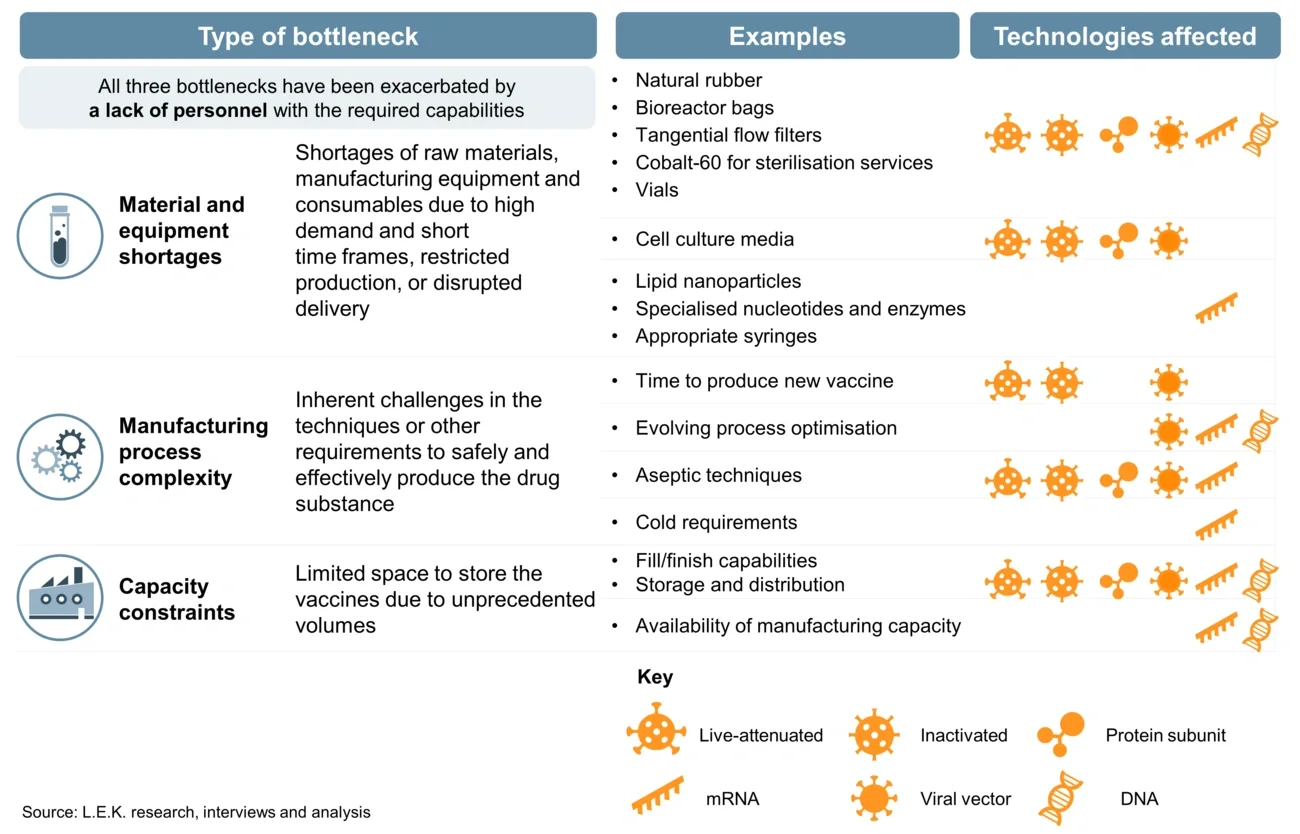
Material and equipment shortages impact a range of steps in the production process, including active pharmaceutical ingredient (API) or vector manufacturing, fill-and-finish services, and even testing and quality control. Shortages of materials and components — such as natural rubber, vials, syringes, flow filters and cobalt-60 for irradiation sterilisation — have all been reported. There have also been challenges in sourcing sufficient reagents, such as enzymes and lipids for mRNA- and DNA-based vaccines, and cell culture media for other vaccine technologies.
