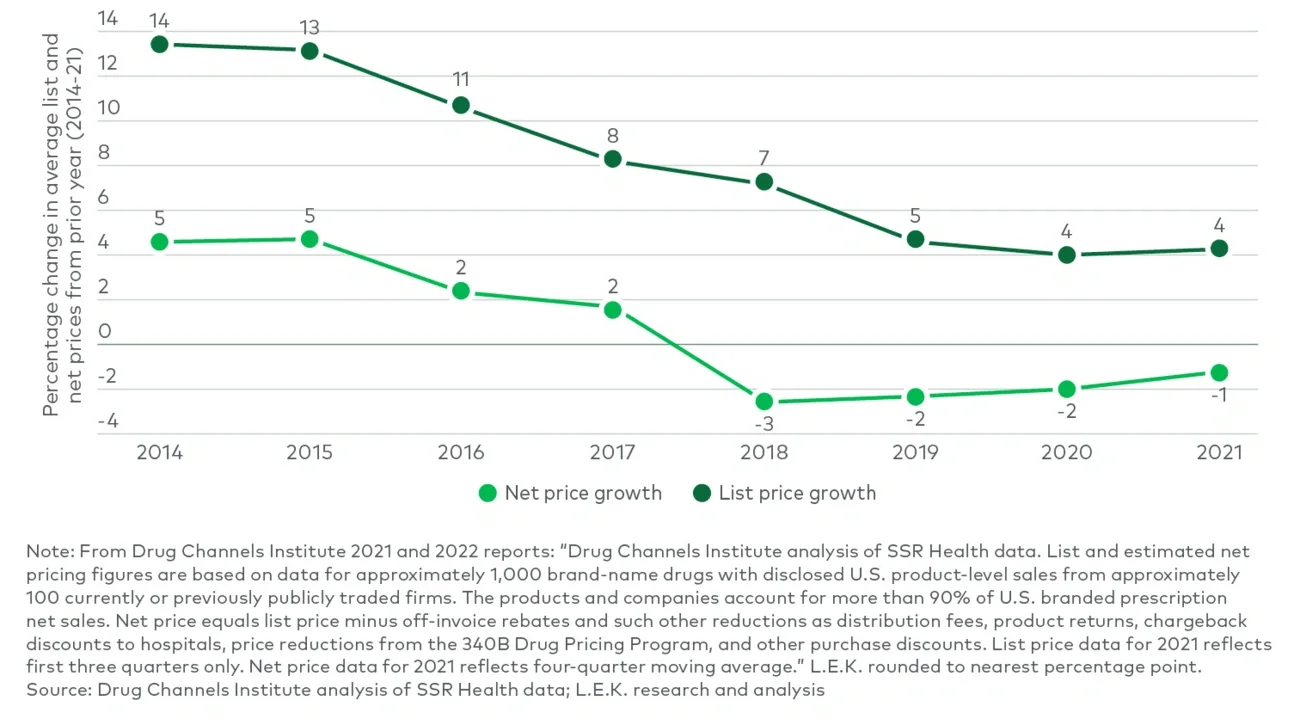
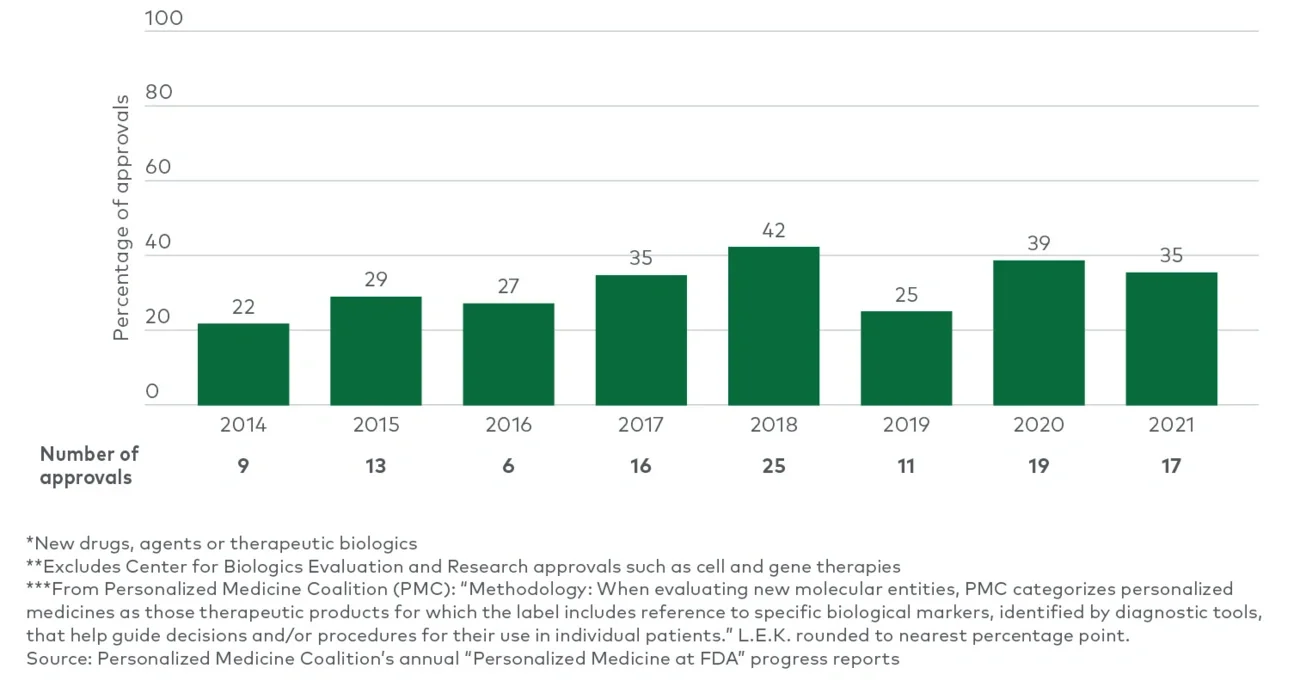
结论
综合来看,这些趋势正在推动整个医疗生态系统的根本性转变。为了在不断变化的市场环境中获胜,行业领导者必须对这些趋势进行监测,并调整其公司战略,以做出与组合投资、新产品上市策略、融资以及如何与行业参与者展开合作等方面相关的决策。L.E.K.一直以来都在密切关注所有关键趋势的发展,能够帮助广大生物制药公司有效解决其战略需求。
如欲了解更多,请联系lifesciences@lek.com。
尾注
1The New York Times. “Tracking Coronavirus Vaccinations Around the World.” Accessed Feb. 11, 2022. https://www.nytimes.com/interactive/2021/world/covid-vaccinations-tracker.html
2Intellia Therapeutics. “Intellia and Regeneron Announce Landmark Clinical Data Showing Deep Reduction in Disease-Causing Protein After Single Infusion of NTLA-2001, an Investigational CRISPR Therapy for Transthyretin (ATTR) Amyloidosis.” https://ir.intelliatx.com/news-releases/news-release-details/intellia-and-regeneron-announce-landmark-clinical-data-showing
3Nature Reviews Drug Discovery. “No shortage of fresh IPO capital in 2021.” https://www.nature.com/articles/d41573-022-00018-0
4Alliance for Regenerative Medicine. “Regenerative Medicine in 2021: A Year of Firsts and Records.” http://alliancerm.org/wp-content/uploads/2021/08/ARM-H1-2021-Report.pdf
5Alliance for Regenerative Medicine. “Regenerative Medicine: New Paradigms.” https://alliancerm.org/wp-content/uploads/2022/01/SOTI-Presentation-FINAL.pdf
6Gilead Sciences. “Yescarta® CAR T-Cell Therapy Quadruples Median Event-Free Survival Duration Over Standard of Care in Second-Line Relapsed or Refractory Large B-Cell Lymphoma.” https://investors.gilead.com/news-releases/news-release-details/yescartar-car-t-cell-therapy-quadruples-median-event-free
