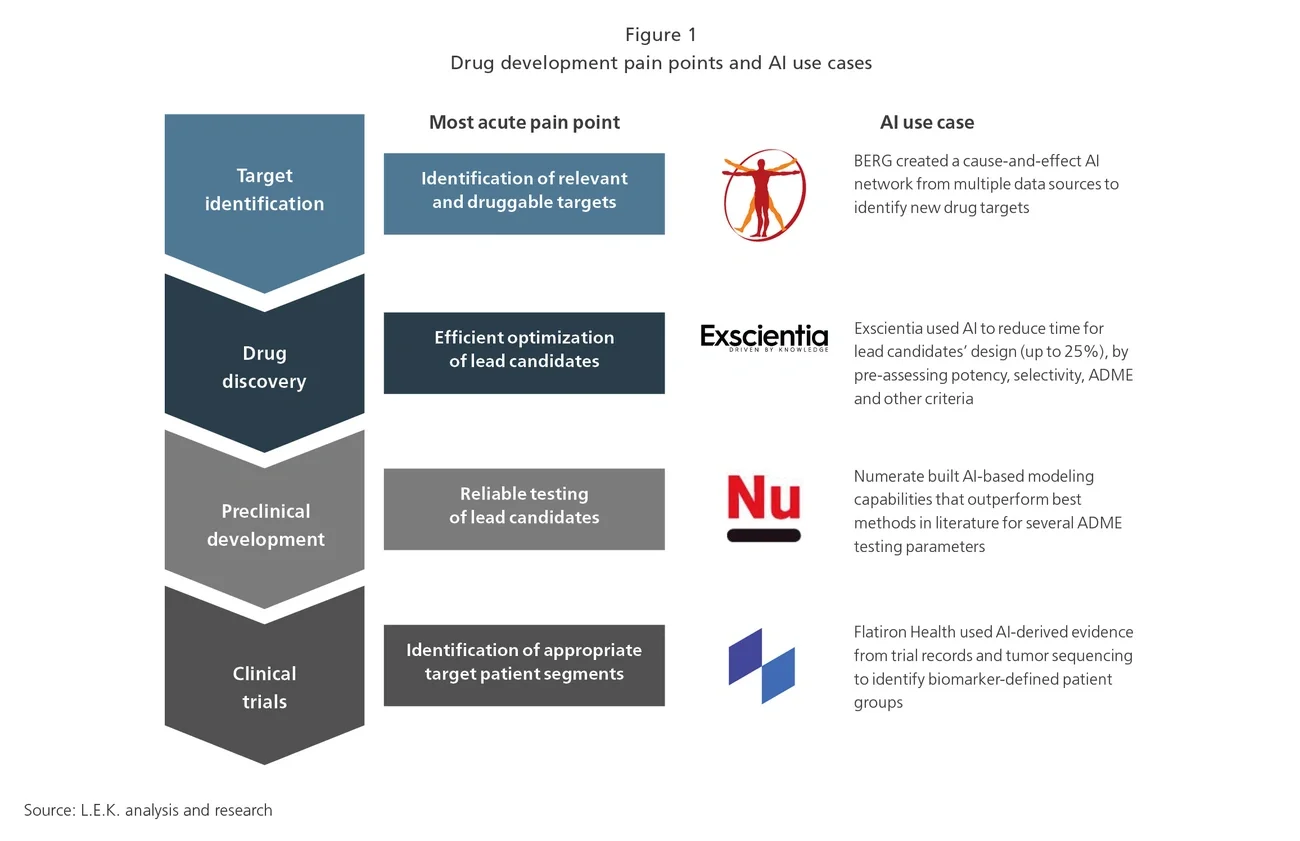
Biopharma tends to be concerned with failures at the clinical stage, with approximately 70% of the total drug development costs incurred in the clinic. However, failure at this stage is often caused by suboptimal compounds, so enhancing earlier stages of drug development should result in higher success rates, highlighting the importance of addressing inefficiencies across the drug development spectrum.
There is widespread recognition among biopharma companies that the R&D process is inefficient and that this inefficiency will keep growing if left unaddressed. This dynamic is partly driven by the increasingly complex nature of the biology underpinning the discovery of new molecules and increasing regulatory requirements. Other contributing factors include the facts that most attainable targets or drugs have already been used and that defining clinical trials is getting harder as patient populations need to be defined more precisely to show the desired effect.
AI has the potential to help curb this trend through promising innovative solutions that address drug development pain points, focusing on:
- Improving the quality of candidates (e.g., improved target validation and lead optimization, drug repurposing)
- Optimizing clinical trial design (e.g., biomarker based screening, patient stratification)
- Reducing time to complete activities (e.g., ADME1 parameter prediction, toxicology profiling, patient recruitment)
- Reducing costs (e.g., enhanced use of in silico database-trained methods, optimized data collection and analysis)
In addition to streamlining steps across the R&D process, AI solutions also have the potential to redefine the process itself by cutting out steps altogether, for example by moving from target to lead directly, or bypassing the hit optimization process.