Manufacturing process complexity arises from the need for rapid production and the prompt resolution of technical issues. Each technology comes with its own specific challenges. Whilst processes to manufacture traditional modalities − such as live-attenuated or inactivated virus vaccines − are well established, these require time to scale up. Innovative vaccines − such as viral vector- and mRNA-based vaccines − had never been manufactured at scale before the pandemic, hence optimised processes and expertise were lacking. The ultra-low-temperature storage of mRNA-based vaccines posed a particular challenge to rapid manufacturing and distribution on this scale, necessitating significant investment. For example, Pfizer invested $2 billion in late 2020 to build its own cold-chain network. As with all innovative approaches, the supporting industry, such as contract development manufacturing organisations (CDMOs, lipid nanoparticle producers) is still nascent and required time to ramp up production to meet demand.
Capacity constraints have also been a major bottleneck. Adding production lines or building new facilities to ramp up production capacity is time-consuming. Outsourcing production to CDMOs or other partners requires technology transfer, which has proven to be particularly difficult for innovative technologies; one example is the contamination of c. 60 million doses of Johnson & Johnson COVID-19 vaccine with an ingredient used to produce AstraZeneca’s COVID-19 vaccine. Additionally, the complex supply chains associated with vaccine manufacturing contribute to capacity constraints (e.g. Pfizer’s vaccine uses a reported 280 ingredients sourced from 19 countries).
Vaccine stakeholders have put in place a range of actions to overcome production bottlenecks in the pressure cooker environment of the pandemic (see Figure 3). On top of investments in vaccine manufacturing capabilities, vaccine stakeholders have optimised their processes, have secured additional capacity through partnerships and are increasingly focusing on local supply chains.
Figure 3
COVID-19 vaccine stakeholders have invested in and built capacity, and partnered to secure and enhance manufacturing capabilities in response to bottlenecks
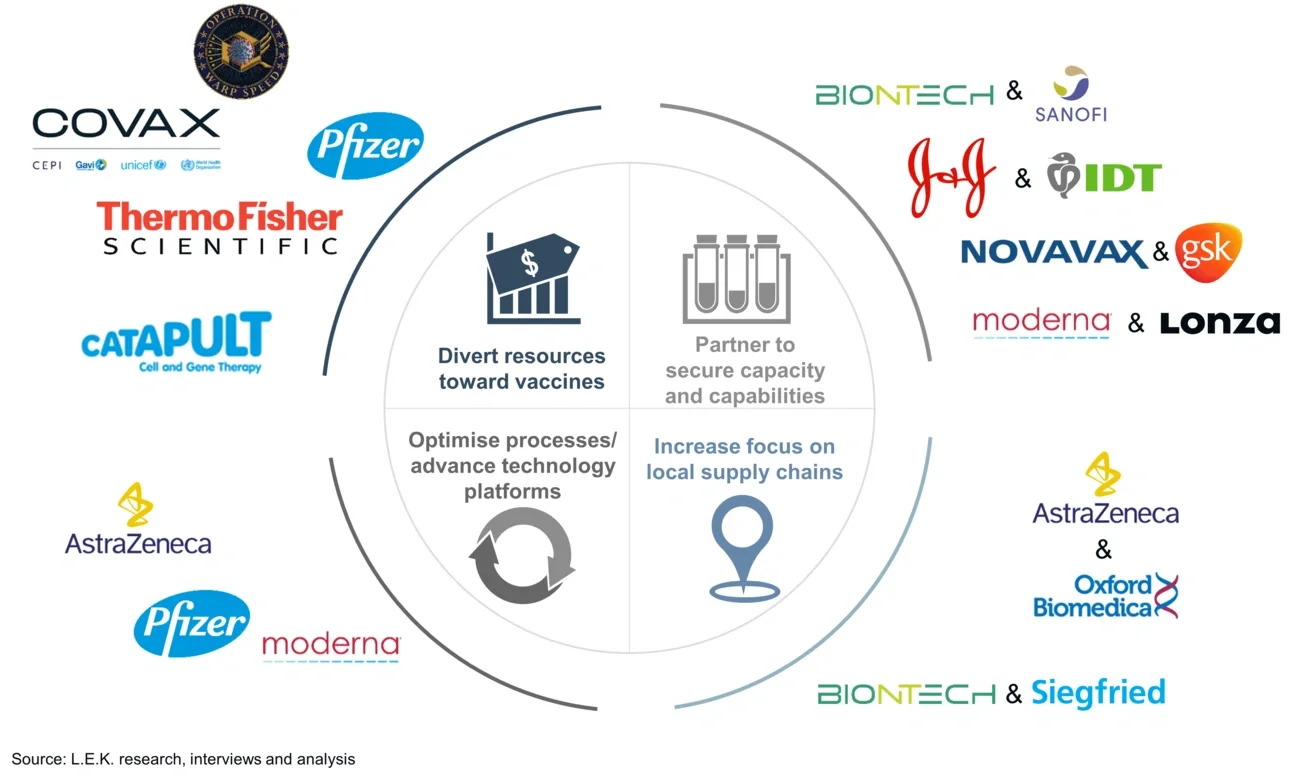
To overcome material and equipment shortages, stakeholders with existing capabilities have diverted production capacity toward COVID-19 vaccines, temporarily using existing production lines whilst building new capacity. The c. $10 billion funding from governments and other sources — such as Operation Warp Speed in the US and the global Coalition for Epidemic Preparedness — has in part been invested in capacity expansion. Pharma companies and CDMOs have also directly invested in additional capabilities, such as Pfizer’s purchase of seven lipid manufacturing machines from Polymum Scientific at a cost of c. $200 million each. Finally, manufacturers have increasingly focused on local supply chains because the pandemic threatened global trade. Oxford/AstraZeneca, Valneva, Novavax and CureVac are all producing COVID-19 vaccines in the U.K. Two rapid response centers are under construction in the U.K. and are expected to be ready by the end of 2021.