7Bristol Myers Squibb. “U.S. Food and Drug Administration (FDA) Accepts for Priority Review Bristol Myers Squibb’s Supplemental Biologics License Application for Breyanzi (lisocabtagene maraleucel) as a Second-Line Therapy for Relapsed or Refractory Large B-cell Lymphoma. https://news.bms.com/news/corporate-financial/2022/U.S.-Food-and-Drug-Administration-FDA-Accepts-for-Priority-Review-Bristol-Myers-Squibbs-Supplemental-Biologics-License-Application-for-Breyanzi-lisocabtagene-maraleucel-as-a-Second-Line-Therapy-for-Relapsed-or-Refractory-Large-B-cell-Lymphoma/default.aspx
8bluebird bio. “bluebird bio Submits Biologics License Application (BLA) to FDA for betibeglogene autotemcel (beti-cel) Gene Therapy for Patients With ß-Thalassemia Who Require Regular Red Blood Cell Transfusions.” https://investor.bluebirdbio.com/news-releases/news-release-details/bluebird-bio-submits-biologics-license-application-bla-fda
9bluebird bio. “bluebird bio Announces FDA Priority Review of Biologics License Application for eli-cel Gene Therapy for Cerebral Adrenoleukodystrophy (CALD) in Patients Without a Matched Sibling Donor.” https://investor.bluebirdbio.com/news-releases/news-release-details/bluebird-bio-announces-fda-priority-review-biologics-license-0
10BioMarin Pharmaceutical. “BioMarin Announces Stable and Durable Annualized Bleed Control in the Largest Phase 3 Gene Therapy Study in Adults with Severe Hemophilia A; 134-Participant Study Met All Primary and Secondary Efficacy Endpoints at Two Year Analysis.” https://investors.biomarin.com/2022-01-09-BioMarin-Announces-Stable-and-Durable-Annualized-Bleed-Control-in-the-Largest-Phase-3-Gene-Therapy-Study-in-Adults-with-Severe-Hemophilia-A-134-Participant-Study-Met-All-Primary-and-Secondary-Efficacy-Endpoints-at-Two-Year-Analysis
