As the vaccination programme builds momentum, experts from L.E.K. Consulting’s Healthcare and Consumer practices have come together to create a framework for modelling recovery scenarios to help businesses plan for the upturn. This article focuses on the implications for the travel and leisure sector, one of the hardest-hit parts of the economy, but the lessons apply more widely across the consumer landscape.
The COVID-19 crisis caused previously unimaginable levels of medical and humanitarian catastrophe, ripping through the global economic fabric and devastating business communities around the world. For much of 2020, some sectors like travel and leisure were effectively entirely shut down as we closed venues and borders, and ordered quarantine and ‘self-isolation’, prompting furlough and redundancy rates amongst the highest ever seen in the modern era. However, 12 months on and the world’s scientific community have delivered amongst the most remarkable medical achievements: a vaccine approved by regulators in fewer than 300 days since clinical trials commenced. As the most ambitious vaccine roll-out in history ever undertaken starts to gain traction, we ask: how should travel and leisure businesses think about, and plan for, a successful bounce back?
We developed a post-pandemic planning framework (PPPF) to assist companies in modelling how the impact of the vaccine roll-out and subsequent opening up of the economy may influence the content and timing of their recovery plans. The PPPF includes five steps, each of which has an impact on the next.
1. The vaccination programmes
Assuming high efficacy rates in preventing symptoms and transmission, the critical first question in the PPPF is to consider how rapidly the global vaccination programme can be delivered. While the UK is currently amongst the top five countries globally in terms of administered vaccine doses per 100 people, in-country vaccination programme roll-out appears uneven at present, which will impact the sequencing of travel corridor reopening. Manufacturing capacity bottlenecks are constraining the pace of roll-out, but these problems will ease, and the global programme of vaccine development is gaining momentum. We consider these themes in turn.
How rapidly will we be able to scale in-country vaccination programmes?
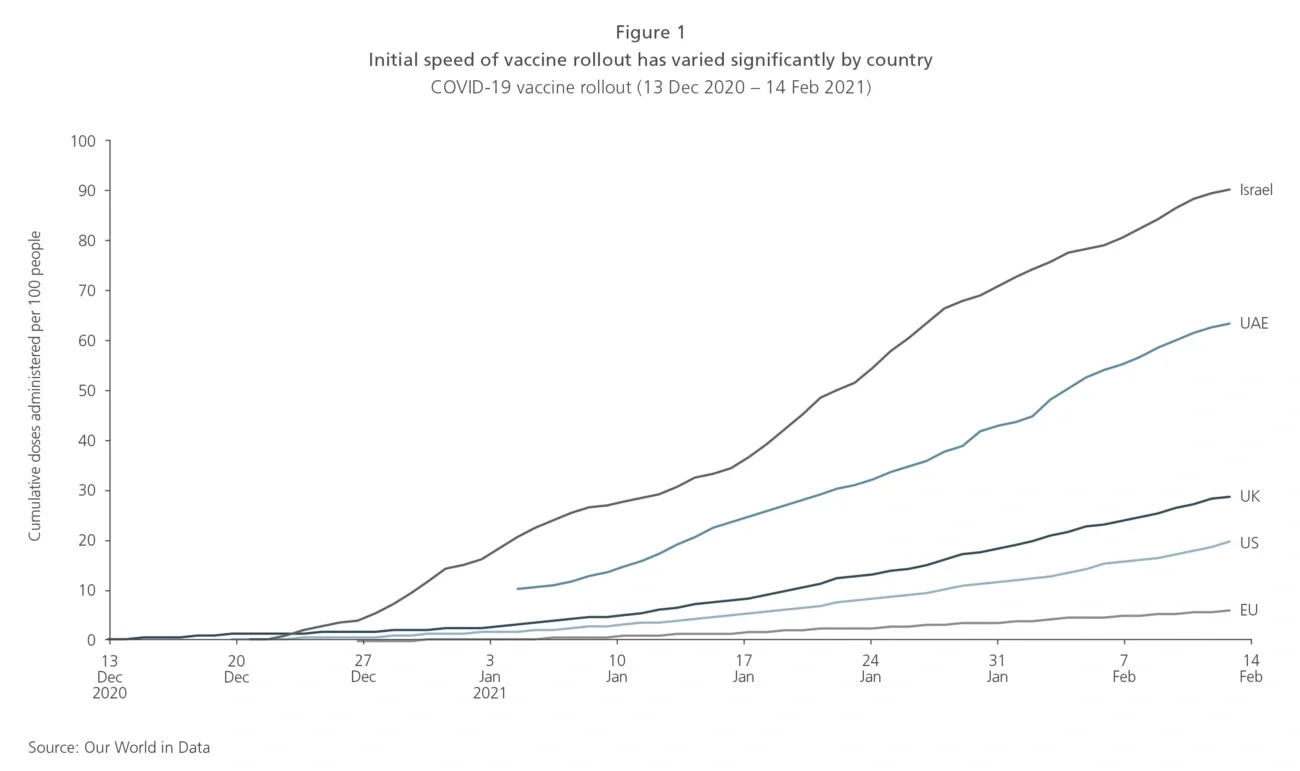
Worldwide vaccination programmes are now underway, with some countries like Israel, the UAE and the UK performing particularly well, whilst others are just getting started. For domestic travel and leisure businesses, this is already good news, but for international travel to rebound, the progress of vaccination programmes in the destination market and other key origination markets for those destinations are also critically important. There are notable flows of sun-seeking passengers into Spain, Turkey and Greece from the UK, Germany, Benelux and Nordics ― vaccination programmes across these markets will set the pace of the recovery.
In the US, the new administration under President Biden has committed to an accelerated vaccine roll-out strategy to provide 100 million vaccine shots in 100 days. Notably, European countries appear slow and have not yet shown much evidence of an accelerated vaccine roll-out plan. Current estimates suggest Europe could be six to nine months behind the progress already made in the UK, with vaccination of most target priority groups not complete until the end of 2021 or even 2022.
For approved vaccines, how quickly will manufacturing capacity ramp up?
Part of the challenge lies upstream in vaccine manufacturing, which is a highly complex process to manage and to scale. Many of the vaccine candidates in the development pipeline have been ‘manufactured at risk’ (i.e. they have been manufactured before achieving regulatory approval). This has led to some ability to stockpile vaccine whilst the regulatory approval process is underway. Manufacturing capacity is usually increased through two approaches: scaling up (increasing the size of vaccine batches) and scaling out (creating multiple production streams). Both of these approaches are in progress to achieve the volumes of COVID-19 vaccine required globally.
Notwithstanding the considerable efforts to increase capacity, there have been recent reports of interrupted supply, which cannot be ruled out going forward. For at least 9-12 months, continued shortages of vaccines will be likely across most countries.
How many more vaccines are likely to be approved over the next 12 months?
However, there is a reassuring level of momentum in the global programme of vaccine development (see Figure 1). Since Comirnaty, the Pfizer/BioNTech COVID-19 vaccine, became the first approved by a UK regulatory agency (the MHRA) anywhere in the world, eight others have also received authorisation (e.g. Oxford/AstraZeneca, Moderna) in at least one country; more than 300 remain in development.
Vaccine development timelines have been radically shortened without impacting safety standards. Regulatory agencies around the world have further reduced timelines through several initiatives, for instance, reviewing data more frequently through the trial process (‘rolling reviews’). Whilst every trial carries some possibility of delay or failure (e.g. GSK/Sanofi’s vaccine candidate did not show sufficient immune response in the elderly), the sheer number of candidates in development suggests a large number may be approved over the coming months.