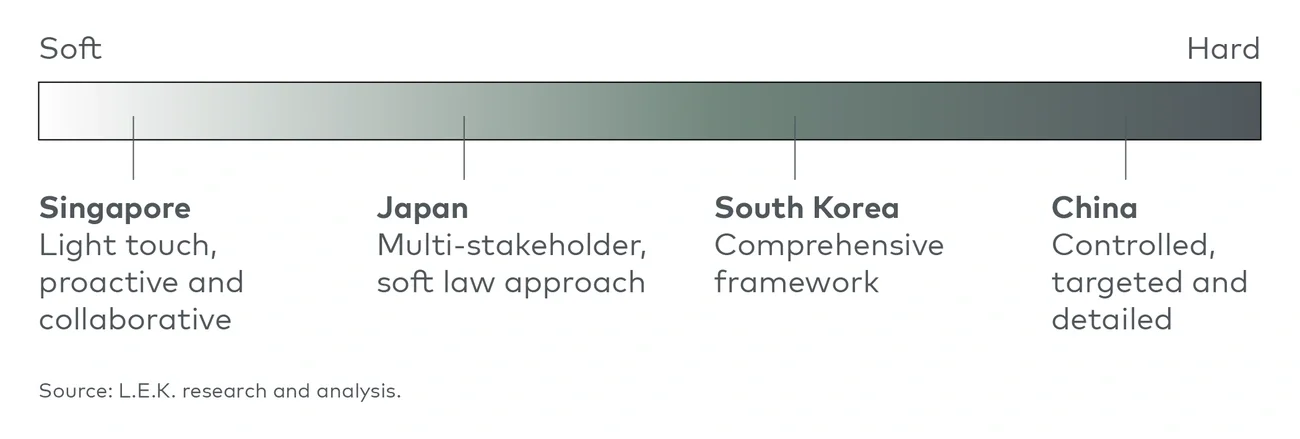
Singapore: Light, practical and gradual
The development and implementation of generative AI tools hold immense promise for addressing pressing challenges in Singapore’s healthcare sector, where government projections highlight an urgent need to expand the medical workforce by 40% by 2030 to support an aging population. Generative AI tools can attenuate the need for such a rapid surge in medical personnel.1
Singapore has taken a proactive approach to generative AI development. In June, the government published a discussion paper that presented an approach to regulation focused on the development of technical tools, standards and technology. Singapore’s approach to regulating generative AI has been collaborative, with an emphasis on voluntary measures.
Central to this approach has been the development of a governance testing framework called “AI Verify” that draws from global standards and principles. This framework is designed to enable companies to voluntarily assess and validate their AI systems.2 By bringing technology giants into this initiative, Singapore’s government aims to harness the collective expertise of the industry to develop robust AI standards and practices that can guide the responsible use of AI. The dedicated IT system of the Ministry of Health of Singapore, which rebranded in July as Synapxe, is a case in point, as it plays an important role in catalyzing the development and implementation of generative AI tools by incorporating the technology into the National Electronic Health Record system, launched in 2011 to act as a centralized archive for health records and aggregated data from healthcare providers.
Synapxe is working with Microsoft3 to enhance collaboration in generative AI and cloud innovation. A result of this collaboration is the Secure GPT for Healthcare Professionals system, based on the Azure OpenAI service. The system aims to put in place the infrastructure for the development of large language models and related healthcare applications, the automation of tasks and the efficient gathering of information.4 This partnership highlights the government’s openness and its proactive stance on working closely with industry leaders to cultivate innovative use cases and establish best practices for generative AI in healthcare.
Japan: Multi-stakeholder framework and guidance
Japan’s government is taking a sector-specific and soft law-based approach that promotes AI governance through nonbinding guidance,5 echoing strategies embraced by the U.K. and U.S.
In May, Japan established an AI strategy council of experts with a particular focus on generative AI, a major topic of discussion during the Japan-led G7 Hiroshima Summit and integral to the “G7 Hiroshima Process on Generative Artificial Intelligence” report. Despite the nonbinding nature of the country’s guidance, Japan has not been passive in regulating AI, implementing both general and sector-specific rules in areas such as transportation and finance.6
In the healthcare sector, Japan’s approach to generative AI is still taking shape. While specific regulations and guidance are yet to be unveiled, there has been notable mention of support for AI applications in the space, including in areas such as drug discovery and the use of software as a medical device.