To overcome manufacturing process complexity, which remains a key challenge, efforts have been made to retrofit facilities (e.g. AstraZeneca and Pfizer in the EU) and improve yields (e.g. self-amplifying RNA vaccines may require lower doses, substantially reducing manufacturing volume requirements). Manufacturers have leveraged technology transfer of techniques, equipment and, more importantly, skilled personnel to enable the mass production of innovative vaccines at a remarkable speed, and progress is set to continue. For instance, the ultra-cold chain requirements for mRNA vaccines are likely to be resolved in the short to medium term by using chemical alterations of mRNA and LNP formulae, lyophilisation, and high-quality containers to increase long-term stability. Moderna and CureVac have both already announced new data supporting a one-to-three-month shelf life at 2-8°C for their COVID-19 vaccines.
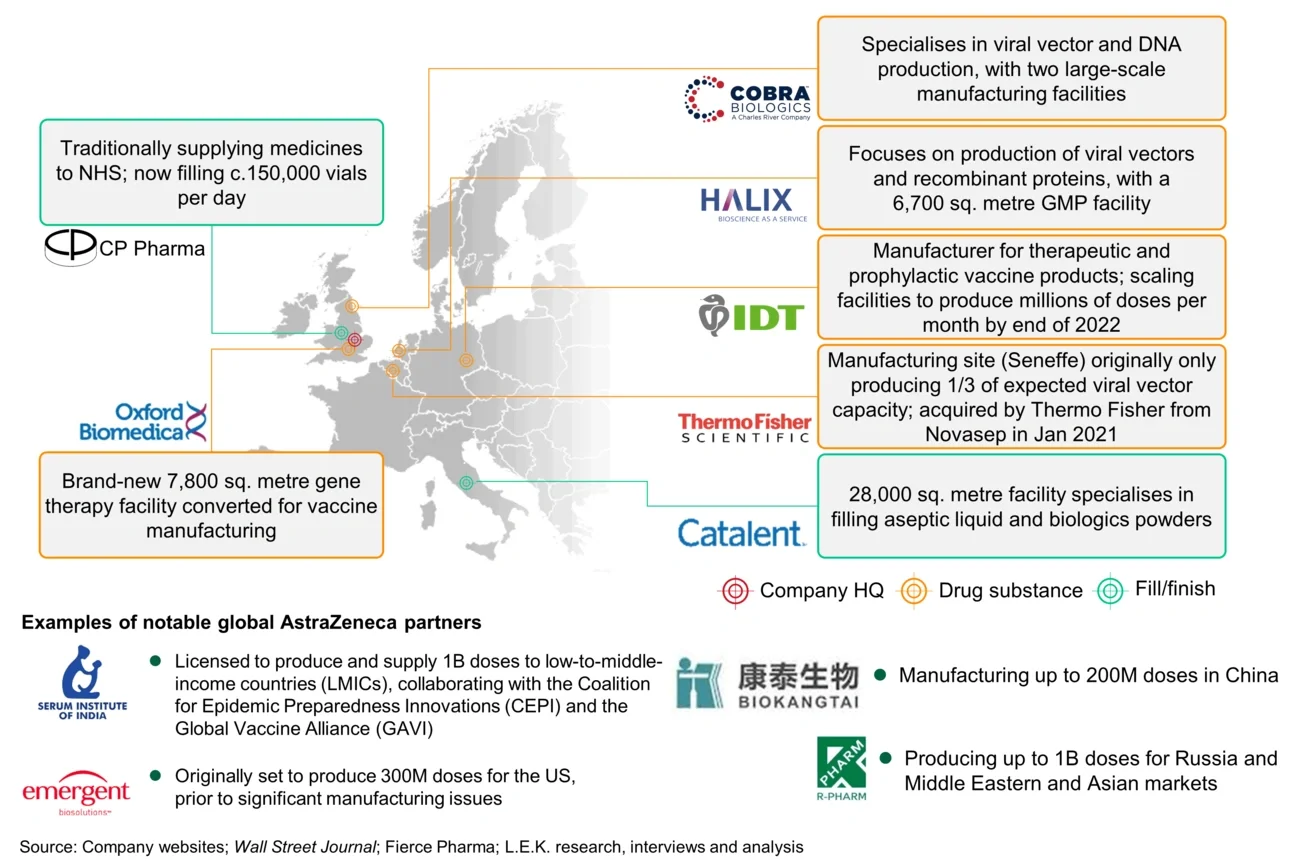
To overcome capacity constraints, vaccine manufacturers have established a broad range of partnerships, not only with CDMOs but also with their peers, such as Pfizer/BioNTech with Novartis, whilst at the same time investing in building new facilities. For example, AstraZeneca has partnered with several CDMOs in Europe, including IDT Biologika, Cobra Biologics and Catalent (see Figure 4) to maximise global production capacity for manufacturing the drug substance, as well as fill-and-finish. To meet demand, some existing capacity has been repurposed. One example is Catalent shifting some of its viral vector capacity, originally intended for gene therapies, to serve the viral vector demand for AstraZeneca’s vaccines.
Figure 4
Vaccine developers, such as AstraZeneca, have established a broad range of partnerships in Europe and beyond to maximise global production capacity
The pandemic has accelerated the development and commercialisation of new vaccine platforms, with multiple vaccines launching in record time (mRNA, viral vector, and — potentially soon — protein nanoparticle).
Going forward, pharma is likely to use these newfound capabilities to develop vaccines and therapeutics beyond COVID-19. For instance, mRNA-based vaccines hold significant advantages over existing options in terms of efficacy, rapid production processes and the flexibility to switch quickly between multiple strains for infectious diseases. This makes them well suited to diseases such