Key takeaways
- The COVID-19 pandemic has had a profound impact on hospitals, bringing new challenges, opportunities and resilience for medtech and pharma companies.
-
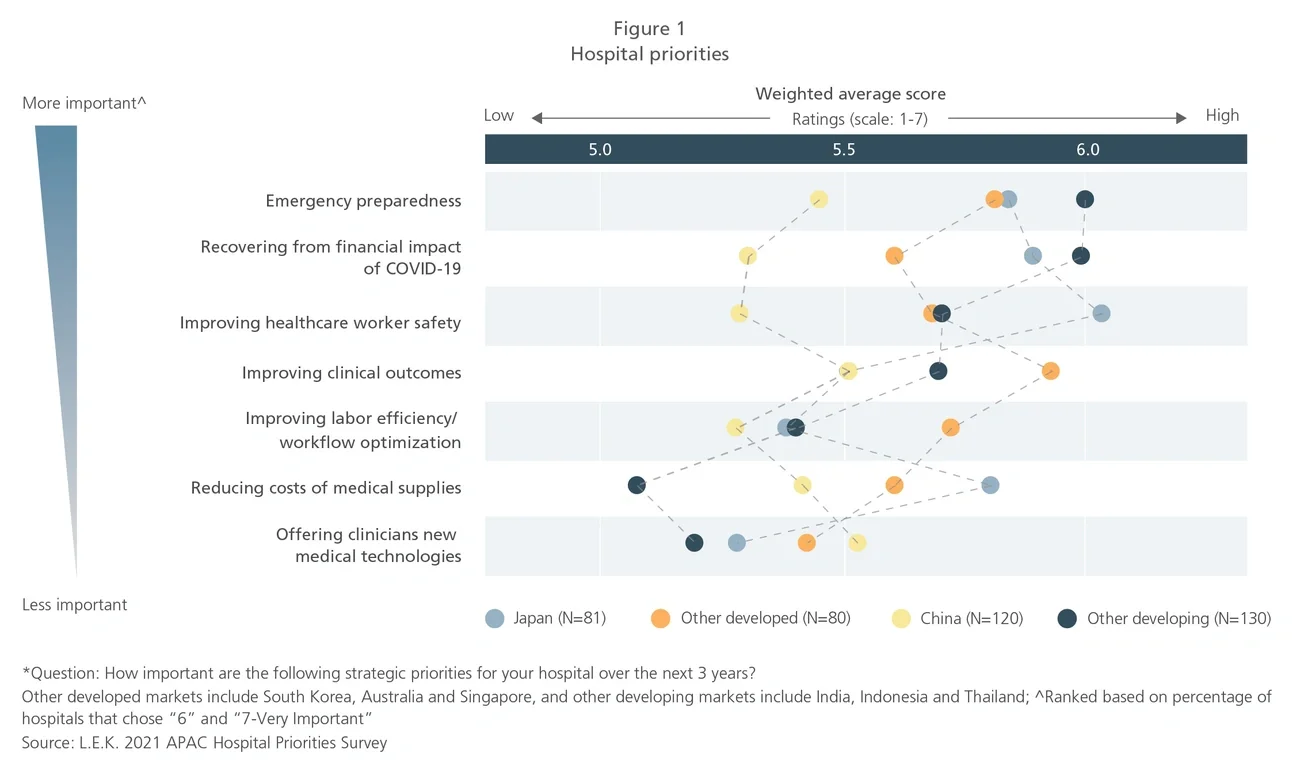
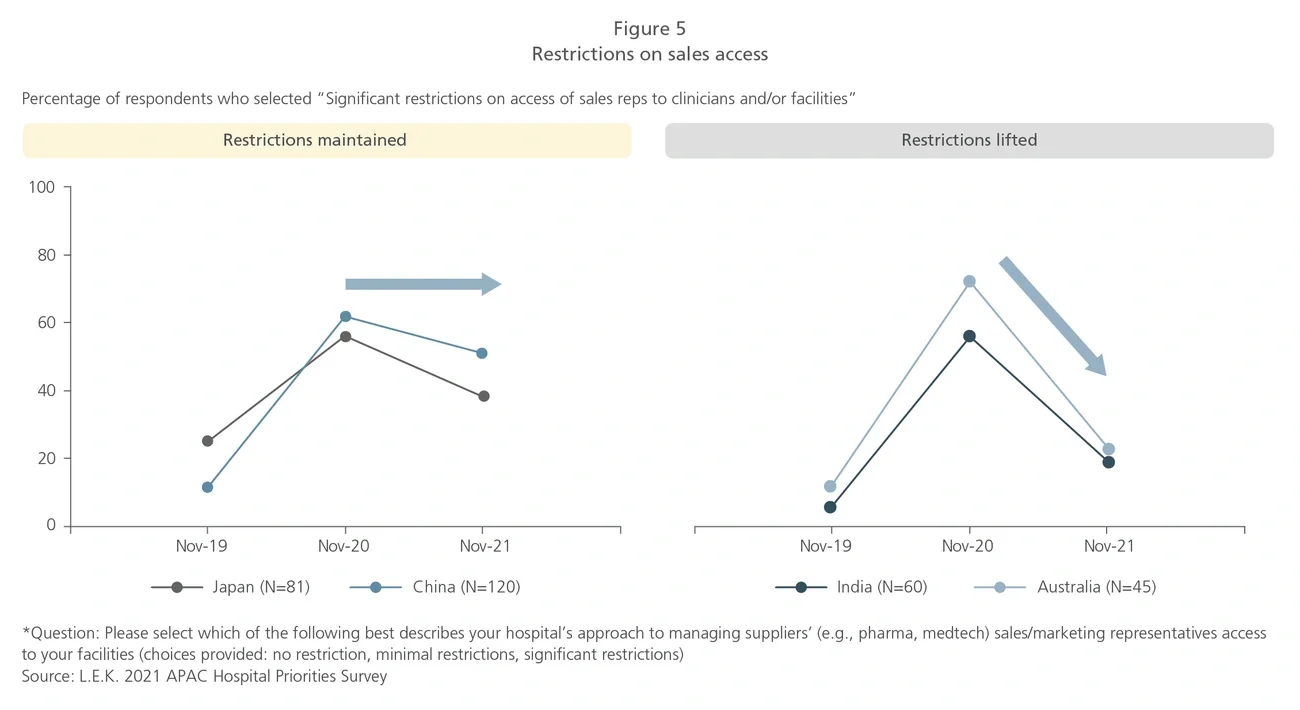
With the shift of hospital priorities towards emergency preparedness and financial recovery to manage the impact of COVID-19, medtech and pharma companies should expect greater scrutiny of economics