Caution holds back adoption
To date, biopharma has been more cautious in adopting AI than other industries, with a number of real issues holding back progress.
First, there are unclear “real-world” benefits. Publicly documented use cases demonstrate the efficacy of AI under specific conditions at specific stages of development, but they often fail to define the tangible impact on R&D cost, timelines or overall probability of success. AI companies have also only recently started to publicly share compelling evidence of their ability to address drug development pain points.
Second, due to the complex nature of machine learning, interpreting AI insights can be challenging. Biopharma stakeholders often find it hard to understand the decision-making process underpinning an AI solution, resulting in a lack of buy-in to findings.
Third, the availability of relevant and readily usable biopharma proprietary data is limited, and it often takes significant time to clean, identify and extract the data required to enable AI. Information often resides in multiple locations and across functions, and the amount of data relevant to a given target or drug may be limited or hard to access.
Fourth, biopharma has an internal resistance to change, and the industry is known for its cautious nature. This is particularly true of R&D teams, which tend to be wary of new approaches given how much is at stake if things go wrong. This may be exacerbated by a lack of experience within the industry, as few biopharma stakeholders have the relevant expertise in AI to fully appreciate the potential and limitations of its application in drug development.
Finally, the AI landscape is evolving rapidly. New companies are emerging, and existing organizations are continuously evolving their propositions and business models. It is challenging for biopharma to “pick winners” at this nascent stage of market development.
Lowering the barriers
Despite the barriers to deployment, biopharma’s interest in AI remains high. The major players are either investing in AI technology or leading an AI-focused effort, alone or jointly with others, and face pressure not to fall behind the competition. AI providers are increasingly targeting their solutions to address specific industry needs, matching biopharma expectations and building trust by providing more precise, comprehensible and substantiated evidence.
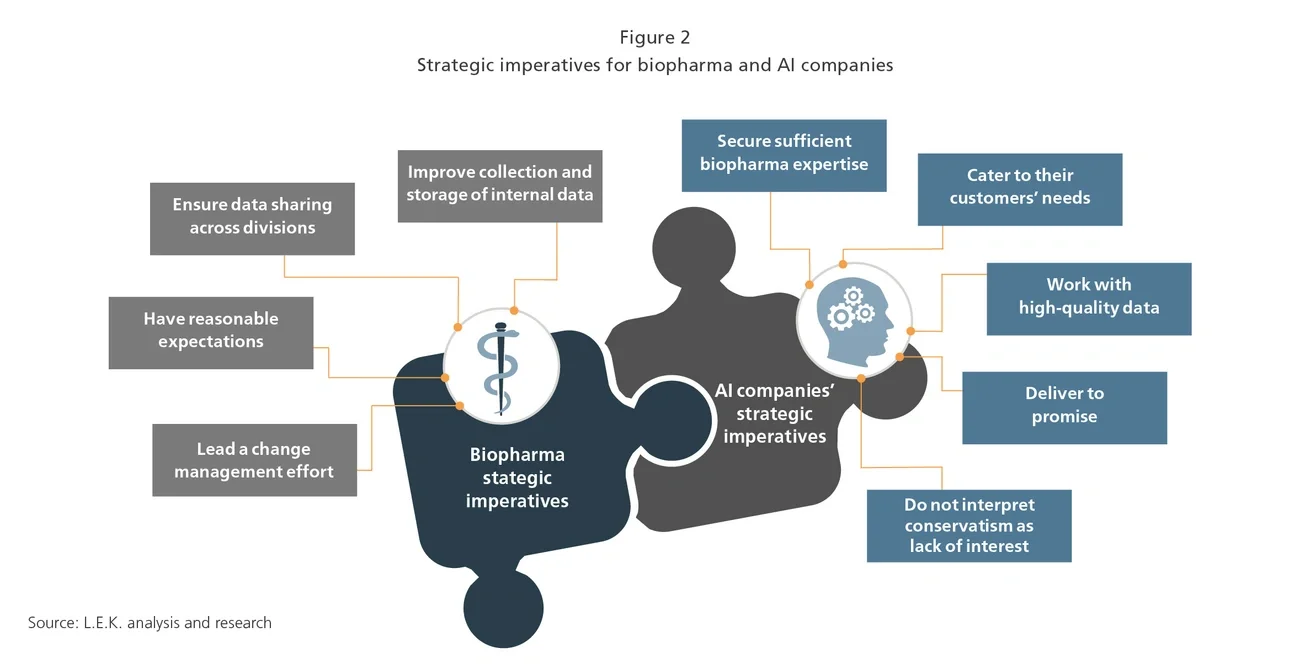
To accelerate the advance of AI, biopharma companies and AI providers can follow a few key principles to lower barriers to adoption (see Figure 2).