There have already been some noteworthy developments in generative AI tools in Japan, including:
- Multiple homegrown generative AI tools that are already coming onto the scene to support doctors with everyday tasks such as generating patient summaries, anticipating complications and tracking prescriptions.
- The development of Al Fast Doctor, which used ChatGPT to develop LHTM-2, a specialized language model tailored to Japanese medical practices that has successfully passed Japanese medical exams and is aimed at assisting doctors by providing answers to common inquiries and aiding in diagnoses.7
- CalqKarte, another tool powered by ChatGPT, that is instrumental in recording and formatting clinical consultation notes within electronic medical records (EMRs), further streamlining administrative processes.8
Ultimately, the demand for generative AI tools in healthcare in Japan is driven by the need to alleviate workloads for healthcare providers, an issue exacerbated by the relatively slow adoption of technology to streamline administrative tasks.
Impending workplace reforms that will cap overtime hours for doctors starting in 2024 are also expected to be a key demand driver for innovative technology solutions.
South Korea: Comprehensive regulatory framework
South Korea has no regulations specific to generative AI, but the country’s approach to AI regulation mirrors that of the European Union in proposing an “AI Act” master bill. This legislation would consolidate various AI-related laws into a unified framework to regulate AI and establish ethical guidelines and a strategic policy roadmap. Some aspects of this approach support the development of generative AI.
One distinctive facet is an emphasis on corporate self-regulation rather than government regulation. In this, the government’s only role is to provide clear rules that ensure legal certainty and the protection of individuals.9
Within the proposed regulatory framework, there have been notable concerns around the vagueness of what constitutes “high-risk AI,” a category that has been applied to both healthcare and medical technology applications. It remains unclear how this determination is made and what systems will fall under the label. Legislation is complicated by an absence of clearly defined penalties for violations and the uncertainty surrounding enforcement. Some commentators have floated concerns about South Korea’s approach potentially hindering innovation and the country’s competitiveness on the global AI stage.
Nevertheless, South Korea’s thriving health data ecosystem is well positioned to provide a solid foundation for innovation and the development of generative AI tools. According to the Health Insurance Review and Assessment Service, South Korea boasts an impressive adoption rate of EMR systems, which are widely used as part of a near-universal digitization trend, encompassing the digital storage of clinical images and electronic hospital administration databases and the expanding use of remote sensor technology, making vast volumes of patient data readily accessible and analyzable.10
While South Korea enforces stringent legal protocols regarding patient data privacy and consent, updates to existing laws in 2020 allow for the sharing of de-identified or pseudonymized data, striking a delicate balance between fostering innovation and safeguarding patient privacy.
With a strong data infrastructure, a focus on corporate self-regulation and generating legal certainty, South Korea has set the stage for innovation in AI to flourish while avoiding stifling any advantages domestic companies might have. Companies like Naver Health and Kakao Brain, for example, are actively partnering with clinical institutions and university hospitals to facilitate the integration of generative AI into medical practice:
- As of May, Naver Health has been collaborating with a large domestic ophthalmic clinic to develop a multimodal chatbot service that can review medical images for diagnostic support via an AI vision correction prediction tool. Moreover, the company aims to introduce a “vision chat” service that leverages AI to analyze patient data alongside EMRs, facilitating more effective patient communication and care.11
- In December 2022, Kakao Brain completed joint research agreements with nine university hospitals propelling advancements in medical imaging through the development of AI models, which allowed it to develop a research AI web service to interpret chest X-rays that it plans to release in 2024.
China: Controlled development and targeted regulation
China is moving quickly to develop AI technologies and regulate their use. The country has rolled out a steady stream of regulations over the past two years, including the first set of rules specific to generative AI in July.
“We need to appreciate that China is ahead in regulating generative AI. China has taken the first and most ambitious steps at regulating AI in the world,” says Rebecca Arcesati, lead analyst at the Mercator Institute for China Studies in Berlin.