11Vertex Pharmaceuticals, accessed on Seeking Alpha. “Vertex Pharmaceuticals Incorporated’s (VRTX) CEO Reshma Kewalramani Presents at 40th Annual JPMorgan Healthcare Conference — Transcript.” https://seekingalpha.com/article/4478807-vertex-pharmaceuticals-incorporatedsvrtx-ceo-reshma-kewalramani-presents-40th-annual
12CSL Behring. “uniQure and CSL Behring Announce Primary Endpoint Achieved in Hope-B Pivotal Trial of ETRANACOGENE DEZAPARVOVEC Gene Therapy in Patients with Hemophilia B.” https://www.cslbehring.com/newsroom/2021/hope-b-gene-therapy-for-hemophilia-b-topline-results
13Alnylam Pharmaceuticals. “40th Annual J.P. Morgan Healthcare Conference.” https://alnylampharmaceuticalsinc.gcs-web.com/static-files/a152a390-748b-46df-92bc-5c064c0bad3c
14Ionis Pharmaceuticals. “Corporate Overview.” https://ir.ionispharma.com/static-files/ebc240f3-749c-436c-93cd-53a03ae8c425. “AstraZeneca and Ionis sign deal to develop and commercialise eplontersen.” https://www.astrazeneca.com/media-centre/press-releases/2021/astrazeneca-ionis-to-collaborate-on-eplontersen.html
15Pharmaceutical Technology. “Mapping the RNA Therapeutics R&D Landscape in 2022.” https://www.pharmaceutical-technology.com/analysis/mapping-the-rna-therapeutics-rd-landscape-in-2022/
16Chemical and Engineering News. “RNA-Editing Race Intensifies as Big Pharma Buys In.” https://cen.acs.org/pharmaceuticals/drug-discovery/RNA-editing-race-intensifies-Big/99/i39
17Drug Discovery Today. “The Rise and Rise of Protein Degradation: Opportunities and Challenges Ahead.” https://www.sciencedirect.com/science/article/pii/S1359644621003640
18Informa Pharma Intelligence. Citeline. “Pharmaprojects.” Accessed January 2022. https://citeline.informa.com/; Query: “Mechanism Of Action is Protein degrader” and “Global Status is Pipeline”.