as Zika or malaria for which currently available vaccines are either insufficiently efficacious, or for seasonal diseases such as influenza. Additionally, new vaccine platforms are likely to play a role beyond infectious diseases, with a number of cancer vaccines in clinical stages of development (e.g. for advanced melanoma). These future developments will benefit from the rapid technological advancements made with COVID-19 vaccines, enabling faster clinical development and manufacturing scale-up ahead of commercialisation.
The vast efforts made to overcome COVID-19 vaccine-related production bottlenecks have led to significantly increased capacity. Post-COVID, the pharma industry will look to repurpose much of this capacity to meet growing demand in other areas such as oncology, with a particular focus on fill-and-finish lines, where capacity has been traditionally limited. In addition, the spectre of vaccine nationalism and the disruptions to global trade seen during the crisis have led manufacturers to rethink their supply chain approach and to repatriate manufacturing closer to distribution sites. Although the exact details of any potential repatriation of supply chains are still being worked on (e.g. which APIs are prioritised and timing), the topic of pharma manufacturing has moved up on various nations’ agendas.
The crucial role that CDMOs have played in COVID-19 vaccine production reinforces their importance in the industry. We expect partnerships between pharma and CDMOs to become increasingly strategic, with longer and deeper agreements.
The openness of pharma companies to partner with each other during the pandemic also creates opportunities for continued, broader collaborations going forward. Such collaborative efforts could be replicated to improve the likelihood of success in areas of high unmet need, both in infectious diseases (e.g. antimicrobial resistance) and beyond.
Overall, pharma’s concerted efforts in COVID-19 vaccine development and manufacturing have not only significantly accelerated and strengthened governments’ pandemic responses, but have also created the foundations for better infectious disease control and advances in critical areas outside of infectious diseases, such as oncology. Collaborations among pharma companies or between pharma companies and CDMOs, as well as newly created production capacity, will play a crucial role in enabling future progress.