and sensitivity on direct and immediate budgetary impacts compared to pre-COVID-19 times.
-
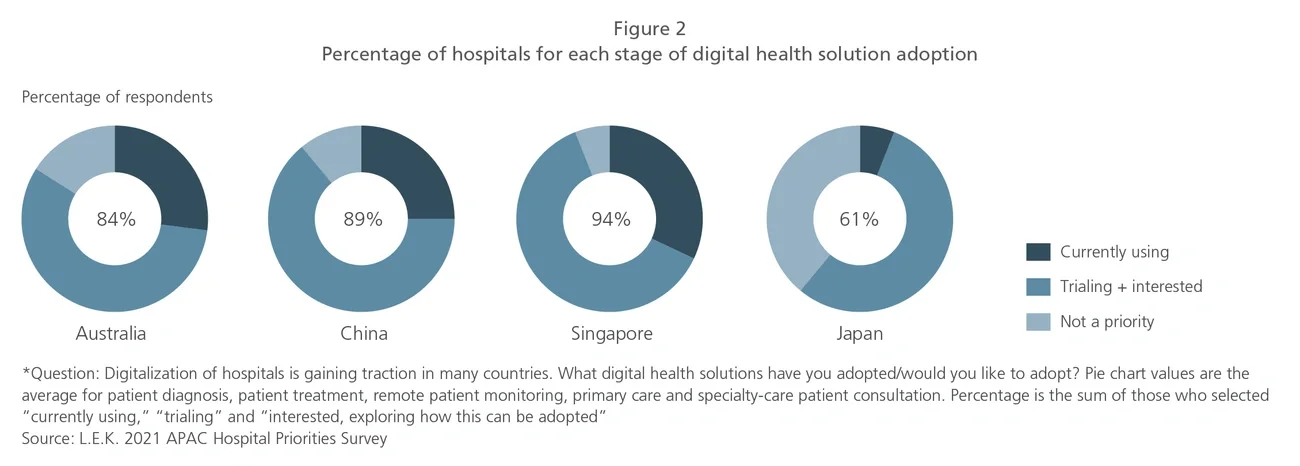
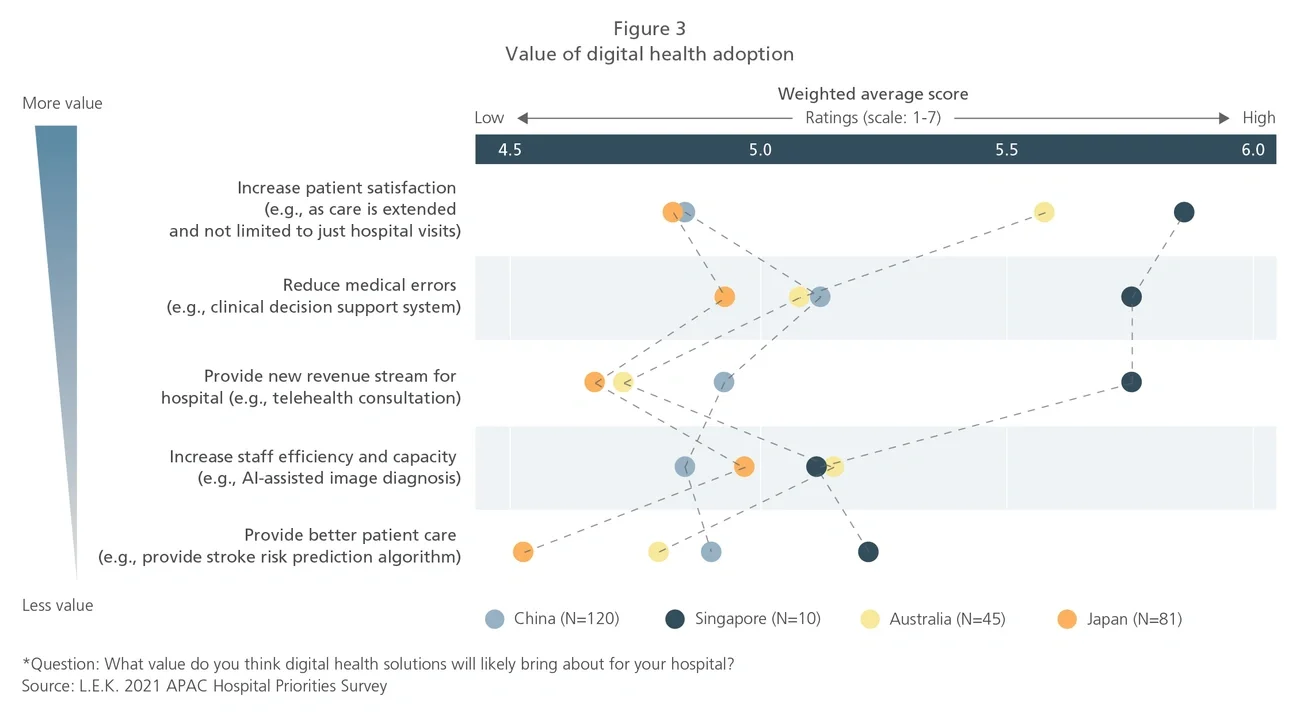
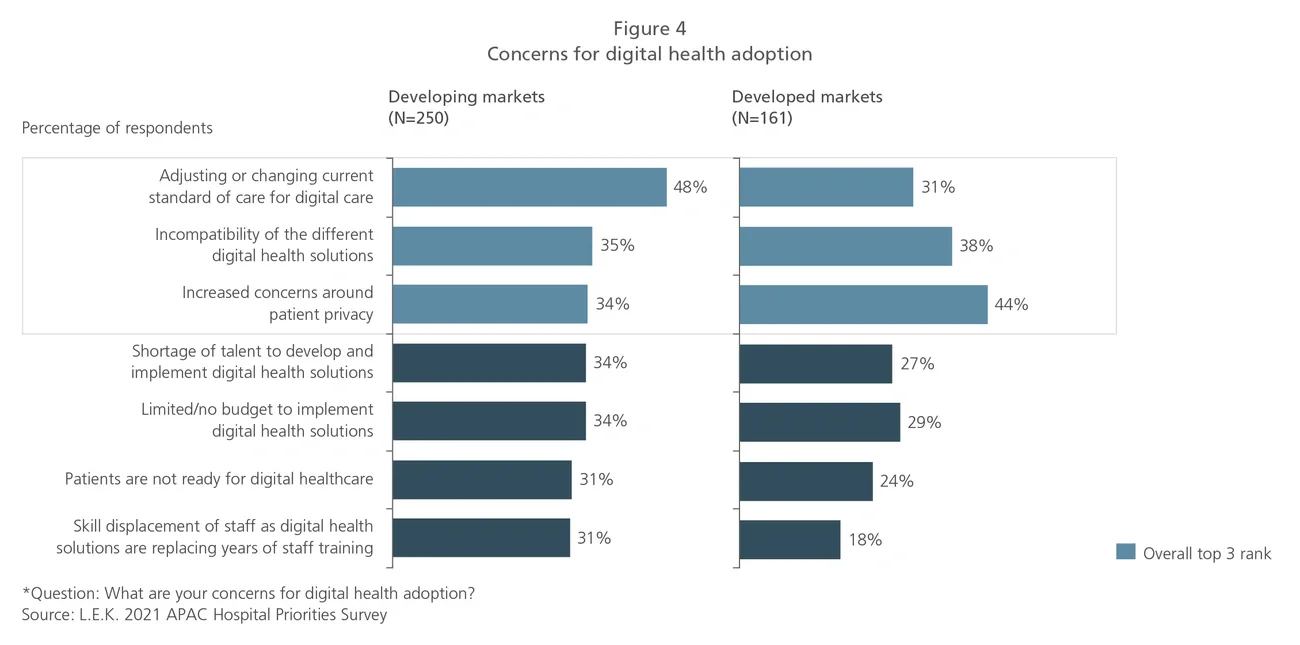
However, new opportunities arise for those who are able to quickly pivot their strategy and support hospitals with the acceleration in adoption of digital health solutions and digital communication tools.
-