19Drug Channels. “Tales of the Unsurprised: Brand-Name Drug Prices Fell for the Fourth Consecutive Year.” https://www.drugchannels. net/2022/01/tales-of-unsurprised-brand-name-drug.html; Drug Channels. “Surprise! Brand-Name Drug Prices Fell (Again) in 2020.” https://www.drugchannels.net/2021/01/surprise-brand-name-drug-prices-fell.html
20Centers for Medicare & Medicaid Services. “CMS Proposes Medicare Coverage Policy for Monoclonal Antibodies Directed Against Amyloid for the Treatment of Alzheimer’s Disease.” https://www.cms.gov/newsroom/press-releases/cms-proposes-medicare-coverage-policy-monoclonal-antibodies-directed-against-amyloid-treatment
21Business Insider. “Investors Aren’t Buying EQRx’s Pitch for ‘Radically Lower’ Drug Prices. The CEO of the $3 Billion Startup Tells Us Why She’s Doubling Down Despite a Stock Drop.” https://www.businessinsider.com/eqrx-ceo-on-spacs-drug-pricing-and-2022-growth-plans-2022-1
22EQRx. “Progress and Pipeline.” Accessed Feb. 13, 2022. https://www.eqrx.com/progress-and-pipeline/#pipeline
23EQRx. “New Pharma — 40th Annual J.P. Morgan Healthcare Conference Presentation.” https://investors.eqrx.com/static-files/9a9e69da-28f3-4b5e-9386-3fd5dd7f8c04
24EQRx. “EQRx presentation delivered at the 40th Annual J.P. Morgan Healthcare Conference on Monday, January 10, 2022 at 3:45 PM.” https://investors.eqrx.com/static-files/9de5fee9-6b1a-4355-8084-ea57fa7927d6
25Secretary Xavier Becerra, U.S. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. “Comprehensive Plan for Addressing High Drug Prices.” https://aspe.hhs.gov/sites/default/files/2021-09/Competition%20EO%2045-Day%20Drug%20Pricing%20Report%209-8-2021.pdf
26U.S. Department of Health and Human Services. “Biden-Harris Administration Improves Transparency and Oversight of Prescription Drug and Medical Costs.” https://www.hhs.gov/about/news/2021/11/17/biden-harris-administration-improves-transparency-and-oversight-of-prescription-drug-and-medical-costs.html
27The White House. “President Biden Announces Prescription Drug Pricing Plan in Build Back Better Framework.” https://www.whitehouse.gov/briefing-room/statements-releases/2021/11/02/president-biden-announces-prescription-drug-pricing-plan-in-build-back-better-framework/
28Covington. Inside EU Life Sciences. “New German Government Plans Significant Changes with Impact on the Healthcare, Life Sciences and Food Sector.” https://www.insideeulifesciences.com/2021/11/29/new-german-government-plans-significant-changes-with-impact-on-the-healthcare-life-sciences-and-food-sector/
29FiercePharma. “Japan Sees ‘Drug Lag’ as Foreign Pharmas Pass Up the Market amid Pricing Pressure, Industry Group Warns.” https://www.fiercepharma.com/pharma-asia/japan-sees-drug-lag-as-foreign-pharmas-skip-market-amid-pricing-pressure-industry-group
30Pharma DJ. “China’s NRDL Explained: 67 New Drugs, 62% Price Cut, 80% Success Rate and $47,000 Annual Cap.” http://www.pharmadj.com/en/cms/detail.htm?item.id=1abe2390597111ecbee6fa163e42049a
31GlobalData Healthcare, Pharmaceutical Technology. “Virtual Sales Reps’ and Physicians’ Engagement to Remain a New Standard Post- Covid-19.” https://www.pharmaceutical-technology.com/comment/virtual-engagement-covid-pharmaceutical-companies/
32FiercePharma. “Amgen Cuts 500 U.S. Jobs, Primarily Sales Reps, as Covid-19 Speeds a Shift to Digital.” https://www.fiercepharma.com/marketing/amgen-cuts-several-hundred-u-s-jobs-primarily-sales-reps-as-covid-19-speeds-a-shift-to#
33Reuters. “Exclusive: Pfizer to Cut U.S. Sales Staff as Meetings with Healthcare Providers Move to Virtual.” https://www.reuters.com/business/healthcare-pharmaceuticals/exclusive-pfizer-cut-us-sales-staff-meetings-move-virtual-2022-01-11/
34American Medical Association. “Policy Research Perspectives: Recent Changes in Physician Practice Arrangements: Private Practice Dropped to Less Than 50 Percent of Physicians in 2020.” https://www.ama-assn.org/system/files/2021-05/2020-prp-physician-practice-arrangements.pdf
35The Wall Street Journal. “Novartis Rethinks Sales Strategy for New Cholesterol Drug Launch.” https://www.wsj.com/articles/novartis-rethinks-sales-strategy-for-new-cholesterol-drug-launch-11637510400
36FiercePharma. “JPM: Novartis Teams up with NHS England in ‘World-First’ Access Deal for PCSK9 Hopeful Inclisiran.” https://www.fiercepharma.com/pharma/novartis-teams-up-nhs-england-world-first-access-agreement-for-inclisiran
37Novartis Pharmaceuticals. “Kymriah® Treatment Center Locator.” https://www.us.kymriah.com/treatment-center-locator/ Kite Pharma, A Gilead Company. “Find a Large B-Cell Lymphoma YESCARTA® Authorized Treatment Center.” https://www.yescarta.com/lbcl/ find-a-treatment-center/ Spark Therapeutics. “Patient Support Services.” https://mysparkgeneration.com/patient-support.html
38Defined as those with potential for at least 18 months of treatment effects; based on cell and gene therapy pipeline from December 2019.