China’s interim measures on generative AI focus on services available to the public and showcase a deliberate effort to balance innovation with responsible usage.12 Regulations issued in final form in July turned out to be far less restrictive than an initial draft first circulated in March, reflecting an emphasis on industry development. Key changes included the removal of fines and an abandonment of real-name verification requirements. Instead, the regulations highlighted Beijing’s penchant for using regulations as a tool to both control technological development and foster innovation. These regulations do not extend to the application, research, development or internal use of generative AI services within organizations.
While the sector-specific approach in China is different from that of the EU, which has sought to cover a broader spectrum of AI,13 China’s regulations generally align with global standards and emphasize critical aspects of the technology, such as the quality of training data, bias mitigation, IP protection and privacy safeguards. A key difference in China, however, is a requirement that generative AI systems reflect “core socialist values,” which raises questions about the stability of the environment for foreign generative AI systems such as ChatGPT, which has been banned, and the potential censorship of AI systems, which could have implications for the global development of AI.
More practically, China has spread responsibility for the development of AI to different government departments, entrusting them to enhance rules within their respective industries. This industry-specific approach aims to give full play to specialized expertise and mirrors the approach being taken by the U.K.
However, while China’s national approach to generative Al regulation emphasizes growth, municipal governments also play a pivotal role in shaping the regulatory landscape. Certain municipal governments have taken measures that could introduce complexities into the development of generative Al. For instance, the Beijing government has formulated regulations that impose limits on the use of generative Al in healthcare, notably prohibiting its use for the automatic generation of medical prescriptions.14 As generative Al regulations evolve, companies must closely monitor not only national regulations in China but those at the provincial and city levels as well, as they could impact the feasibility and investment of generative Al applications in healthcare, influencing the development of the sector.
In healthcare specifically, China has aimed its regulatory focus at generative AI in applications available to the public. A logical next step would be for regulators to focus on non-patient-facing applications such as diagnosis chatbots or communication tools that aim to ease the burden on healthcare professionals. According to BMJ Open, a peer-reviewed, open-access medical journal, China faces a scarcity of doctors, ranking 77th globally in terms of doctors per capita, with pronounced rural-urban healthcare inequalities. Generative Al tools like Ping An Good Doctor, which aids doctors in handling patient inquiries,15 and MedGPT, an advanced chatbot and medical document platform,16 can play an important role in bridging this gap. These innovations alleviate the burden on healthcare professionals, who have long been strained due to rising patient numbers.
Next steps for healthcare and generative AI
Regulators are playing catch-up with the rapid advancements in generative AI and frameworks remain a work in progress. However, for businesses in generative AI and healthcare, this presents an opportune moment to gain firsthand experience, define a vision for generative AI and positively inform regulators’ perspectives on its use in healthcare.
To do this, companies must first develop an internal vision and governance structure for the use of generative AI, ensuring alignment with broader strategic goals. Additionally, the strategic implementation of smaller pilot-scale projects across various applications could provide invaluable insights into the benefits and risks associated with different use cases, mitigating regulatory risk. Furthermore, fostering partnerships and coalitions with industry players and associations is essential to building consensus and collectively influencing policy, thereby amplifying the impact of individual efforts in shaping regulation.
L.E.K. Consulting is a registered trademark of L.E.K. Consulting LLC. All other products and brands mentioned in this document are properties of their respective owners. © 2023 L.E.K. Consulting LLC
Endnotes
1Tan K., Strait Times, 2023
2Chiang, S., CNBC, June 2023
3Frontier Enterprise, July 2023
4Abdullah, Strait Times, 2023
5Nussey, S., Kelly, T., Reuters, July 2023
6Habuka, H., CSIS, February 2023
7Fast Doctor Co. Ltd., PR Times, 2023
8KandaQuantum Inc., PR Times, 2023
9Yulchon LLC, March 2023
10Digital Health South Korea, Intralinks, 2019
11ETNews, May 2023
12Wu, Y., China Briefing, July 2023
13Wu, Y., China Briefing, October 2022
14South China Morning Post, August 2023
15Shenshen Z., Shine, 2023
16MedCloud.PH, MedGPT, 2023