The winners in the new era will be those that adapt to the needs of their customers and create win-wins during this unprecedented time.
L.E.K. Consulting conducted the 2021 APAC Hospital Priority Survey with 411 hospital executives across both public and private hospitals in countries1 across the Asia-Pacific (APAC) region. The fieldwork was executed in collaboration with GRG Health.2 This article aims to provide an overview and understanding of the rapidly changing landscape in which hospitals are operating and identifies key trends within the region. Insights are provided into how hospitals across the region have adapted to the impact of the COVID-19 pandemic, and how this has shaped their priorities for the future.
Given these insights, the following trends have emerged from the survey:
-
Shifting priorities to manage the impact of the pandemic
-
Accelerating adoption of digital health solutions to expand capacity and improve clinical decision-making
-
Leveraging digital channels to maintain engagement with physicians
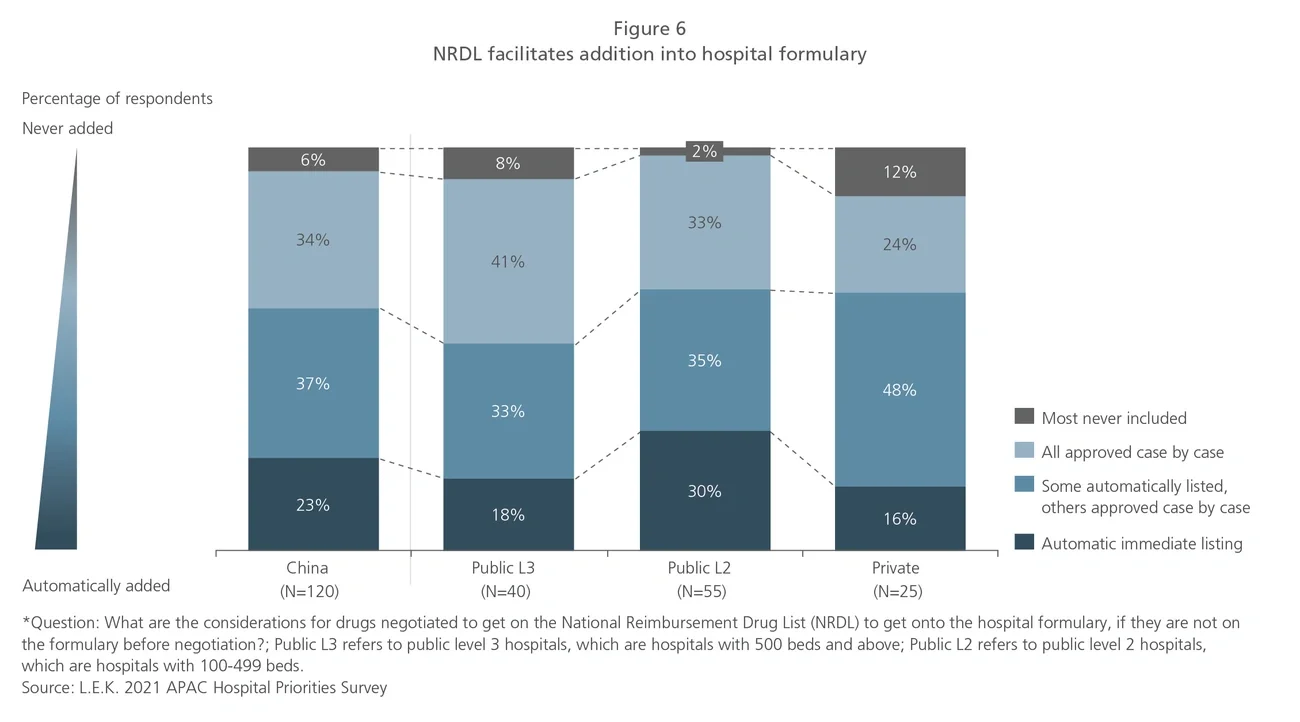
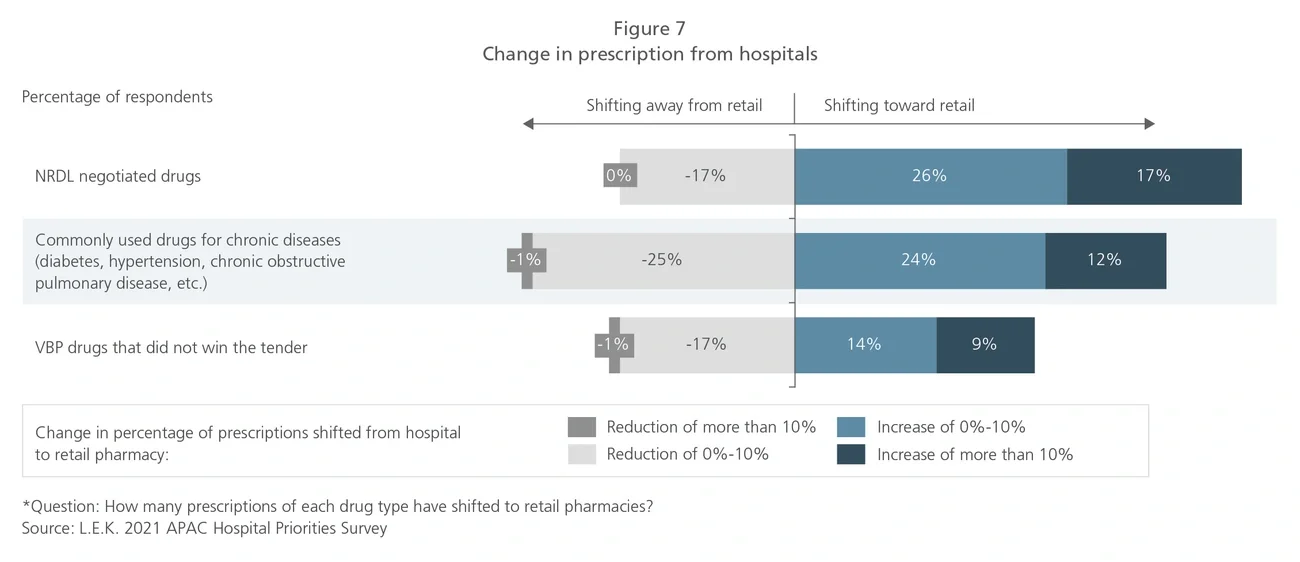
This article also features the cost containment efforts for pharma products through the use of VBP (volume-based procurement) and the NRDL (National Reimbursement Drug List) in China, and through the introduction of formulary guidelines in Japan.