39NEWDIGS FoCUS Project. MIT. “Updated Projection of US Durable Cell and Gene Therapies Product-Indication Approvals Based on December 2019 Development Pipeline.” https://newdigs.mit.edu/sites/default/files/NEWDIGS-Research-Brief-2020F207v51-PipelineAnalysis.pdf
40The Personalized Medicine Coalition. “The Personalized Medicine Report — 2020: Opportunity, Challenges, and the Future.” https://www.personalizedmedicinecoalition.org/Userfiles/PMC-Corporate/file/PMC_The_Personalized_Medicine_Report_Opportunity_Challenges_and_the_Future.pdf
The Personalized Medicine Coalition. “Personalized Medicine at FDA: The Scope & Significance of Progress in 2020.” https://www.personalizedmedicinecoalition.org/Userfiles/PMC-Corporate/file/PM_at_FDA_The_Scope_Significance_of_Progress_in_2020.pdf
The Personalized Medicine Coalition. “Personalized Medicine at FDA: A Progress & Outlook Report.” https://www.personalizedmedicinecoalition.org/Userfiles/PMC-Corporate/file/PM_at_FDA_The_Scope_and_Significance_of_Progress_in_2019.pdf
The Personalized Medicine Coalition. “Personalized Medicine at FDA: A Progress & Outlook Report.” https://www.personalizedmedicinecoalition.org/Userfiles/PMC-Corporate/file/PM_at_FDA_A_Progress_and_Outlook_Report.pdf
The Personalized Medicine Coalition. “Personalized Medicine at FDA: 2017 Progress Report.” https://www.personalizedmedicinecoalition.org/Userfiles/PMC-Corporate/file/PM_at_FDA_2017_Progress_Report.pdf
The Personalized Medicine Coalition. “Personalized Medicine at FDA: 2016 Progress Report.” https://www.personalizedmedicinecoalition.org/userfiles/pmc-corporate/file/pm-at-fda.pdf
The Personalized Medicine Coalition. “Personalized Medicine at FDA: 2015 Progress Report.” https://www.personalizedmedicinecoalition.org/Userfiles/PMC-Corporate/file/2015_Progress_Report_PM_at_FDA.pdf
41Exact Sciences. “Multi-Cancer Early Detection Blood Test.” https://www.exactsciences.com/Pipeline-and-Data/Multi-Cancer-Early-Detection
42GRAIL. “Grail Confirms Q2 2021 Introduction of Galleri, First-of-Kind Multi-Cancer Early Detection Blood Test.” https://grail.com/press-releases/grail-confirms-q2-2021-introduction-of-galleri-first-of-kind-multi-cancer-early-detection-blood-test/
43Natera. “Natera Receives Final Medicare Coverage for Its Signatera™ MRD Test in Stage II-III Colorectal Cancer.” https://www.natera.com/company/news/natera-receives-final-medicare-coverage-for-its-signatera-mrd-test-in-stage-ii-iii-colorectal-cancer/
44Being Patient. “New Era for Alzheimer’s Trials: Blood Tests Bring Younger Participants.” https://www.beingpatient.com/c2n-alzheimers-bloodtest-in-clinical-trials/
45BioPharma Dive. “As Three Biotechs Head to Wall Street, a Battered Sector Braces for a Pullback.” https://www.biopharmadive.com/news/biotech-ipo-performance-investors-2021/616808/
46Nasdaq. “2021 Year in Review: What’s New with the Innovators of the Nasdaq Biotechnology Index?” https://www.nasdaq.com/articles/2021-year-in-review-whats-new-with-the-innovators-of-the-nasdaq-biotechnology-index
47Evaluate Vantage, Evaluate. “Evaluate Vantage 2022 Preview.” https://info.evaluate.com/rs/607-YGS-364/images/Vantage%202022%20Preview%20Report_Final.pdf
48HKTDC Research. “Biotechnology, Medical & Healthcare Industry in Hong Kong.” https://research.hktdc.com/en/article/MzEzOTQ1MjMz